This book is a unique guide to emerging stem cell technologies and the opportunities for their commercialisation. It provides in-depth analyses of the science, business, legal, and financing fundamentals of stem cell technologies, offering a holistic assessment of this emerging and dynamic segment of the field of regenerative medicine.
• Reviews the very latest advances in the technology and business of stem cells used for therapy, research, and diagnostics
• Identifies key challenges to the commercialisation of stem cell technology and avenues to overcome problems in the pipeline
• Written by an expert team with extensive experience in the business, basic and applied science of stem cell research
This comprehensive volume is essential reading for researchers in cell biology, biotechnology, regenerative medicine, and tissue engineering, including scientists and professionals, looking to enter commercial biotechnology fields.

eBook - ePub
Stem Cells in Regenerative Medicine
Science, Regulation and Business Strategies
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Stem Cells in Regenerative Medicine
Science, Regulation and Business Strategies
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Part I
The stem cell business
Chapter 1
Therapeutic Stem Cells Answer a Strategic Breakthrough Need of Healthcare
Alain A. Vertès
London Business School, London, UK
Introduction
The Tractatus de Herbis (Anonymous, 1440) is one of the earliest dictionaries ever written to provide the names and pictures of ‘simples’, that is, the medicinal plants used during the Middle Ages in everyday therapeutic practice (Riddle, 1974). From this plant-based approach to treating human and animal ailments, the pharmaceutical industry has developed through a process that first aimed at isolating active pharmaceutical principles from extracts. The most telling examples here are perhaps the case of licorice roots (Figure 1.1), reportedly efficacious in curing a number of diseases from the common cold to liver diseases, that has been used in Europe since pre-historic times (Fiorea et al., 2005), or more recently the bark of the cinchona tree that contains quinine, and in Europe that of the willow tree that contains salicin, and the development of aspirin as a modern analgesic drug prepared as pure acetylsalicylic acid, produced on an industrial scale and marketed for the first time in 1899 by the German firm of Friedrich Bayer & Co (Elberfeld, Germany) (now Bayer AG, Leverkusen, Germany) (Tainter, 1948; Sneader, 2000; Brune and Hinz, 2004; Lukovic et al., 2014). Despite having decreased in importance due to the deployment of high throughput techniques to identify and optimise small molecules that act upon targets of well-defined mechanisms of action, natural products still remain a source of important drugs as recently exemplified by the discovery in 1966 of taxol, a compound produced by endophytic fungi in the bark of the Pacific yew tree (Nicolaou et al., 1994). Notably, ethnobotanic medicine, which encompasses the healing traditions of populations worldwide, remains to this day relevant in drug discovery (Fabricant and Farnsworth, 2001). In the foundational years of the modern pharmaceutical industry, pure chemicals were soon being produced by chemical synthesis as a necessity, given the difficulty in procuring the biological raw materials from the Orient and South America, particularly triggered by the blockade of the Continent during the Napoleonic Wars (Crouzet, 1964), to produce drugs such as quinine and morphine (Brune and Hinz, 2004). This first transformation was facilitated by earlier developments in chemistry achieved particularly for the production of dyes along the Rhine in the cities of Basel, Frankfurt, and Köln, which served as the cradle of the modern pharmaceutical industry through a combination of critical success factors comprising skilled workers, a plentiful water resource and easy transportation at the crossroads of several countries representing distinct markets (ibid.).The rise and improvements in ancillary technologies and sciences, such as pharmacology, molecular biology, cell biology, microbiology, human genetics, robotics, as well as bioinformatics have further paved the way for the development of drugs of increasing safety and efficacy to treat an array of indications of increasing complexity. These advances have promoted the emergence and maturation of several technological platforms to develop novel pharmaceutical modalities (Figure 1.2).
Figure 1.1 Liquorice. From folio f. 53v of the Tractatus de Herbis © M. Moleiro Editor (www.moleiro.com). Reproduced with permission (see plate section for color representation of this figure).
