
- 345 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Plastics Failure Analysis and Prevention
About this book
This book contains analysis of reasons that cause products to fail. General methods of product failure evaluation give powerful tools in product improvement. Such methods, discussed in the book, include practical risk analysis, failure mode and effect analysis, preliminary hazard analysis, progressive failure analysis, fault tree analysis, mean time between failures, Wohler curves, finite element analysis, cohesive zone model, crack propagation kinetics, time-temperature collectives, quantitative characterization of fatigue damage, and fracture maps. Methods of failure analysis are critical to for material improvement and they are broadly discussed in this book. Fractography of plastics is relatively a new field which has many commonalities with fractography of metals. Here various aspects of fractography of plastics and metals are compared and contrasted. Fractography application in studies of static and cycling loading of ABS is also discussed. Other methods include SEM, SAXS, FTIR, DSC, DMA, GC/MS, optical microscopy, fatigue behavior, multiaxial stress, residual stress analysis, punch resistance, creep-rupture, impact, oxidative induction time, craze testing, defect analysis, fracture toughness, activation energy of degradation.Many references are given in this book to real products and real cases of their failure. The products discussed include office equipment, automotive compressed fuel gas system, pipes, polymer blends, blow molded parts, layered, cross-ply and continuous fiber composites, printed circuits, electronic packages, hip implants, blown and multilayered films, construction materials, component housings, brake cups, composite pressure vessels, swamp coolers, electrical cables, plumbing fittings, medical devices, medical packaging, strapping tapes, balloons, marine coatings, thermal switches, pressure relief membranes, pharmaceutical products, window profiles, and bone cements.
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Chapter 1
Failure Mechanisms
Plastics Failure Due to Oxidative Degradation in Processing and Service
Myer Ezrin, Amanda Zepke, John Helwig, Gary Lavigne and Mark Dudley, University of Connecticut, Institute of Materials Science, Storrs, CT 06269-3136, USA
INTRODUCTION
People cannot live without oxygen and water. But these are deadly enemies of polymers, both in processing of plastics formulations and in service. Water is a problem mainly for condensation polymers which degrade by hydrolysis. In this paper the focus is on oxidative degradation.
Oxygen degrades polymers to lower molecular weight (MW) by reacting with polymer free radicals to form peroxy free radicals (ROO•) and hydroperoxides (ROOH). Free radicals have an unshared electron and react in any way they can to restore the atom or molecule to a balanced structure. Often that leads to chain scission. As MW goes down most polymer properties suffer. As little as 5-10% reduction in MW may cause failure. Avoiding contact with oxygen and using an antioxidant (AO) as a free radical scavenger are means of preventing degradation.
The high temperature required to process plastics is the major cause of degradation in injection molding, extrusion, blow molding, etc. High temperature is needed to fuse polymers and to reduce melt viscosity to a level that the machines can handle. Mechanical shear of the melt and the presence of oxygen, even in small amounts, are major factors in degradation due to processing. The chain carbon atoms attached to a branch, such as methyl group (CH3), tend to split off a hydrogen atom, creating a free radical at a tertiary carbon atom.
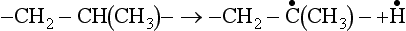
Very little oxygen is needed to react with free radicals during processing. Polymer suppliers usually have very little AO in the resin as sold to processors. Unless additional AO is added, polymer is likely to degrade in process. Polyolefins, which have only carbon-carbon chain bonds (PE, PP, EP and other copolymers) are particularly susceptible to oxidative degradation, in service as well as in processing. Even if additional AO is added, severe processing conditions (high temperature, high shear, long residence time in the barrel), use of regrind, etc. may deplete most of the AO, leaving too little to withstand conditions in service.
Even in moderate service conditions, such as a PE eyewash squeeze bottle on a laboratory wall, oxidative degradation can lead to failure in long term applications. Such a PE bottle, which had been on a laboratory wall for 15-20 years, cracked when tested in a safety inspection. Here, too, additional AO is needed to survive many years of service.
