![]()
1
BRAIN CIRCUITRY IN AGEING AND NEURODEGENERATIVE DISEASE
Sharna Jamadar
Introduction
As we age, our bodies undergo widespread and significant changes. Our hair greys and becomes thinner, our skin wrinkles, our eyesight and hearing become worse. Many people experience changes in their thinking as they get older, finding it more difficult to remember events and solve complex problems. It is not all doom and gloom for older people, however: older people have accumulated knowledge to help them solve many problems that may strike younger people as novel and complex (Salthouse, 2012); and many older people report a positive quality of life (Farquhar, 1995). Being retired, volunteer work, and social relationships with children, family and friends all have a positive impact on the older person’s life (Netuveli, Wiggins, Hildon, Montgomery & Blane, 2006).
Since our bodies and cognitive abilities show clear evidence of age-related change, it is not surprising that our brain also shows signs of ageing. There are at least two reasons to study brain ageing. Firstly, cognitive decline is considered an inevitable consequence of the ageing process, and this decline is tightly locked to changes in the integrity of the brain. By understanding the influence of age on the brain, we will develop better mechanistic models of how some people seem to age more successfully than others, and whether we can develop interventions to counteract age-related cognitive decline. Secondly, brain ageing is a strong risk factor for many neurodegenerative and psychiatric illnesses, with age serving as the greatest risk factor for illnesses such as Alzheimer’s disease (Guerreiro & Bras, 2015) and Parkinson’s disease (Reeve, Simcox & Turnbull, 2014). It is important to understand brain ageing not only so we can understand the mechanism of age as a disease risk factor, but also so we can distinguish healthy from pathological ageing, thereby initiating therapeutic interventions earlier in the disease process.
In this chapter, we will review the effects of ageing on the human brain, with particular focus on cortico-basal ganglia and cortico-cerebellar networks.
Cortico-basal ganglia and cortico-cerebellar circuits: structure and function
Cortico-basal ganglia circuits
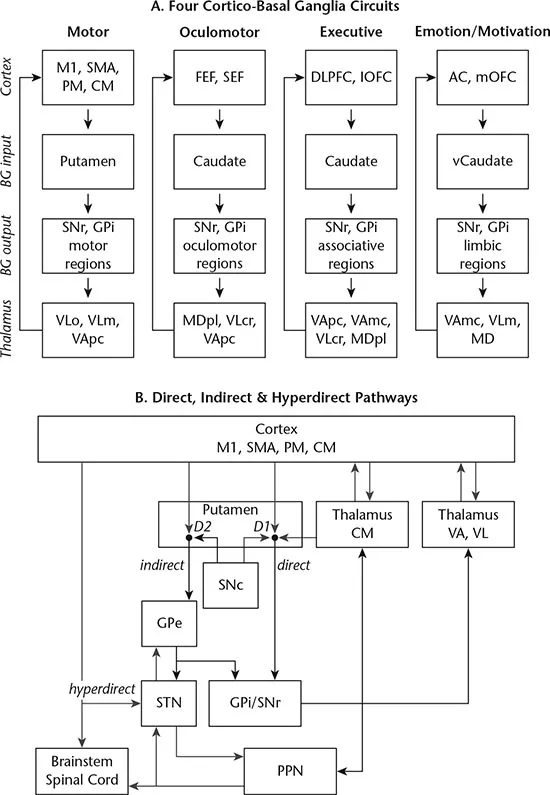
The basal ganglia comprise the striatum (caudate, putamen, nucleus accumbens), globus pallidus, substantia nigra, subthalamic nucleus and pons. The components of the basal ganglia are categorised according to their function within the cortico-basal ganglia circuits. The caudate, putamen and nucleus accumbens form the input nuclei of the basal ganglia; the globus pallidus pars interna and substantia nigra pars reticulata form the output nuclei; and the globus pallidus pas externa, subthalamic nucleus and substantia nigra pars compacta form the intrinsic or relay nuclei.
The output nuclei project to the thalamus and brain stem. The descending pallidal and nigral projections to the brainstem provide pathways for direct influence on motor circuits (e.g. oculomotor control via superior colliculus, locomotion via the pedunculopontine nucleus). The projections to the brainstem are also the likely location of cerebellar inputs to the basal ganglia via the pontine nuclei (Hoshi, Tremblay, Féger, Carras & Strick, 2005). Thalamic projections are directed to the ventral anterior, ventrolateral and intralaminar thalamic nuclei, which are then projected to the cortex (primarily frontal cortex). (Many excellent reviews of basal ganglia anatomy are available, including Lanciego, Luquin & Obeso, 2012; Wichmann & DeLong, 2013).
