
eBook - ePub
Fast Facts: Myelodysplastic Syndromes
Determining Risk, Tailoring Therapy, Supporting Patients
- 76 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Fast Facts: Myelodysplastic Syndromes
Determining Risk, Tailoring Therapy, Supporting Patients
About this book
Myelodysplastic syndromes (MDS) are a group of clonal myeloid blood disorders that affect older people. As the global population ages, the incidence of these disorders is increasing. As such, healthcare professionals need to be aware of MDS as a possible diagnosis and know how to identify those patients whose condition is most likely to transform to acute myeloid leukemia (AML). Fast Facts: Myelodysplastic Syndromes explains how these disorders are classified, how they present and how prognosis and the risk of transformation to AML are assessed. It also outlines the most appropriate management options for patients of different ages, together with an overview of the latest research under way to further understand MDS and how to treat it. This highly readable resource is the ideal primer on MDS for all members of the multidisciplinary team involved in the care of patients with MDS, including hematologists, hematology trainees, clinical nurse specialists and primary care providers.
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
Medicine3Diagnosis and classification
There is no specific diagnostic test for MDS; they must be distinguished from the diagnosis of dysplasia associated with marrow failure syndromes and other clonal disorders, as well as non-clonal non-malignant causes, such as infections, nutritional deficiencies and intoxications (Figure 3.1). Diagnosis requires excellent morphological evaluation, as well as cytogenetic and molecular data, in the context of peripheral blood cytopenias. Clinical correlation is also important to rule out familial syndromes and non-neoplastic causes of dysplasia.
MDS should be suspected in a patient with otherwise unexplained cytopenia. Although the diagnosis may be suggested by the blood counts and peripheral blood smear, ultimately, analysis of bone marrow samples is required to make a firm diagnosis. This allows examination of the morphological, histological and genetic changes that are required to reach and subclassify a diagnosis of MDS. Because there is no single test for MDS and some aspects of diagnosis, such as the interpretation of dysplastic changes, can be subjective, it is often a challenging condition to confidently diagnose.

Figure 3.1 Clonal and non-clonal causes of dysplasia and diseases that overlap with MDS morphology or clinical presentation. Figure not drawn to scale. AA, aplastic anemia; CCUS, clonal cytopenia of undetermined significance; ICUS, idiopathic cytopenia of undetermined significance; IDUS, idiopathic dysplasia of undetermined significance; LGL, large granular lymphocyte leukemia; MPN, myeloproliferative neoplasm; PRCA, pure red cell aplasia.
Differential diagnoses
There is a long list of differential diagnoses for cytopenia and many non-malignant causes of dysplastic features that mimic those of MDS (Table 3.1). Initial review should focus on excluding non-clonal causes before focusing on a possible MDS diagnosis. The emergence of a greater array of genetic tests has aided the diagnosis of MDS, but equally has created a more complex diagnostic landscape.
Once a diagnosis of MDS is reached, the next stage is to subclassify and then risk stratify the disorder. This will inform the patient’s prognosis and help to determine the most appropriate management strategy.
TABLE 3.1
Differential diagnosis of suspected MDS
• Chronic liver disease
• Drug-induced cytopenia
• Alcohol excess
• Cytotoxic medications
• Vitamin B12/folate deficiency
• Autoimmune disease
• Anemia in chronic disease
• Parasitic manifestations
• HIV infections
• Other stem-cell disorders
Full blood count and peripheral blood smear
Full blood count. The presence of a cytopenia is a prerequisite for the diagnosis of MDS. According to the WHO classification of MDS revised in 2016, cytopenia is defined as hemoglobin less than 10 g/dL (100 g/L), platelets less than 100 × 109/L and absolute neutrophil count less than 1.8 × 109/L.1
A peripheral blood smear should be created once a cytopenia has been identified so that morphological assessment can be undertaken. Peripheral blood morphology can be normal in MDS, but there are often changes that hint at the diagnosis (Table 3.2 and Figure 3.2). The first review of a film, and therefore suspicion of MDS, will generally be made in the laboratory and, as such, the presence of experienced and competent laboratory staff is essential.
Common features include macrocytosis in mature erythrocytes or hypogranularity and/or abnormal nuclear appearances in mature neutrophils (see Figure 3.2). Changes in platelets can be more subtle but may also be present.