A complicating factor in processing is formulations containing peroxides to crosslink the polymer. Peroxide causes crosslinking by decomposing to free radicals (ROOR → 2RO). The high content of peroxy free radicals formed abruptly reacts with the polymer to cause crosslinking. These free radicals may react with the AO, leaving the system without enough AO for the polymer to survive processing and service. The AO system must be chosen accordingly.
Commonly used methods of analysis to determine if failure is due to oxidative degradation are differential scanning calorimetry (DSC) for oxidative induction time (OIT), (ASTM D3895) or oxidative induction temperature (ASTM D3350). Infrared spectroscopy (IR) may detect bound oxygen as carbonyl (C=O), which forms increasingly as AO becomes exhausted. A third method is change in MW measured as an increase in melt flow rate (MFR), (ASTM D1238). This is a very practical method because it relates directly to MW, i.e., a small reduction in MW gives a large increase in MFR. The applicable relationship is n=KM3.4. Gel permeation chromatography (GPC) is also useful for monitoring MW changes in processing or service.
The DSC methods require about an hour or less, after establishing test conditions, and are most useful for comparing materials, e.g., before and after processing, or after service. They are a practical method of determining the relative amount of AO remaining. When a sample’s AO content is zero, oxidation exotherm starts very soon after oxygen is admitted into the DSC cell. Additional information on the DSC methods is given in the next section. IR is useful mainly to detect bound oxygen, which occurs when most or all of the AO has been depleted.
Examples are given below of failure due to oxidative degradation for (1) HDPE power cable jacket; (2) PE low voltage cable in a power plant control room; (3) PP rotors in a hot water system; (4) EPDM hot water check valve; and (5) EVA (ethylene vinylacetate) hot melt adhesive degraded in a heated reservoir.
A recent case of PP failure in hot water heaters, most likely due to oxidative degradation, was reported in Consumer Reports, July 1999, p. 8. PP that replaced copper dip tubes brings cold water to the bottom of the heater. The PP has been disintegrating into small pieces and clogging pipes and other water delivery systems, and preventing normal operation of the hot water heater. Class action law suits have been filed in some states. Sixteen million heaters were made between 1993 and 1996 with PP dip tubes that may be defective.
EXPERIMENTAL METHODS
DSC - OXIDATIVE INDUCTION TIME AND OXIDATIVE INDUCTION TEMPERATURE
The OI time method requires selecting an isothermal temperature, first equilibrated in nitrogen, then changing to oxygen for the test. For PE and PP a common temperature is 200°C. The OI temperature method is much simpler, because a routine programmed temperature run is made using oxygen from the beginning. At some temperature an exotherm will be observed. OI time testing is critically dependent on selecting an appropriate isothermal temperature. Switching from nitrogen to oxygen may cause an upset in the baseline which could complicate interpretation of the result, i.e., did the exotherm start right away (zero minutes OIT) or is it at a higher value, which may be difficult to detect with some polymers.
The instrument used was a TA Instruments model 2920. The OI time method generally followed the guidelines of ASTM D3895. The isothermal temperature depends some-what on the material being tested, selected to give a time to exotherm of about 60 minutes for the most highly stabilized samples. Surface to volume ratio and sample weight affect the response to oxygen, affecting how readily and reproducibly the OI time is determined. The ASTM method calls for extrapolation of the initial part of the exotherm to the baseline (see DSC figures below). Extrapolation introduces some variability because not all samples in a group have similar exotherm slopes. A more realistic measure of OI time or OI temperature is the initial onset time or temperature (see DSC figures below). That is when the reaction with oxygen starts. Since the deflection off the baseline is often slight and gradual, a common or standard method needs to be used to detect the initial onset point because the temperature or time is not important by itself, only in comparison with similar samples with different process or service history. Uniform contact with the sample pan bottom is ideal. If OI time is required, an OI temperature run is helpful in selecting an appropriate isothermal temperature.