The basal ganglia form a number of segregated but parallel loops that originate in the cortex, innervate the thalamus via the basal ganglia, and then project back to the cortex. The cortico-basal ganglia motor loop is the best characterised, given that pathology within this network is associated with several major disorders of movement, including Parkinson’s disease and Huntington’s disease (see Chapters 3 and 4). A key feature of the cortico-basal ganglia networks is their closed-loop nature: the cortical regions that are the main inputs to the circuit are also the main target for outputs (Kelly & Strick, 2004). The circuits are labelled according to their functions, and include motor, oculomotor, executive and affective/motivation circuits (Figure 1.1A). Within each network, there are direct and indirect pathways of information flow (Figure 1.1B), with the direct pathway supporting activation and the indirect pathway supporting inhibition (DeLong & Wichmann, 2015). There is also evidence for a hyperdirect pathway within the motor circuit, which projects directly from the cortex to the subthalamic nucleus, conveying strong excitatory signals with faster velocity than the direct and indirect pathways (Nambu, 2008). This pathway seems to be important for cancelling motor plans (Nambu, Tokuno & Takada, 2002). Excitatory connections within the network (direct, indirect and hyperdirect) are glutamatergic, and inhibitory connections are GABAergic projections (ibid.). The opposing behaviour of the direct and indirect pathway is established by differing response to dopamine: direct pathway neurons are facilitated by dopamine D1 receptors, whereas the indirect pathway is inhibited by dopamine, through activation of the D2 receptors (Wichmann & DeLong, 2013).
Cortico-cerebellar circuits
The cerebellum comprises a series of highly regular repeating units, each of which contains the same basic cellular organisation (see Haines & Mihailoff, 2018; Lisberger & Thach, 2013 for reviews). The cerebellum has a highly convoluted outer layer of grey matter, and internal core of white matter, containing four deep cerebellar nuclei: the fastigial, emboliform, globose and dentate nuclei. The cerebellar cortex is macroanatomically divided into the midline vermis, lateral hemispheres and flocculonodular lobes. Signals to and from the brain stem, spinal cord and cortex enter the cerebellum via the three cerebellar peduncles: the inferior, middle and superior cerebellar peduncles. Most of the output of the cerebellum project via the cerebellar deep nuclei to the cerebellar peduncles; projections from the flocculonodular lobe to the vestibular nuclei in the brainstem are the exception to this pattern. Unlike cortical and subcortical brain regions, which process somatosensory and motor input from contralateral regions of the body, the cerebellum processes input from ipsilateral sensory organs and limbs. Thus, when the cerebellum receives input from the body and sensory organs via the cortex, the signals must again cross the midline from the contralateral to the ipsilateral side, given the originally crossed representation in the sensorimotor cortex.
The cerebellum can also be divided into three systems or modules on the basis of their inputs and presumed function (Figure 1.2A). The vestibulocerebellum receives inputs from the ipsilateral vestibular system (via primary and secondary vestibulocerebellar fibres) and visual areas and projects directly through the flocculonodular lobe to the brainstem vestibular nuclei (via vestibulospinal and reticulospinal tracts). This system is important for balance and eye movements. The spinocerebellum covers the medial regions of the cerebellum, the vermis and the intermediate sections of the cerebellar hemispheres. The vermal spinocerebellar system receives visual, auditory, vestibular and somatosensory input from the body (via the posterior and anterior spinocerebellar tracts and the cuneocerebellar fibres) and projects via the fastigial nucleus to the brainstem and cortex. The vermal spinocerebellum is important for posture, locomotion and eye movements. The adjacent, more lateral intermediate hemispheric spinocerebellar system also receives somatosensory input from the limbs but also the motor cortex, and output neurons project to the brainstem via the emboliform and globose nuclei. This intermediate hemispheric spinocerebellar network is responsible for the control of distal muscles of the limbs and fingers. The cerebrocerebellum encompasses the lateral cerebellar hemispheres and receives almost all of its input from the contralateral cortex via the pontine nuclei (pontocerebellar fibres). Cerebrocerebellar output projects back to the cortex (motor, premotor, prefrontal) via the contralateral dentate nucleus. This module is responsible for the planning and control of fine motor movements, and in the timing of these movements. There is also significant evidence to suggest that cerebrocerebellar fine control and timing processes are also applied to cognitive functions, including working memory, executive function, and language (Buckner, 2013).
The repeating structure and similarity of the cellular organisation and physiology suggests that similar computational processes are carried out throughout the cerebellum. While it was once controversial to suggest that the cerebellum played a role in non-motor processes, it is now widely agreed that the structure plays a role in cognitive processes (Buckner, 2013; Koziol et al., 2014). The dysmetria of thought hypothesis (Schmahmann, 1991) draws upon evidence showing that the cerebellum modulates the rate, rhythm, force and accuracy of motor outputs, and proposes that the structure also regulates the speed, consistency, capacity and appropriateness of cognitive and limbic processes. This ‘universal cerebellar transform’ (Schmahmann, 2000) integrates multiple internal representations of the body and external environment with external stimuli and self-generated responses to optimise cognitive and motor actions according to the current context. Central to this theory is the concept of the ‘internal model’: neural representations that encode input–output relationships between motor commands and their consequences that are acquired through experience-dependent learning (Ramnani, 2006). The internal forward model (Figure 1.2B) encodes and updates representations of input–output mappings learned in real-world situations via error feedback. In this model, when a signal is sent to generate an action, an eff...