Acupuncture in Manual Therapy is a comprehensive overview of manual therapy interventions combined with acupuncture management of musculoskeletal disorders covering all major joints of the body. The book is intended to offer physiotherapists and students of acupuncture a clinical reasoning model, whilst supporting suggested interventions with the relevant evidence base in order to alleviate pain, restore function and facilitate rehabilitation within musculoskeletal management. It places an emphasis on the neurophysiological processes of pain management stressing the importance of using the correct intervention for the relevant presenting pain mechanism. Each chapter discusses suggested manual intervention, acupuncture evidence and physiological processes underpinning this combined approach to musculoskeletal pain and dysfunction including meridian acupuncture, auricular acupuncture, electroacupuncture, tens and trigger point acupuncture as a means of addressing the varying pain patterns presented within musculoskeletal rehabilitation.This book provides knowledge from distinctive and prominent contributors from the world of manual therapy and acupuncture whilst this expertise is further supported by the provision of student case studies to offer support and enhance learning.- Twelve chapters addressing each major joint of the musculoskeletal system- Over 100 images- A clinical reasoning model for manual therapy and acupuncture intervention- Relevant case studies that support the student undertaking acupuncture training

- 248 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Acupuncture in Manual Therapy
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
MedicineSubtopic
Acupuncture1 Clinical reasoning in Western acupuncture
Background 1
Theoretical knowledge underpinning the model 1
Acupuncture mechanisms 2
Nociception 2
Supraspinal effects 2
Neurohormonal responses 2
Clinical reasoning model: the layering method 3
Local effects 3
Segmental effects 3
Supraspinal effects 5
Conclusion 6
References 17
Background
Using acupuncture to treat musculoskeletal disorders should follow a clinical reasoning process (CRP), the thinking behind practice, as identified by physiotherapists for manual therapy interventions (Jones & Rivett 2004), the norm being to identify predominant tissue and pain mechanisms presented by the patient as a means of identifying effective intervention. The layering method is a clinical reasoning model (CRM) developed specifically for clinicians to treat musculoskeletal conditions with acupuncture, using a mechanism-based approach (Bradnam 2007). It aims progressively to target different physiological processes within the central nervous system (CNS) in order to provide the best effect for each individual. The layering method is a Western approach to acupuncture, but does allow a clinician to integrate traditional Chinese acupuncture (TCA) point selection into clinical reasoning.
An orthodox physiotherapy assessment and diagnosis is made with identification of likely contributors to the patients’ disability in terms of:
• Associated anatomical structures;
• Tissue sources;
• Tissue healing; and
• Pain mechanisms. (Jones & Rivett 2004)
An acupuncture treatment plan will be formulated to target structures identified as sources of the physical impairment. Applying acupuncture mechanisms in this manner will also allow progression of treatment if the initial approach does not achieve the desired effect; if pain mechanisms change, or if the condition resolves or becomes chronic.
Theoretical knowledge underpinning the model
The following knowledge must underpin the model:
• An understanding of how acupuncture affects the CNS;
• The clinical presentation of pain mechanisms; and
• The tissue healing process and time frames for these processes to be achieved.
The practical implementation of the model relies on:
• A knowledge of acupuncture points;
• A good knowledge of anatomy;
• A knowledge of segmental and peripheral nerve innervation of muscles and skin; and
• A full understanding of the neuroanatomy of the autonomic nervous system (ANS).
Acupuncture mechanisms
Nociception
Three categories of acupuncture mechanisms have been described; peripheral, spinal, and supraspinal (Lundeberg 1998). Firstly, on needling, nociceptive afferents are stimulated and release vasodilatory neuropeptides into the muscle and skin they innervate, forming the basis of the local or peripheral effects of acupuncture (Sato et al 2000). This phenomenon, an axon reflex, releases neuropeptides into human skin such as calcitonin gene-related peptide (CGRP) and substance P (Weidner et al 2000). Sensory neuropeptides modulate immune responses and hence will assist in tissue healing (Brain 1997). Secondly, acupuncture will act within the spinal cord, known as spinal effects or segmental effects. To initiate spinal effects, the sensory stimulus must be applied to tissues that share an innervation with the appropriate spinal cord level (Fig. 1.1). Dorsal horn neurons activated by painful inputs may be inhibited by acupuncture via a gate control mechanism, producing a spinally mediated analgesic response. Neurons of the ANS efferent fibres can be influenced and both sympathetic and parasympathetic activity may be affected, depending on the position of the needles.
• High-intensity (HI) needling may immediately increase sympathetic outflow to tissues supplied by the segment, which is then followed by a decrease in outflow.
• Low-intensity (LI) or non-painful input could reduce sympathetic outflow in the segment (Sato et al 1997).
