![]()
PART I: MEMBRANE MATERIAL AND MEMBRANE DEVELOPMENT
1
SPINNING EFFECT OF POLYETHERSULFONE HOLLOW FIBER MEMBRANE PREPARED BY WATER OR POLYVINYLPYRROLIDONE IN TERNARY FORMULATION
Nurmin Bolong, Ahmad Fauzi Ismail, and Mohd Razman Salim
1.1 INTRODUCTION
The art in membrane making has been found to be influenced by many factors starting from the dope contents and formulation up to the fabrication process, including posttreatment and module preparation of the membranes. Since Leob and Sourirajan invented the first flat anisotropic cellulose acetate membranes by the phase inversion process in the late 1950s [1], the synthetic polymeric membrane technology has obtained much attention from both academia and industry. Compared to the flat sheet, hollow fiber membranes possess the following advantages: (1) much larger membrane area per unit volume of membrane module which results in higher productivity, (2) large packing density that offers the lowest cost per unit membrane area (3), self-mechanical support which enables back-flushing to recover the membrane flux, (4) good flexibility and easy handling during module fabrication and in the operation [2–4].
The ability of producing self-made membranes allows a greater chance to produce and achieve a successful treatment system since during the production process of membrane it can be tailored to suit the requirements. However, many important factors need to be taken into account and properly controlled in order to allow production of membranes that meet the desired characteristics. In general, membrane fabrication involves trial-and-error method which covers optimal material formulation, selection of critical operating variables, and proper conditions during fiber spinning. Although a great deal of works have been carried out in the past, fabrication of membrane for specified application is still extremely difficult and becomes an interesting subject of study.
One of the common polymer materials used for membrane formation is polyethersulfone (PES). PES is principally favorable due to the characteristics of wide temperature limits, wide pH tolerances, fairly good chlorine resistance, easiness to fabricate membranes in a wide variety of configurations and modules, wide range of pore sizes ranging from 10 Å to 0.2 µm, and good chemical resistance to aliphatic hydrocarbons, alcohols, and acids [5, 6].
In the immersion process, choice of the additive or the nonsolvent system has strong influence in membrane fabrication. In particular, to increase hydrophilicity of the resultant membrane, a hydrophilic polymer, polyvinylpyrrolidone (PVP), is often blended into relatively hydrophobic PES [7, 8]. The addition of a second polymer such as PVP in the casting solution may produce highly porous membranes with well-connected pores and surface properties [6]. Water is also frequently used as a nonsolvent [9], and can form a complex multiple hydrogen bonding with solvents such as N,N-disubstituted amides (e.g., N-methyl pyrrolidone, NMP) [10]. Furthermore, water can be used to control the porosity of the membranes by inducing macrovoids [11].
In this work, an attempt is made to study and compare the effect of water and PVP as additives in the polymer solution toward PES as a base polymer in fabricating hollow fiber membranes by the phase inversion technique. The hollow fiber membranes are characterized by pure water permeation (PWP) and sodium chloride rejection. The effect of spinning conditions, particularly that of the dope extrusion rate (DER), is investigated. Furthermore, dry-wet or wet-wet spinning influences are also compared.
1.2 EXPERIMENTAL
Hollow fiber membranes were fabricated in the laboratory using a simple phase inversion technique [12]. The homemade hollow fiber membranes were spun at spinning parameters given in Table 1.1, using water as coagulant both in the coagulation tank and as the bore fluid. Prior to spinning, a spinning dope of an exclusive formulation consisting of PES (Radel A-300 with a molecular weight of about 15,000 Dalton (Da), purchased from Amoco Performance Products Inc., Atlanta, GA), water (deionized) or PVP (Kollidon® 15, or PVP K-15) with a weight average molecular weight (Mw) of 10,000 Da (E. Merck KGaA, Darmstadt, Germany), and N-methyl-2-pyrrolidone (NMP, >99%, Aldrich Chemical Comp. Inc., Milwaukee, WI) was prepared to produce an asymmetric hollow fiber membrane.
