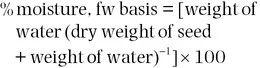![]()
1 Seed Storage, Germination, Quality, and Enhancements
Alan G. Taylor
School of Integrative Plant Science, Horticulture Unit, Cornell AgriTech, Cornell University, Geneva, New York 14456, USA
Before vegetables are harvested, before their growth and development, even before the seedling becomes photosynthetically competent, the source of the vegetable—the seed—holds major keys to final product yield. To fully comprehend the complexities of vegetable crop growth and development, we must understand seed physiology related to storage, germination, quality, and enhancements. The challenge is to present an in-depth knowledge of vegetable seeds that covers considerable diversity with respect to botanical classification, seed size, and composition. This chapter and book encompasses 33 common vegetable crop seeds from ten plant families (Table 1.1). This diverse group of plants makes a comprehensive overview of vegetable seeds difficult to achieve, especially since there is little scientific literature on seed physiology of many small-seeded crops of minor economic importance. Due to these limitations, we enlist several approaches to develop a coherent picture of vegetable seeds. Relative differences between seeds are shown with regard to seed size and composition, storage longevity, seed and seedling morphology, temperature requirements for germination, and the use of reserve materials during emergence. More generalized information is presented on factors and events associated with storage, germination, and aging. Finally, the process of aging and other aspects of seed physiology and technology are illustrated using specific crop examples.
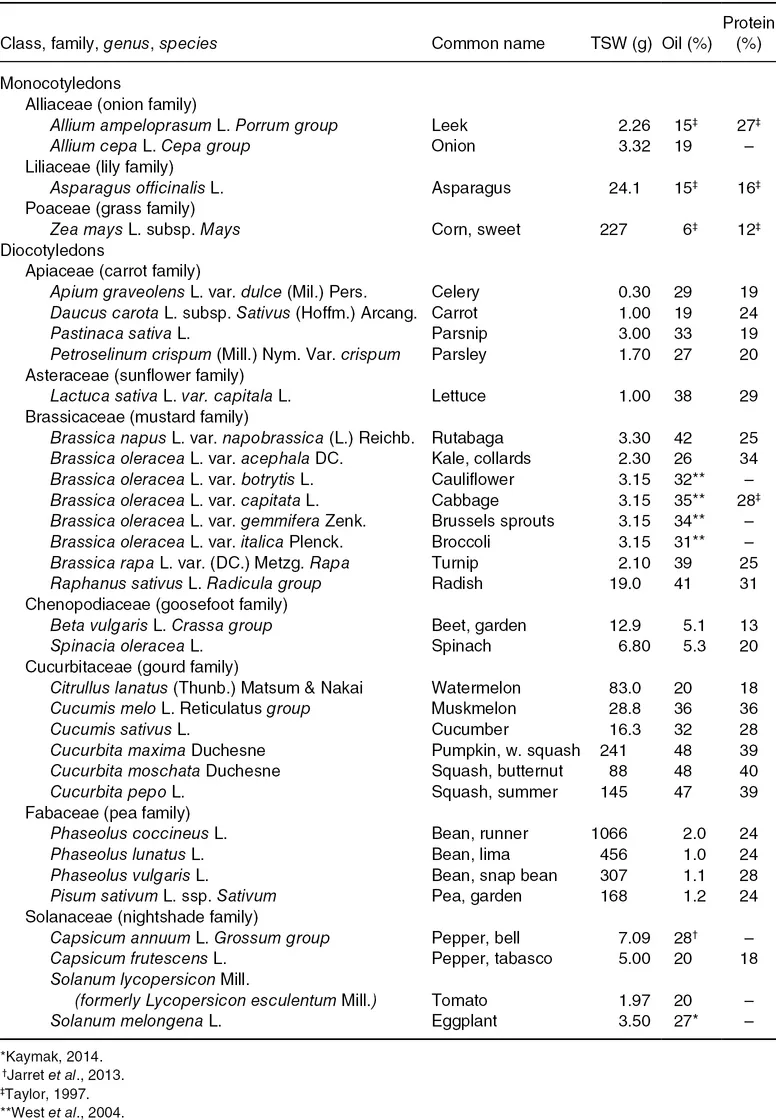
Table 1.1. Botanical and common names (Maynard and Hochmuth, 2007). Thousand seed weight (TSW), percent oil and protein content from representative seed samples (Royal Botanic Gardens Kew, 2017, except where noted).
The focus of this chapter is on postharvest aspects of seeds; the period of seed development and production stages are not addressed. We start with storage of seeds with low moisture content as most vegetable crop seeds have unique characteristics that permit them to withstand desiccation (Leopold and Vertucci, 1986). Desiccation tolerance is essential for long-term survival and allows for a time interval between seed production and crop production. Most vegetable seeds imbibe water readily and, provided with a suitable environment, will germinate and resume active growth. The seed uses its reserve materials following germination and then becomes an active photosynthetic seedling, fixing its own carbon and producing energy.