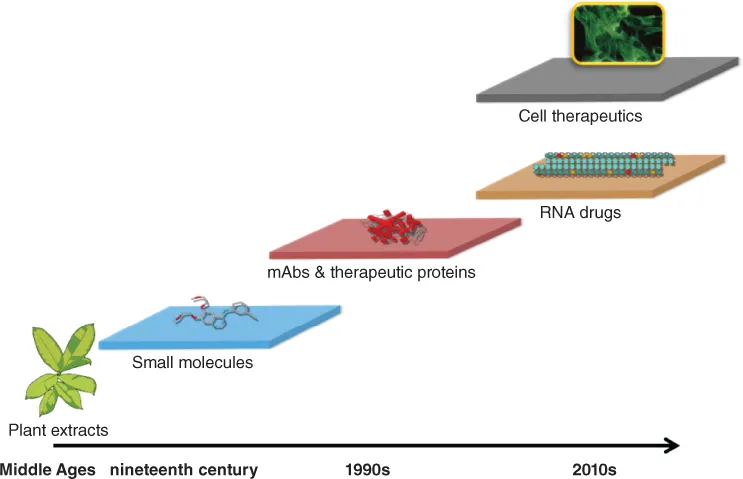
Figure 1.2 Platform technologies that have supported the development of pharmaceuticals throughout the ages. The pharmaceutical industry is deeply rooted in chemistry; however, novel technological platforms have emerged in recent years that have enabled medical practitioners to treat diseases which remained largely intractable using small molecules. In particular, the technology of monoclonal antibodies (mAbs) has revolutionised healthcare since the commercialisation of the first molecule of this class in the late 1990s (Brodsky, 1988; Pescovitz, 2006; Nelson et al., 2010; Buss et al., 2012). Other biotechnological products such as therapeutic proteins are now also part of the pharmacopeia (Pavlou and Reichert, 2004). Nucleic acids drugs (e.g. siRNAs, miRNAs, RNA aptamers, antisense oligonucleotides) and cell therapeutics (e.g., mesenchymal stem cells, hematopoietic stem cells, pluripotent stem cell-derived cells and tissues, tissue-specific stem cells, T-cells and engineered T-cells as well as NK cells) constitute novel pharmaceutical modalities that should come of age starting in the 2010 decade (Opalinska and Gewirtz, 2002; Pecot et al., 2011; Daley, 2012).
Strategic breakthrough need
The greatest challenge in medicine is to develop drugs with positive risk vs. clinical benefits ratios and to understand the bases of adverse reactions to drugs. The first biotechnological embodiment of the properties of stem cells was to enable the development of safer drugs using: (1) hepatocytes and cardiomyocytes to unravel toxicities of compounds in development earlier in the discovery process; (2) cells derived from iPS cells sourced from patients to better reproduce the biology of diseases; and (3) mini-organs, generated, for example, by bioprinting technologies, to enable testing compounds in development on a chip or under the native three-dimensional architectures of organs (Mironov et al., 2003; Nishikawa, Goldstein and Nierras, 2008; Jensen, Hyllner and Bjorquist, 2009; Baker, 2011; Wobus and Löser, 2011). These technologies are already being used in the research laboratories of academic or industrial laboratories (Vertès, 2010). On the other hand, one of the recent developments of this renewed strategic focus of the pharmaceutical industry is encompassed in the concept of personalised medicine, which aims to provide the right treatment to the right patient at the right time, so as to maximise efficacy while minimising adverse side effects and optimising the economic aspects of healthcare (Hamburg and Collins, 2010; Towse and Garrison, 2011). This challenge constitutes the strategic breakthrough need that must be addressed in the coming decade.
The ‘magic bullet’ concept revisited
This need to develop personalised and tailored drugs that maximise efficacy and reduce side effects by precisely targeting specific infectious organisms or molecular defects but not the host tissue, for example, in a cancer patient, was first advocated by Paul Ehrlich (Winau, Westphal and Winau, 2004; Strebhardt and Ullrich, 2008). These ‘magic bullets’ would comprise essentially two functional elements: the first functional group would recognise and bind to its targets, while the second would provide the therapeutic action. Immunotoxins have been notably developed using this basic architecture (Brodsky, 1988; Torchilin, 2000).
The pharmaceutical industry of today relies on several technological platforms, with the technology of small molecules having the longest tradition of use. Biologics, therapeutic proteins comprising enzymes and most importantly monoclonal antibodies (mAbs), represent a class of pharmaceuticals that has gained a strong foothold in the market since the beginning of the genetic engineering era in the early 1980s, a technological deployment that has accelerated in the late 1990s to take its full place in the pharmacopeia in the mid-2000s (Galambos and Sturchio, 1998).
Inventing treatments of the future is a complex process. The first step is to define the ideal target product profile that the novel drug needs to exhibit, comprising elements related to reduced toxicity, increased efficacy, or easier delivery as compared to the standard of care. Target product profiles can be very specific and with well-quantified thresholds. Notably, the standard of care is typically a moving target, and this dynamics needs to be forecasted early in the process when designing clinical trials and particularly when selecting endpoints, since the new drug could become obsolete even before it reaches the market. An example here is ...
Table of contents
- Cover
- Title Page
- Copyright
- Table of Contents
- List of contributors
- Foreword
- Preface
- Part I: The stem cell business
- Part II: Stem cells as research tools
- Part III: Stem cells as veterinary medicines
- Part IV: Stem cell therapeutics
- Part V: Perspectives
- Index
- End User License Agreement
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Stem Cells in Regenerative Medicine by Alain A. Vertes, Nasib Qureshi, Arnold I. Caplan, Lee E. Babiss, Alain A. Vertes,Nasib Qureshi,Arnold I. Caplan,Lee E. Babiss in PDF and/or ePUB format, as well as other popular books in Biological Sciences & Biotechnology. We have over 1.5 million books available in our catalogue for you to explore.