![]()
Management of Specific Diseases
Oh H, Oshima Y (eds): Microincision Vitrectomy Surgery. Emerging Techniques and Technology.
Dev Ophthalmol. Basel, Karger, 2014, vol 54, pp 150-158 (DOI: 10.1159/000360461)
______________________
Idiopathic Macular Hole
Hideyasu Oh
Hyogo Prefectural Amagasaki Hospital, Amagasaki, Hyogo, Japan
______________________
Abstract
Idiopathic macular hole is a disease that arises from adhesion in the vitreomacular interface and can theoretically be treated by vitrectomy surgery. Surgical techniques include removal of the vitreous with or without simultaneous peeling of the internal limiting membrane (ILM), fluid-air exchange, and gas tamponade. Since the advent of microincision vitrectomy surgery, macular hole surgery has been performed with minimal invasiveness, and significant visual improvement is a common outcome. This chapter describes the pathology of this disease, including presurgical evaluation using optical coherence tomography (OCT), and then shows the fundamental techniques required for the surgery. Also important is the understanding of the postsurgical ‘healing’ process of the disease, which may confirm the fact that the subjective improvement is closely related to the retinal imaging obtained by OCT. More recent advances are the inverted ILM peeling technique for larger macular holes and 27-gauge vitrectomy that can potentially minimize the surgical invasiveness mainly by smaller wound construction and the reduced volume of irrigation during surgery.
© 2014 S. Karger AG, Basel
Macular holes had long been regarded as an incurable disease until Kelly and Wendel [1, 2] reported that vitrectomy in combination with intraocular gas tamponade can be the first modality to treat this disorder with a remarkable success rate. Gass [3, 4] first reported the potential pathogenesis of idiopathic macular holes based on biomicroscopic findings. He proposed that the vitreous cortex remaining on the macula after spontaneous posterior vitreous separation will contract and lead to tear formation at the macula in an eccentric fashion. After the advent of optical coherence tomography (OCT), investigators were able to show the presence of localized vitreomacular adhesion at the central fovea in stage 1 and stage 2 macular holes [5, 6]. The adhesion is surrounded by a shallow posterior hyaloid separation [called perifoveal posterior vitreous detachment (PVD)] [5, 7], and its existence has convinced observers that antero-posterior traction also plays a crucial role in pathogenesis [8].
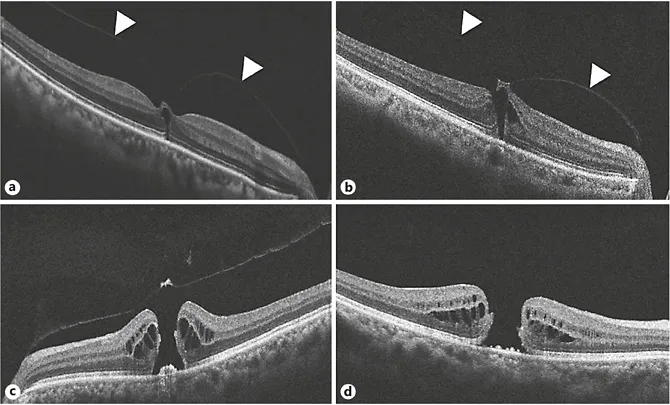
Based on these key findings, researchers adopted a OCT-based classification of macular holes, which is highly compatible in clinical settings (fig. 1): stage 1a = retinal split, foveal cyst or foveal detachment with a vitreous cortex attachment to the fovea; stage 1b = dehiscence of photoreceptor layer with a vitreous cortex attachment to the fovea; stage 2 = full-thickness hole covered by a flap-like tissue extending from the perifoveal retina; stage 3 = full-thickness hole with the detached vitreous, in which an operculum is suspended, and stage 4 = full-thickness hole with complete vitreous detachment.
Fig. 1. Spectral domain OCT image of macular holes: stage 1 (a), stage 2 (b), stage 3 (c), and stage 4(d). Note the perifoveal PVD (arrowheads).
More recently, the International Vitreomacular Traction Study (IVTS) Group newly proposed OCT-based anatomic definitions and classification of the closely related vitreomacular diseases and incorporated not only full-thickness macula holes, but also vitreomacular adhesion and vitreomacular traction [9]. In this new proposal, vitreomacular adhesion is defined as perifoveal vitreous separation with remaining vitreomacular attachment and unperturbed foveal morphology. In contrast, vitreomacular traction is characterized by anomalous vitreous detachment accompanied by anatomic distortion of the fovea, including pseudocyst, macular schisis, cystoid macular edema, and subretinal fluid.