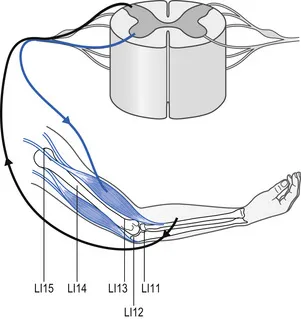
Figure 1.1 Dermatome and myotome innervation from C5 nerve root.
Lastly, acupuncture may influence alpha-motoneurons housed in the ventral horn of the spinal cord to alter reflex activity in muscles supplied by the segment (Fig. 1.1). At present the effect on motoneurons is still unclear: an immediate change in excitability has not been demonstrated in contrast to clinical observations (Chan et al 2004).
Supraspinal effects
Acupuncture can influence neuronal structures within the brain (Stener-Victorin et al 2002) and these are known as supraspinal effects. Analgesic pathways such as diffuse noxious inhibitory controls (DNIC) and beta-endorphin mediated descending pain inhibitory pathways from the hypothalamus will be activated with appropriate needling (Stener-Victorin et al 2002). Autonomic outflow is also under central control via the medullary vasomotor centre and can be influenced by the acupuncture stimulus.
Neurohormonal responses
Responses affecting the immune, endocrine, and reproductive systems of the body can be affected by acupuncture (Carlsson 2002, Stener-Victorin et al 2002; White 1999). Recent advances in brain imaging technologies such as functional magnetic imaging (fMRI) and positron emission tomography (PET) have allowed investigations of the brain and have elucidated the effect of acupuncture on the CNS. Several analgesic points in the extremities will stimulate blood flow to cortical and subcortical brain regions (Lundeberg 1998). Activation is relatively non-specific and closely related to areas activated by painful stimuli, through what is known as the pain matrix (Lewith et al 2005). Studies show an increase in blood flow in the hypothalamus (Table 1.1) and a decrease in the limbic system (Table 1.2), a brain region where affective and emotional responses to pain are integrated with sensory experience. However, most of the brain regions activated by acupuncture are closely related to those areas mediating placebo analgesia and expectation (Lewith et al 2005), and it is unclear how much of the change is due to the acupuncture stimulus and how much is due to non-specific effects. Recently studies using transcranial magnetic stimulation (TMS) have shown that acupuncture modulates motor cortical excitability and that the effect (excitation or inhibition) is specific to the investigated muscle and the site of needle placement (Lo et al 2005; Maioli et al 2006). Maioli et al (2006) showed that changes lasted for fifteen minutes following the removal of the needle stimulus, suggesting longer term plastic changes in motor cortical excitability.
Table 1.1 Suggested points to stimulate blood flow to hypothalamus
| Meridian | Points |
|---|---|
| Large intestine | LI4 |
| Lung | LU5 |
| Gall bladder | GB34, GB40 |
| Spleen | SP6 |
| Stomach | ST36 |
| Liver | LIV3 |
Biella et al (2001); Fang Kong et al (2004); Hsieh et al (2001); Hui et al (2000); Wu et al (1999, 2002); Yan et al 2005; Zhang et al (2003)
Table 1.2 Suggested points for deactivation of limbic system
| Meridian | Points |
|---|---|
| Large intestine | LI4 |
| Gall bladder | GB34 |
| Spleen | SP6 |
| Stomach | ST36 |
| Liver | LIV3 |
Hsieh et al (2001); Hui et al (2000, 2005); Kong et al (2002); Wu et al (1999,2002); Zhang et al 2003
Clinical reasoning model: the layering method
Clinical reasoning within acupuncture intervention requires that the clinician ask a series of questions as to what is required from the needle. The question provides a problem-solving pathway as to effects on pain and tis...
Table of contents
- Cover
- Title Page
- Front-matter
- Copyright
- Contributors
- Dedication
- Acknowledgements
- Preface
- Table of Contents
- Chapter 1: Clinical reasoning in Western acupuncture
- Chapter 2: The temporomandibular joint
- Chapter 3: Cervical spine
- Chapter 4: The shoulder
- Chapter 5: The elbow
- Chapter 6: The thoracic spine
- Chapter 7: The lumbar spine
- Chapter 8: The sacroiliac joint and pelvis
- Chapter 9: The hip
- Chapter 10: Anterior knee pain
- Chapter 11: Foot and ankle
- Chapter 12: Transcutaneous electrical nerve stimulators for pain management
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Acupuncture in Manual Therapy by Jennie Longbottom in PDF and/or ePUB format, as well as other popular books in Medicine & Acupuncture. We have over 1.5 million books available in our catalogue for you to explore.