
eBook - ePub
Perspiration Research
- 124 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Perspiration Research
About this book
Research into perspiration has developed dramatically during the last 15 years, continually improving our understanding of the pathogenesis of sweating disorders. It has become clear that, in addition to its temperature-regulating function, perspiration offers bactericidal protection as well. In this book, select authors further broaden our perspective on perspiration. Contributions cover a variety of new aspects, offering insight into the sweat glands' major role during the onset of disorders such as parapsoriasis, lichen planus, and lichen amyloidosis. They also highlight the importance of Malassezia - an allergen in sweat that exacerbates atopic dermatitis and cholinergic urticarial. Further roles of the sweat glands are discussed, including as storage of stem cells for replenishing epidermal cells in the case of thermal burns or as water retention sites for replenishing moisture in the stratum corneum. In addition, a novel analysis of the sweat glands' three-dimensional structures, using high-speed en-face optical coherence tomography (OCT), is introduced. Offering an in-depth overview of the latest knowledge in perspiration research, this book serves as an essential reference for all medical staff and researchers in the field.
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
MedicineSubtopic
CardiologyYokozeki H, Murota H, Katayama I (eds): Perspiration Research.
Curr Probl Dermatol. Basel, Karger, 2016, vol 51, pp 109-119 (DOI: 10.1159/000447370)
Curr Probl Dermatol. Basel, Karger, 2016, vol 51, pp 109-119 (DOI: 10.1159/000447370)
______________________
Perspiration Functions in Different Ethnic, Age, and Sex Populations: Modification of Sudomotor Function
Jeong-Beom Leea · Jeong-Ho Kima · Hiroyuki Murotab
aDepartment of Physiology, College of Medicine, Soonchunhyang University, Cheonan, Republic of Korea; bDepartment of Dermatology, Course of Integrated Medicine, Graduate School of Medicine, Osaka University, Osaka, Japan
______________________
Abstract
The sudomotor mechanism, wich contributes to tolerating thermal environments, is affected by not only the body temperature, but also sex, ethnicity, exercise training, region, season, and heat adaptation. Aging attenuates the sudomotor function by the decreased peripheral sensitivity to acetylcholine and demyelination of innervating nerves. Women show less sudomotor activity than men. Heat adaptation with sudomotor modification is induced by repetitive physical and/or thermal training. Short-term heat acclimation increases sweat gland activity. Long-term heat acclimation results in a reduction in the sweating response to stimuli. Residents of tropical areas sweat less and more slowly than residents of temperate areas. Short-term heat acclimation enhances the sweating response. Long-term heat acclimation, from seasonal change or migration, diminishes the sweating response. Also, deacclimation can be induced by migration from a tropical area to a temperate area. Body composition, especially brown adipose tissue, and weight affect thermal responses. Further studies should investigate BAT and endocrinal pyrogens as additional factors.
© 2016 S. Karger AG, Basel
Humans can tolerate a wide range of thermal environments, but physiological function is dependent upon the maintenance of thermal homeostasis. When exposed to hyperthermic conditions, via environmental factors and/or increased metabolism, heat dissipation becomes vital for survival. In humans, the primary mechanism of heat dissipation is evaporative heat loss, secondary to sweat secretion from the eccrine glands. With a large surface area of hairless skin, sweating is an efficient evaporative cooling mechanism in humans. Evaporation of sweat is an important heat-loss process in the control of internal body temperature in a hot environment, in which the ambient temperature is higher than the skin temperature.
Central sudomotor mechanisms integrate inputs from the core and skin thermoreceptors, and produce signals to control the sweat glands [1-3]. Efferent sweat fibers originate in the preoptic area and anterior hypothalamus, and descend to the postganglionic sympathetic plexus (sudomotor neurons) through the ipsilateral brain stem and medulla [4]. The sweat glands generate a sweat response according to the central sudomotor nerve activity [1, 5-9]. In addition, peripheral conditions (local skin temperature and blood flow) can modulate the sweating response [10].
The autonomic nervous system regulates sudomotor function. Sudomotor response can be tested using several tools, such as thermoregulatory sweat testing, the Quantitative Sudomotor Axon Reflex Test (QSART), silicone impressions, the sympathetic skin response, the acetylcholine (ACh) sweat-spot test, and quantitative direct and indirect axon reflex testing [11]. QSART evaluates the postganglionic sympathetic sudomotor axons by measuring the axon reflex-mediated (AXR) and directly activated (DIR) sweat response over time, following stimulation of sweat glands with 10% ACh [4, 12, 13]. QSART has been a routine postganglionic sudomotor function (HCFA 95923) clinical laboratory test of the Mayo Clinic since 1983. DIR is based upon the response of muscarinic receptors, whereas AXR represents the nicotinic receptor response [12, 13]. Forearm sweat glands have been widely used to approximate sweating activity of the whole body [14, 15]. According to Low et al. [16], QSART provides close to maximal sweat gland stimulation. A large number of previous reports using QSART on the forearm have potentially demonstrated overall (whole body) sudomotor sensitivity.
Neural control of sweating, primarily regulated by the integration of internal and skin temperatures, is paramount for temperature regulation; however, a variety of other thermal and nonthermal factors also modify the sweating response. In this study we mainly focus on age, sex, ethnicity, exercise training, region, season, and heat adaptation.