Removing the vitreous is, considering the pathology of macular holes and the need for gas injection at the end of surgery, an ideal and requisite procedure. Nowadays, vitrectomy combined with internal limiting membrane (ILM) peeling is a standard technique for a vitreoretinal surgeon to master during early learning periods. In some cases - like that of the author - the mentor will start teaching by operating on this disease because the procedures encompass most skills required for vitrectomy surgery: core vitrectomy, induction of posterior hyaloid separation, ILM peeling, and gas tamponade.
Indications and Preoperative Evaluation
Stage I macular holes sometimes show spontaneous closure during physiological PVD. Hence, they can be followed up with close observation unless distortion or visual impairment is too severe for patients to tolerate. Stage 2 or higher macular holes are good indications for surgical intervention.
Preoperative examination should include visual acuity measurement, the Amsler chart to document and for patients to recognize metamorphopsia, slit-lamp biomicroscopy, and OCT. Especially in stage 2 macular holes, OCT findings are important not only for visualizing the extent of vitreomacular traction, but also for intraoperative maneuvers. If there is marked traction and the flap is highly elevated on OCT, it might be advisable to avoid pulling in the direction, as it could lead to further enlarging of the macular hole during posterior hyaloid separation. Also crucial is to always pay attention to the size of macular holes since they are known to have a substantial influence on anatomical success and visual prognosis [10, 11].
Surgical Techniques
Wound Construction
In the era of microincision vitrectomy surgery, angled incision is preferred to create the wound for trocar placement [12, 13] because it improves wound sealing and thus prevents postoperative complications such as hypotony and endophthalmitis [14-16]. Even with this technique, however, the sealing is not always complete [17] and wound-related complications can still occur. Therefore, careful detection of the leakage from the wounds is always advised during the removal of trocars, and the surgeon should not hesitate to place a suture if any trace of leakage is detected. One technique for better sealing is to press only the external flap of the wound with a forceps for a few seconds rather than to rub the entire wound back and forth repeatedly, which may conversely lead to reopening and impaired sealing.
Core Vitrectomy and Creation of Posterior Hyaloid Separation
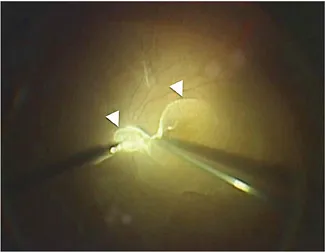
After the three-port trocar is inserted in place and the infusion cannula is connected, a wideviewing system such as BIOM® (Oculus) or RESIGHT® (Carl Zeiss Meditec) is introduced and then core vitrectomy is commenced. A wide-viewing system is nowadays an indispensable device for modern vitreous surgery since it commands a far wider fundus view compared to the conventional direct-viewing system using contact lenses. It saves the need to suture a fixation ring for contact lens and also makes it much easier for the surgeon to switch between viewing inside the eye and outside the eye - he/she only needs to push aside the metal frame that holds the objective lens. Following core vitrectomy, the peripheral vitreous should also be removed and during this process the illumination needs to be set closer to the cutter probe for better visualization of the vitreal collagen bundles that the surgeon is going to cut. After injecting a small volume of triamcinolone for visualization, posterior hyaloid separation is created by using either a backflush needle or a cutter probe. The latter is more useful in that it can be placed very close to the disc where a strong vitreal adhesion exists and can suck the vitreous from its side. An alternative way of creating the separation is to suck the vitreous cortex between the papillomacular bundle in cases with a high perifoveal PVD documented on preoperative OCT (fig. 2). After hyaloid separation from the posterior pole is achieved, it is important to move the cutter just anteriorly and not tangentially because this may pull the vitreous base and lead to potential formation of retinal breaks. On the other hand, insufficient completion of this step implies the existence of vitreal remnants on the retina, which potentially may lead to postoperative complications such as retinal breaks. Inappropriate completion may also interfere with the more delicate maneuvers like ILM peeling that follows.
Fig. 2. Visualization of posterior vitreous cortex by triamcinolone acetonide injection. Note the two well-stained sites (arrowheads), the bursa premacularis, and space of Martegiani. The cutter probe is placed at the perifoveal PVD, located between the disc ...