Seed quality is a broad term and encompasses several attributes of seeds including the germination and seedling performance. In this chapter, certain physiological and biochemical processes associated with loss of seed quality are described, and symptoms of seed aging are presented at physiological and whole-plant levels. The sowing environment has a direct effect on germination and stand establishment, and under severe stress, seedling performance is seriously impaired. To ensure stand establishment, high quality seeds are needed for transplant production and for direct seeding. Seed performance is improved by seed enhancements to achieve maximum emergence when sown in suboptimal conditions. In addition, seed-coating technologies are used to improve precision placement of seeds for sowing, and to provide a delivery system for compounds and agents to both protect and enhance seed and plant performance.
Composition and Water Status in Seeds
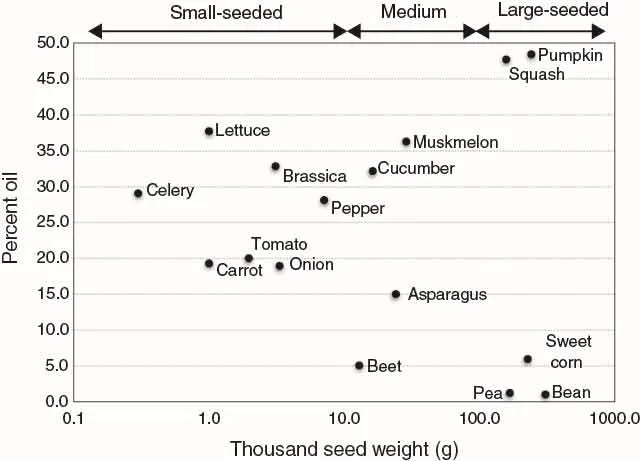
The composition of seeds is important to many aspects of seed physiology, and the percent oil or lipid content, and percent protein, are provided for the 33 common vegetable crop seeds in Table 1.1. Seed size expressed as thousand seed weight (TSW) was calculated; small-seeded vegetables have a TSW of < 10 g, while large-seeded vegetable crops have TSW > 100 g. Intermediate or medium-sized seeds range from 10 to 100 g TSW. The relationship of TSW and percent seed oil content is shown in Fig. 1.1.
There are two groups of seeds based on their storage reserves: starch and lipid storing. Seeds with less than 10% oil are starch-storing seeds, and the starch content of snap bean, pea and sweet corn is 42%, 48%, and 53%, respectively (cited by Taylor, 1997). Table beet seeds contain starch, which is stored in the perisperm (see “Seed and seedling morphology” section). All small-seeded crops (TSW < 10g) are oil-storing seeds with > 18% oil. Though large-sized seeds generally store starch, the large-seeded cucurbits are exceptions with nearly half of their weight as lipids. All seeds contain proteins, and values range between 12% and 40% (Table 1.1).
Seeds in storage are said to be in a dry condition; however, “dry” is a relative term and does not mean that water is absent from seeds. Water is present in seed tissue, and the status of water is related to many aspects of seed physiology, including seed longevity. First, the concentration of water is measured and expressed in meaningful units.
Standardized gravimetric methods to determine the moisture content of seeds were published by the Association of Official Seed Analysts (Elias et al., 2018). Other primary and secondary methods were described and reviewed by Grabe (1989). Seed moisture content calculated on a wet or fresh weight (fw) basis is commonly used in seed testing and in commerce. The following formula is used to determine the percentage of seed moisture content on a fresh weight basis:
The seed moisture content expressed on a fresh weight basis provides a direct assessment of the concentration of water in any quantity of seed. For example, 100 grams of seeds with 10% moisture content contains 10 grams of water and 90 grams of seed dry weight. Another unit to express seed moisture content is g H2O g−1 fresh or dry weight.
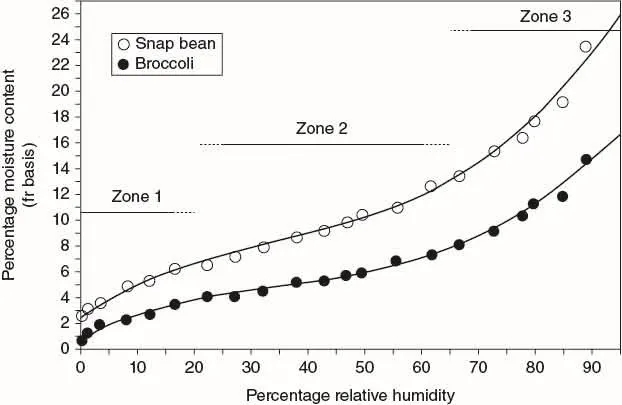
The seed moisture content comes into equilibrium with the relative humidity of the air. The relationship between the equilibrium moisture content and relative humidity reveals a negative sigmoidal-shaped curve known as a moisture isotherm (Iglesias and Chirife, 1982). This relationship is determined by placing seeds in a range of known relative humidity conditions produced in closed containers with the use of saturated salt solutions (Taylor et al., 1992). We prepared a number of saturated salt solutions and desiccants to achieve a range of humidity levels from c. 0% to 89% in equilibrated snap beans (Phaseolus vulgaris) and broccoli (Brassica oleracea italica group) seeds. Three regions or zones of water binding are observed: Zone I, < 20% RH; Zone II, 25–65% RH; and Zone III, > 70% RH (Fig. 1.2).
