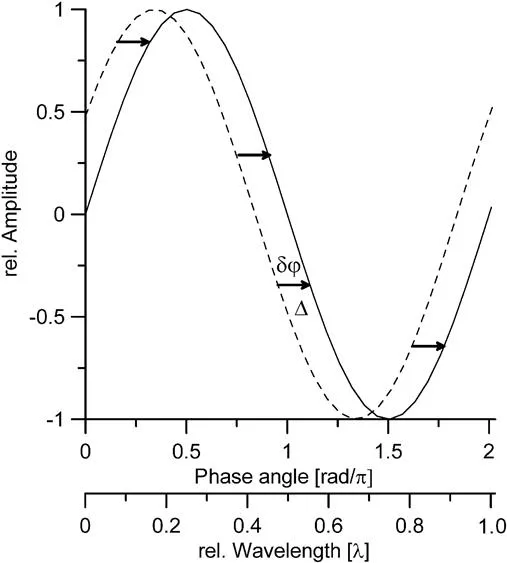
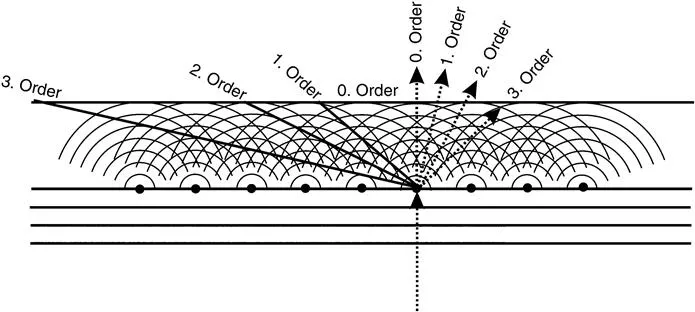
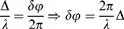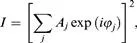Powder Diffraction
Theory and Practice
R E Dinnebier, S J L Billinge, Peter G Bruce, R E Dinnebier, S J L Billinge
- 604 pagine
- English
- ePUB (disponibile sull'app)
- Disponibile su iOS e Android
Powder Diffraction
Theory and Practice
R E Dinnebier, S J L Billinge, Peter G Bruce, R E Dinnebier, S J L Billinge
Informazioni sul libro
Powder diffraction is a widely used scientific technique in the characterization of materials with broad application in materials science, chemistry, physics, geology, pharmacology and archaeology. Powder Diffraction: Theory and Practice provides an advanced introductory text about modern methods and applications of powder diffraction in research and industry. The authors begin with a brief overview of the basic theory of diffraction from crystals and powders. Data collection strategies are described including x-ray, neutron and electron diffraction setups using modern day apparatus including synchrotron sources. Data corrections, essential for quantitative analysis are covered before the authors conclude with a discussion of the analysis methods themselves. The information is presented in a way that facilitates understanding the information content of the data, as well as best practices for collecting and analyzing data for quantitative analysis. This long awaited book condenses the knowledge of renowned experts in the field into a single, authoritative, overview of the application of powder diffraction in modern materials research. The book contains essential theory and introductory material for students and researchers wishing to learn how to apply the frontier methods of powder diffraction
Domande frequenti
Informazioni
Principles of Powder Diffraction
1.1 INTRODUCTION
1.2 FUNDAMENTALS