MOLECULAR WEIGHT-RELATED METHODS
Melt Flow Rate - ASTM D1238
If a sample no longer has AO, some degradation may occur in the MFR test at elevated temperature. That will give a value which is the sum of the change due to the sample’s pretest history and its MFR test. This effect can be judged by running the test at various heating times in the barrel before extrusion. If MFR goes up as test time increases, ...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- Preface
- Chapter 1: Failure Mechanisms: Plastics Failure Due to Oxidative Degradation in Processing and Service
- Durability Study of Conductive Copper Traces Within Polyimide Based Substrates
- Fatigue Behavior of Discontinuous Glass Fiber Reinforced Polypropylene
- Ductile Failure and Delayed Necking in Polyethylene
- Chapter 2: Processing and Assembly: The Role of a Heat Affected Zone (HAZ) on Mechanical Properties in Thermally Welded Low Density Polyethylene Blown Film
- Effects of Processing Conditions on the Failure Mode of an Aliphatic Polyketone Terpolymer
- Orientation Effects on the Weldability of Polypropylene Strapping Tape
- Joint Performance of Mechanical Fasteners under Dynamic Load - Self-Tapping Screws in Comparison with Threaded Inserts in Brass and Plastic
- Defect Cost Analysis
- Chapter 3: Environmental Effects: Environmental Stress Cracking (ESC) of ABS (II)
- Residual Stress Development in Marine Coatings Under Simulated Service Conditions
- Estimation of Long-term Properties of Epoxies in Body Fluids
- Mechanical Performance of Polyamides with Influence of Moisture and Temperature – Accurate Evaluation and Better Understanding
- Temperature-Moisture-Mechanical Response of Vinyl Ester Resin and Pultruded Vinyl Ester/E-Glass Laminated Composites
- Freeze-thaw Durability of Composites for Civil Infrastructure
- Chapter 4: Morphology and Fractography: Fractography of ABS
- Fractography of Metals and Plastics
- Crack Propagation in Continuous Glass Fiber/Polypropylene Composites: Matrix Microstructure Effect
- Fracture Behavior of Polypropylene Modified with Metallocene Catalyzed Polyolefin
- Morphology and Mechanical Behavior of Polypropylene Hot Plate Welds
- The Influence of Morphology on the Impact Performance of an Impact Modified PP/PS Alloy
- Morphological Study of Fatigue Induced Damage in Semi-crystalline Polymers
- Chapter 5: Modelling of Failures and Failure Processes: Failure Analysis Models for Polyacetal Molded Fittings in Plumbing Systems
- Progressive Failure Analysis of Fiber Composite Structures
- Calculating Thermally Induced Stresses Using a Nonlinear Viscoelastic Material Model
- Evaluation of a Yield Criteria and Energy Absorbing Mechanisms of Rubber Modified Epoxies in Multiaxial Stress States
- Chapter 6: Design and Life Prediction: Shelf Life Failure Prediction Considerations for Irradiated Polypropylene Medical Devices
- Determining Etch Compensation Factors for Printed Circuit Boards
- Activation Energies of Polymer Degradation
- Estimation of Time-temperature-collectives at Describing Ageing of Polymer Materials
- Chapter 7: Test Methods: Standard Test Procedures for Relevant Material Properties for Structural Analysis
- Factors Affecting Variation in Gardner Impact Testing
- Radiation Resistance of Multilayer Films by Instrumented Impact Testing
- Aspects of the Tensile Response of Random Continuous Glass/Epoxy Composites
- Comparing the Long Term Behavior of Tough Polyethylenes by Craze Testing
- Chapter 8: Failure Prevention: Design Aids for Preventing Brittle Failure in Polycarbonate and Polyetherimide
- Common Pitfalls in Thin-Wall Plastic Part Design
- Defect Analysis and High Density Polyethylene Pipe Durability
- Practical Risk Analysis - As a Tool for Minimizing Plastic Product Failures
- Attachment Design Analysis of a Plastic Housing Joined with Snap-fits
- Avoiding the GIGO Syndrome – Combining the Real and Virtual Worlds in Analysis of Polymer Product Failures
- Chapter 9: Case Studies: Case Studies of Inadvertent Interactions Between Polymers and Devices in Field Applications
- Case Studies of Plastics Failure Related to Improper Formulation
- Translating Failure Into Success – Lessons Learned From Product Failure Analysis
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Plastics Failure Analysis and Prevention by John Moalli in PDF and/or ePUB format, as well as other popular books in Technology & Engineering & Materials Science. We have over 1.5 million books available in our catalogue for you to explore.