TABLE 3.2
Blood film features suggestive of/associated with dysplasia (but not pathognomonic for MDS)
Erythrocytes | Granulocytes | Platelets |
Anisocytosis | Abnormal segmentation | Giant platelets |
Poikilocytosis | Pseudo-Pelger neutrophils | Platelet anisocytosis |
Polychromasia | Hypersegmentation | Megakaryocytic fragments |
Stippling/granules | Hypogranular and agranular forms | Rarely micromegakaryocytes |
Abnormal hemoglobinization | ||
Nucleated RBCs |
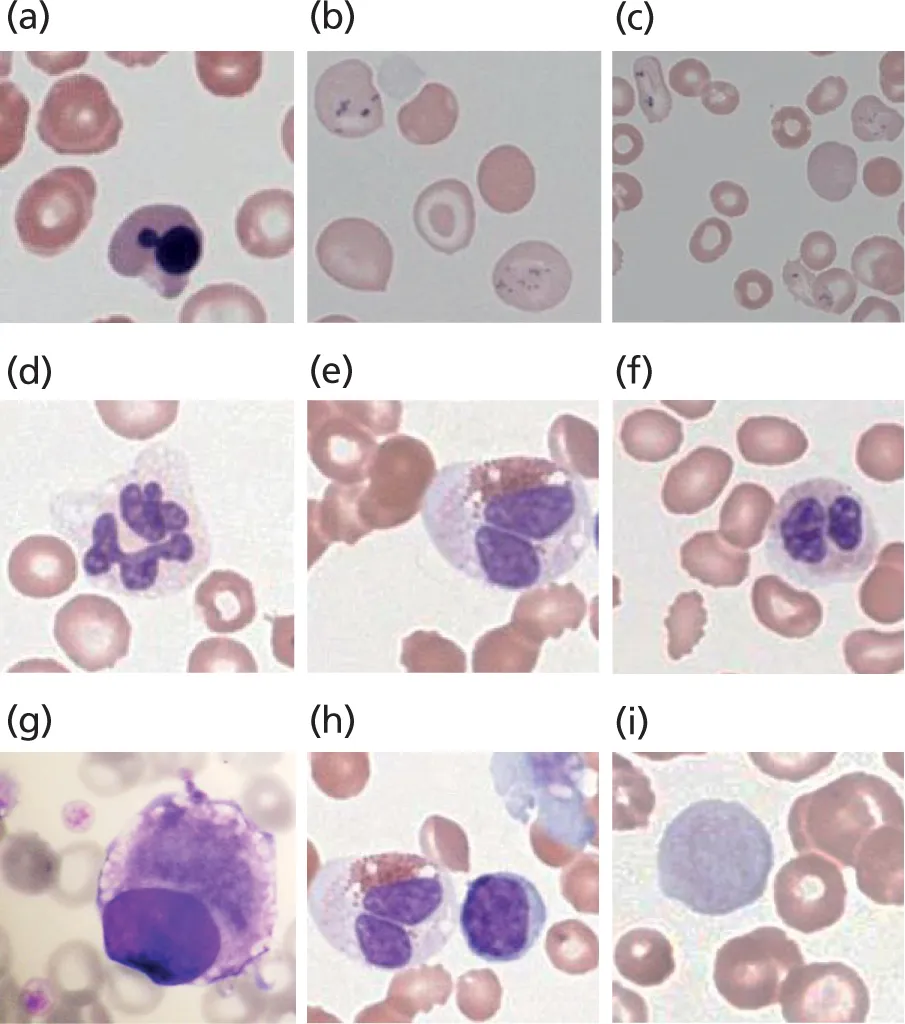
Figure 3.2 Examples of dysplastic features in peripheral blood in MDS. Erythrocytes (MDS-RS-SLD): (a) nuclear blebbing in erythroblast; (b) basophilic stippling and siderotic granules; and (c) abnormal hemoglobinization of RBCs with basophilic stippling and granules. Granulocytes (MDS-RS-MLD): (d)–(f) abnormal segmentation of neutrophils. Platelets: (g) micromegakaryocyte and large platelets; (h) megakaryocytic fragment and abnormal granulation of eosinophil; and (i) giant platelet. All images 600× magnification. Images: NH Lawrence©. MLD, multilineage dysplasia; RS, ring sideroblast; SLD, single-lineage dysplasia.
Bone marrow biopsy
Unless clinically inappropriate, all patients in whom MDS is suspected should undergo bone marrow biopsy. This procedure will allow the acquisition of samples for morphological and histological assessment, immunophenotyping and genetic analysis.
Bone marrow morphology. A marrow aspirate is a liquid sample obtained from the bone marrow cavity. An aspirate smear is made, which will demonstrate the hematopoietic elements within the marrow and must be carefully examined by an experienced hematopathologist. Quantitative and qualitative comments should be made for each of the erythroid, myeloid and megakaryocytic lineages, specifically commenting on whether maturation is normal or abnormal, with a description of any abnormal features that are present. The presence of dysplastic features in 10% or more of any of the hematopoietic lineages is considered significant and compatible with a diagnosis of MDS (Figure 3.3 and Table 3.3). Given that a precise percentage of an abnormal cell type is required for MDS diagnosis, a 500-cell differential should be performed if t...
Table of contents
- Cover
- Title Page
- Copyright
- Contents
- List of abbreviations
- Introduction
- Epidemiology and etiology
- Pathogenesis and presentation
- Diagnosis and classification
- Management
- Supporting patients with MDS
- Useful resources
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Fast Facts: Myelodysplastic Syndromes by Peter Dyer,Martha L. Arellano,P. Dyer,M.L. Arellano in PDF and/or ePUB format, as well as other popular books in Medicine & Family Medicine & General Practice. We have over 1.5 million books available in our catalogue for you to explore.