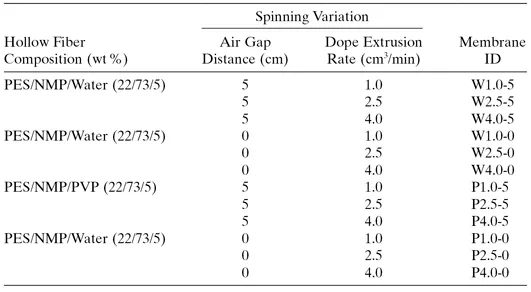
TABLE 1.1. Hollow Fiber Spinning Parameters
The fabricated hollow fiber membranes were characterized by filtration tests using a cross-flow filtration setup as detailed elsewhere [13]. Prior to the filtration test, 30 hollow fibers (approximately of 10 × 10−3 m3 surface area) were potted into a bundle, which was slowly inserted into a stainless steel 30 mm housing and assembled with the permeation test system. After 0.5 hour of pressurization at 90 psig, the PWP flux was first measured at an operating pressure of 80 psig and at ambient temperature. Then the separation experiments were performed using 0.01 M sodium chloride. The sodium chloride solute concentrations in the feed and the permeate were determined by a conductivity meter (WTW handheld meter model LF330).
1.3 RESULTS AND DISCUSSION
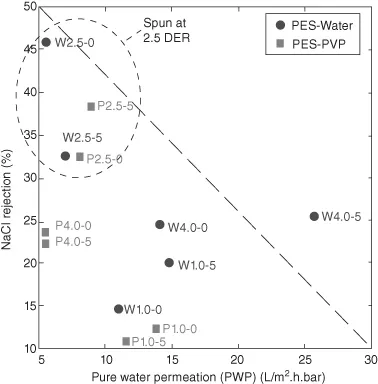
The results of filtration experiments are illustrated in Figure 1.1. The pressure normalized pure water flux (also termed as PWP) was calculated for all the membranes and divided into the two main groups (i.e., PES-water and PES-PVP), which then were plotted against the sodium chloride rejection. Regardless of spinning conditions, the results typically reveal that PVP addition could reach an optimum NaCl rejection of ∼40%. The highest PWP was ∼15 L/m2.h.bar. Whereas, use of water as nonsolvent has shown a larger range of separation performance with up to ∼45% NaCl rejection and the highest PWP of ∼28 L/m2.h.bar.
Water or PVP certainly had an effect on the thermodynamic interaction properties of the casting dopes and the process of demixing and thus on membrane structures and performances. The existence of ternary component in the dope formulation has caused a shift in the composition path toward binodal or liquid–liquid demixing. This thermodynamically indicates that the addition of water or a second polymer such as PVP to the casting solution enhances coagulation [6] and induces occurrence of instantaneous demixing [9]. Addition of both water and PVP generally leads to an increment of PWP compared to pure PES. For example, PES membrane fabricated under the condition 2.5 DER at 5 cm air gap without additive showed PWP = 2.18 L/m2.h.bar and ∼47% NaCl separation. PVP is known to hydrophilize the membrane [13] and influence membrane surface porosity [6]. On the other hand, water is a strong nonsolvent for PES, and primarily affects the phase inversion rate when diffused into the polymer solution.
From the data shown in Figure 1.1, the addition of PVP could enhance sodium chloride rejection but not necessarily contribute to the enhancement of PWP, whereas the addition of water affected PWP and NaCl rejection in a much wider range. This might be related to the overall morphological properties of the membranes and due to the stronger effect of water on demixing rate than PVP.
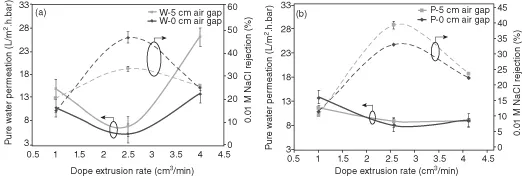
Figure 1.1 further indicates that all the high NaCl rejection data are found in a region surrounded by the broken line where hollow fibers were spun at DER of 2.5 cm3/min. This trend can also be found in Figure 1.2 where PWP and NaCl rejection are plotted versus DER for the addition of (1) water and (2) PVP, respectively. Figure 1.2 shows the effect of DER and air gap on PWP and NaCl rejection. The PWP of both PES/water and PES/PVP membranes were found to have a minimum at 2.5 DER. Conversely, NaCl rejection had a maximum at this DER. This is due to the effect of the shear rate on the macromolecular packing and alignment in the hollow fiber.
Figure 1.3 explains schematically the effect of DER on the macromolecular arrangement. At the optimum DER, the shear force working on the outer surface of the nascent hollow fiber causes polymer molecules to be packed closer, leading to a tighter skin structure with enhanced molecular orientation. As a result, hollow fiber membranes of denser outer skin layers with small pore sizes are formed [14, 15]. Hence, PWP decreases and the solute separation increases. However, further increase in DER beyond a critical point disrupts the molecular orientation and loosens the outer skin structure. This in turn enlarges the pore sizes and thus reduces solute separation and increases water flux. The DER effect on PWP was found more significant for PES-water than PES-PVP formulation, indicating that PES-PVP has stronger molecular chain interaction.