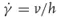![]()
Chapter 1
Rheology of Disperse Systems
Norbert Willenbacher and Kristina Georgieva
1.1 Introduction
The rheology of disperse systems is an important processing parameter. Being able to characterize and manipulate the flow behavior of dispersions one can ensure their optimal performance. Waterborne automotive coatings, for example, should exhibit a distinct low-shear viscosity necessary to provide good leveling but to avoid sagging at the same time. Then, a strong degree of shear thinning is needed to guarantee good pump- and sprayability. The rheological properties of dispersions, especially at high solids content, are complex and strongly dependent on the applied forces and flow kinematics. Adding particles does not simply increase the viscosity of the liquid as a result of the hydrodynamic disturbance of the flow; it also can be a reason for deviation from Newtonian behavior, including shear rate dependent viscosity, elasticity, and time-dependent rheological behavior or even the occurrence of an apparent yield stress. In colloidal systems particle interactions play a crucial role. Depending on whether attractive or repulsive interactions dominate, the particles can form different structures that determine the rheological behavior of the material. In the case of attractive particle interactions loose flocs with fractal structure can be formed, immobilizing part of the continuous phase. This leads to a larger effective particle volume fraction and, correspondingly, to an increase in viscosity. Above a critical volume fraction a sample-spanning network forms, which results in a highly elastic, gel-like behavior, and an apparent yield stress. Shear-induced breakup and recovery of floc structure leads to thixotropic behavior. Electrostatic or steric repulsion between particles defines an excluded volume that is not accessible by other particles. This corresponds to an increase in effective volume fraction and accordingly to an increase in viscosity. Crystalline or gel-like states occur at particle concentrations lower than the maximum packing fraction.
Characterization of the microstructure and flow properties of dispersions is essential for understanding and controlling their rheological behavior. In this chapter we first introduce methods and techniques for standard rheological tests and then characterize the rheology of hard sphere, repulsive, and attractive particles. The effect of particle size distribution on the rheology of highly concentrated dispersions and the shear thickening phenomenon will be discussed with respect to the influence of colloidal interactions on these phenomena. Finally, typical features of emulsion rheology will be discussed with special emphasis on the distinct differences between dispersion and emulsion rheology.
1.2 Basics of Rheology
According to its definition, rheology is the science of the deformation and flow of matter. The rheological behavior of materials can be regarded as being between two extremes: Newtonian viscous fluids, typically liquids consisting of small molecules, and Hookean elastic solids, like, for example, rubber. However, most real materials exhibit mechanical behavior with both viscous and elastic characteristics. Such materials are termed viscoelastic. Before considering the more complex viscoelastic behavior, let us first elucidate the flow properties of ideally viscous and ideally elastic materials.
Isaac Newton first introduced the notion of viscosity as a constant of proportionality between the force per unit area (shear stress) required to produce a steady simple shear flow and the resulting velocity gradient in the direction perpendicular to the flow direction (shear rate):
where
σ =
F/A is the shear stress,
η the viscosity, and the
is the shear rate. Here
A is the surface area of the sheared fluid volume on which the shear force
F is acting and
h is the height of the volume element over which the fluid layer velocity
v varies from its minimum to its maximum value. A fluid that obeys this linear relation is called
Newtonian, which means that its viscosity is independent of shear rate for the shear rates applied. Glycerin, water, and mineral oils are typical examples of Newtonian liquids. Newtonian behavior is also characterized by constant viscosity with respect to the time of shearing and an immediate relaxation of the shear stress after cessation of flow. Furthermore, the viscosities measured in different flow kinematics are always proportional to one another.
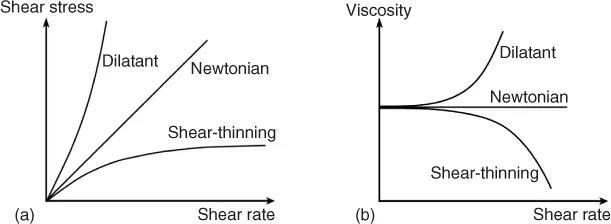
Materials such as dispersions, emulsions, and polymer solutions often exhibit flow properties distinctly different from Newtonian behavior and the viscosity decreases or increases with increasing shear rate, which is referred to a shear thinning and shear thickening, respectively. Figure 1.1a,b shows the general shape of the curves representing the variation of viscosity as a function of shear rate and the corresponding graphs of shear stress as a function of shear rate.
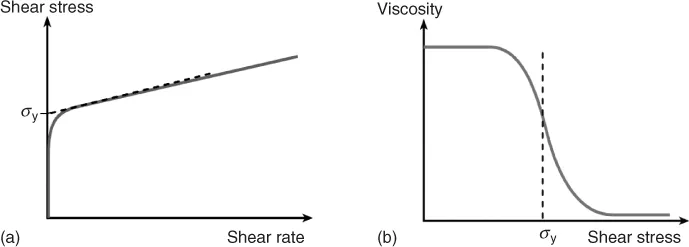
Materials with a yield stress behave as solids at rest and start to flow only when the applied external forces overcome the internal structural forces. Soft matter, such as, for example, dispersions or emulsions, does not exhibit a yield stress in this strict sense. Instead, these materials often show a drastic change of viscosity by orders of magnitude within a narrow shear stress range and this is usually termed an “apparent” yield stress (Figure 1.2a,b). Dispersions with attractive interactions, such as emulsions and foams, clay suspensions, and ketchup, are typical examples of materials with an apparent yield stress. Note that there are various methods for yield stress determination and the measured value may differ depending on the method and instrument used.
The flow history of a material should also be taken into account when making predictions of the flow behavior. Two important phenomena related to the time-dependent flow behavior are thixotropy and rheopexy. For materials showing thixotropic behavior the viscosity gradually decreases with time under constant shear rate or shear stress followed by a gradual structural recovery when the stress is removed. The thixotropic behavior can be identified by measuring the shear stress as a function of increasing and decreasing shear rate. Figure 1.3 shows a hysteresis typical for a thixotropic fluid. Examples of thixotropic materials include coating formulations, ketchup, and concentrated dispersion...