Age and Sex
Numerous studies have used specific techniques to show that age and/or sex affect sudomotor function [1, 17-29]. Researchers have used ACh and/or methylcholine for sweating, and recruited specifically women or men, or subjects of both sexes, across various age groups. The results from such previous studies show that the attenuation of sudomotor function occurs with increased aging in both men and women. There is a progressive increase in onset time and a decrease in sweat rates, density of activated sweat glands (ASGs), and sweat output per gland (SGO), with increasing age in both sexes (fig. 1). A variation in sweat function between the sexes has been reported, but is not consistent in all age groups. Larose et al. [30] recently demonstrated, in a large sample of men between the ages of 20 and 70 years, that the capacity to dissipate heat was compromised as early as the age of 40 years, and declined progressively thereafter. Meanwhile, a slightly different approach is needed in the case of children. Araki et al. [31] and Shibasaki et al. [32] suggested that maturation is an important factor, as postadolescent subjects had a superior ability to activate sweat function compared with preadolescents during muscular exercise. Rees and Shuster [33] did not reveal a significant difference between sweat rates in prepubertal males and females.
There are several possible explanations for the apparent attenuation of sweating with ageing. Some researchers have suggested that the thermoregulation threshold (i.e. sweating threshold) is different between younger and older subjects, such that the threshold for the onset of sweating increases with ageing [22-24, 28]. The biological function of sweat glands stimulated by ACh becomes lower with aging [28]. Inoue et al. [34] suggested that, as no significant change in the central sudomotor drive has been noted, the sluggish sweating response appears to be associated with age-related modifications in the periphery, involving reduced sweat gland sensitivity to cholinergic stimulation, or to sluggish vasodilatation. Low et al. [18] suggested that this change is caused by demyelination of the nerve fibers. Inoue et al. [35] demonstrated that SGO decreases gradually with age, due to progressive sweat gland atrophy. It has been postulated that the age-related impairments in sweating are due to differences in end-organ function, such as cholinergic sensitivity of the muscarinic receptors on the sweat glands [25, 26].
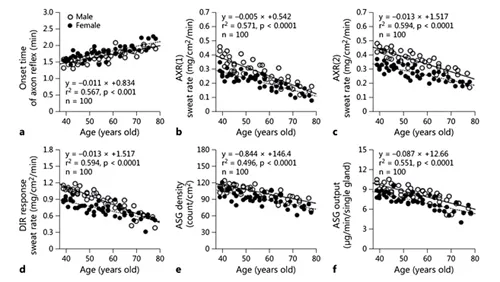
Fig. 1. A study [29] of the sudomotor function related to age and sex in healthy men (n = 52; ○) and healthy women (n = 48; ●) by QSART. a Sweating onset time of the AXR. b AXR(1) sweat rate, AXR(2) sweat rate. c DIR sweat rate. d ASG density. e Activated single SGO. AXR(1) = Sweating by axonal reflex, activating nicotinic receptor during iontophoresis; AXR(2) = local skin area where the sweating activity is induced by the nicotinic receptor: axonal reflex sweating after iontophoresis, for 5 min; DIR: direct responsive sweating induced by the muscarinic receptor after iontophoresis for 5 min. Sweating activities were compared after ACh iontophoresis. Correlation between onset time of the AXR sweating (a), AXR(1) sweating rate (b), AXR(2) sweating rate (c), DIR sweating rate (d), ASG density (e), ASG output (f), and age in male and female subjects.
The nervous system, particularly postganglionic sympathetic nerve fibers that innervate sweat glands, degenerates with age. Namer et al. [36] showed that the physiological function of C- fibers declines with age and that mechanoresponsive C-fiber (classified afferent C-fiber) composition decreases to 58.5% in young subjects, compared with 39.6% in aged subjects, although the proportion of afferent and efferent fibers in young subjects is equal to those found in elderly subjects. Based on the data provided by Namer et al. [36], conduction velocity in aged subject...
Table of contents
- Cover Page
- Front Matter
- New Pathologies of Skin Disorders Identified from the History of Perspiration Research
- Classification of Systemic and Localized Sweating Disorders
- New Findings on the Mechanism of Perspiration Including Aquaporin-5 Water Channel
- Old and New Approaches for Assessing Sweating
- Sweat as an Efficient Natural Moisturizer
- Genetic Disorders with Dyshidrosis: Ectodermal Dysplasia, Incontinentia Pigmenti, Fabry Disease, and Congenital Insensitivity to Pain with Anhidrosis
- Histamine Modulates Sweating and Affects Clinical Manifestations of Atopic Dermatitis
- Sweating in Systemic Abnormalities: Uremia and Diabetes Mellitus
- Abberant Sudomotor Functions in Sjögren’s Syndrome: Comparable Study with Atopic Dermatitis on Dry Skin Manifestation
- Clinical Analysis and Management of Acquired Idiopathic Generalized Anhidrosis
- Dyshidrotic Eczema and Its Relationship to Metal Allergy
- Pathophysiology and Treatment of Hyperhidrosis
- New Etiology of Cholinergic Urticaria
- Sweat Allergy
- Perspiration Functions in Different Ethnic, Age, and Sex Populations: Modification of Sudomotor Function
- Author Index
- Subject Index
- Back Cover Page
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Perspiration Research by H. Yokozeki,H. Murota,I. Katayama,H., Yokozeki,H., Murota,I., Katayama, P. Itin,G. B. E. Jemec,P., Itin,G.B.E., Jemec in PDF and/or ePUB format, as well as other popular books in Medicine & Cardiology. We have over 1.5 million books available in our catalogue for you to explore.