The curves are similar for both kinds of seeds; however, the equilibrium moisture content at a given relative humidity is always greater for snap beans compared to broccoli. Differences between these two species are attributed to seed composition, in particular lipid content, as lipids have little affinity for water. Thus, seeds with high lipid content have lower equilibrium moisture content at a given relative humidity than seeds with low lipid content. In our isotherm, the lipid content for the snap bean and broccoli seeds was 1% and 31%, respectively (Table 1.1).
Another method used to quantify the water status of seeds, which is not affected by seed composition, is to measure the water activity (aw). Water activity is defined as the ratio of the vapor pressure of water in a seed to the vapor pressure of pure water at the same temperature (Bourne, 1991). Water activity is measured by determining the equilibrium relative humidity of the seed’s headspace in a closed container and is expressed as a decimal. For example, a seed (any seed) equilibrated to 50% relative humidity has a water activity of 0.50. Measuring water activity of pelleted seeds (described in the “Seed enhancements” section) was the only nondestructive method to accurately measure the water status of coated seeds (Taylor et al., 1997).
Storage: Moisture Content and Temperature
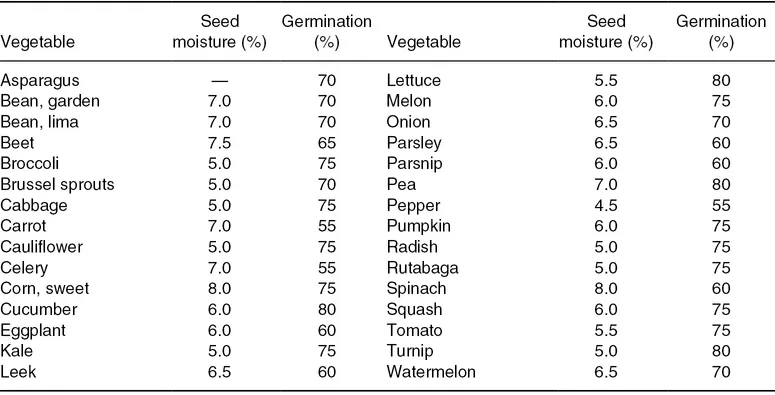
Two major environmental factors that influence seed storage are seed moisture content and temperature. Moisture content, as previously shown, is determined by the storage relative humidity and by seed characteristics, largely lipid content. The concentration of water in the seed tissue directly affects the rate of aging at a given temperature. The moisture isotherm reveals three zones or types (see Fig. 1.2) in which the water binding to seed tissues differ. Type I water is bound tightly and water interacts very strongly with charged groups of proteins (Vertucci, 1993). Type II water is less tightly bound and condenses over the hydrophilic sites of macromolecules (Leopold and Vertucci, 1989). Type III water is bound with negligible energy and forms bridges over hydrophobic moieties (Vertucci, 1993). The status of water in the seed tissue governs the kind of reactions (enzymatic or non-enzymatic) that occurs in storage (Vertucci, 1993). In practice, vegetable seeds are stored in Zone II (Fig. 1.2), and the recommended seed moisture content for vegetable crop seeds stored in hermetically sealed containers is listed in Table 1.2.
Table 1.2. Recommended seed moisture content for vegetable crop seeds in hermetically sealed containers. Minimum percentage germination standards for seeds in interstate commerce in the United States. Data from Federal Seed Act (USDA, 2017).
Temperature has a direct influence on longevity, and the rate of deterioration increases as temperature increases at a given relative humidity. Seeds are stored over a wide range of temperatures depending on the particular needs and available conditions. Seeds for most applications are stored above 0°C; however, for long-term preservation, seeds are stored below 0°C. Seeds with water contents in Types I or II are not injured since the water is non-freezable in the seed tissue. Long-term germplasm preservation has taken advantage of this ability to withstand freezing injury as seeds are stored above liquid nitrogen in the vapor phase at −150 to −180°C (Roos, 1989).
The need to keep seeds cool and dry for long-term preservation has been known for centuries. In the 1960s, “Harrington’s Rules of Thumb” were developed as guidelines for storage (cited by Justice and Bass, 1978). The first two rules relate the influence of moisture content and temperature independently on longevity. The life of the seed is halved by a 1% increase in seed moisture, and this rule applies when seed moisture content ranges from 5% to 14%. The life of the seed is halved by a 5°C increase in storage temperature, and this applies to storage between 0 and 50°C. The most widely quoted rule combines both temperature and relative humidity to storage: “The sum of the temperature in °F and the percentage of relative humidity should not exceed 100.”
Since ...