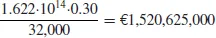![]()
1
INTRODUCTION
Drying can be defined as a unit operation in which a liquid–solid separation is accomplished by the supply of heat, with separation resulting from the evaporation of liquid. Although in the majority of cases water is the liquid being removed, solvent evaporation is also encountered. The definition may be extended to include the dehydration of food, feed, and salts, and the removal of hydroxyl groups from organic molecules.
This book is based on my personal experience gained in the selection and operation of drying equipment while employed by Akzo Nobel, a multinational company that manufactured, at that time, bulk and fine chemicals, pharmaceuticals, and coatings. Since 2000, I gained experience while working as an independent consultant.
Laboratory measurements and investigations concerning the drying of a product should be the first stage of the selection of a new dryer or the replacement of one. This aspect is discussed in Chapter 3. During the next stage, a person should seek the cooperation of a reputable dryer manufacturer. Close cooperation between the manufacturer and the potential user is essential, because one partner is knowledgeable about the equipment and the other person has expertise regarding the product. Since small-scale testing of drying equipment can be carried out, such testing can provide valuable insight into ultimate dryer selection. However, it is important that each partner have some insight into the other's field so that the user can develop value judgments on the equipment being recommended by the manufacturer. The size of the equipment must be checked, using various techniques (e.g., estimating methods, rules of thumb, rough-and-ready calculations). This book covers these techniques for each class of dryer.
Various reasons exist for drying materials to a specific level or range:
1. It is often necessary to obtain a free-flowing material that can be stored, packed, transported, or dosed.
2. Contractual limits exist for many products (e.g., salt, sand, yarn).
3. Statutory limits are in force for some materials (e.g., tobacco, flour).
4. A moisture content within a specified range may have to be obtained for quality control purposes. For many dried foods and feeds, too much moisture may adversely affect shelf life and nutritional value, whereas a moisture content too low, due to overdrying, may cause the loss of valuable nutrients. Moisture contents that are either too high or too low may render a product less enjoyable.
5. The feasibility of subsequent process steps sometimes requires that the moisture content be between specified limits, as in the milling of wheat or the pressing of pharmaceutical tablets. Another example is the low moisture content of rubber chemicals to be used in the vulcanization process of tires. Too much moisture causes the formation of blisters.
6. The onset of mildew and bacterial growth in such textiles as woolen cloth can be prevented by drying the cloth to a specific moisture content.
7. A drying step can be used as a shaping step. The manufacture of fluid cracking catalysts is an example. A spray-drying step produces hard and dry spheres of average diameter 80 μm. However, next, the spheres are leached with water to remove sodium salts. That step is followed by filtration and flash drying.
Typical dryer feeds are:
1. Objects (e.g., bricks)
2. Particulate materials (e.g., sodium sulfate crystals)
3. Filter and centrifuge cakes
4. Sheet material (e.g., paper for newspapers)
5. Pastes (e.g., dibenzoyl peroxide paste)
6. Liquids (i.e., solutions, emulsions, or suspensions)
Drying is an energy-intensive process. In general, heating and evaporation require large quantities of energy. An apple of mass 100 g hanging 4 m above the ground has a potential energy of approximately 4 J. Heating 1 kg of water from 15°C to 100°C requires 356,150 J. Evaporating 1 kg of water at 100°C and atmospheric pressure requires 2,285,000 J. Thus, in terms of energy, thermal effects are in general much more important than mechanical effects. This explains why the energy consumption in phase transformation and the heating in a drying operation exceeds the energy consumption of electromotors. In this respect, there is one more important aspect. The energy to evaporate 1 kmol of liquid is approximately constant for all liquids. Thus, it is possible to evaporate 18 kg of water (which has a kilomolecular weight of 18 kg·kmol−1) with this heat of evaporation. However, it is also possible to evaporate 92 kg of toluene (which has a kilomolecular weight of 92 kg·kmol−1) with this amount of heat. The explanation is that kilomoles of different substances contain the same number of molecules: 6.023·1026 (Avogadro's number). Thus, on evaporating 1 kmol of a substance, the bonds between this number of molecules must be broken. The bonds between the molecules are relatively weak Van der Waals forces and are approximately equal. The evaporation of water occurs more frequently than the evaporation of organic liquids.
The energy consumption of the drying operation in the UK has been reviewed by Bahu and Kemp [1]:
- The energy consumption of drying is 8% of the industrial energy consumption. The industrial energy consumption comprises both processes and buildings.
- The annual water evaporation amounts to 2·1010 kg. This is equivalent to 100-m water columns on 27.2 soccer fields (70·105 m2). As the U.S. economy is about 5.5 times larger than the UK economy, the annual water evaporation in the United States due to drying could be 1.1·1011 kg.
- In 1981, drying required 1.622·1014 kJ. This figure was possibly 10 to 20% lower in 1991.
- (1.622 · 1014)/(2 · 1010) = 8110 kJ per kilogram of evaporated water. This consumption figure includes electricity. Excluding electricity, the consumption figure is possibly 7000 kJ·kg−1. Compared to the heat of evaporation of water at 0°C and atmospheric pressure (i.e., 2500 kJ·kg−1), the consumption figure is quite high. In the chapters to come, the background of this state of affairs is discussed.
- Annual costs are determined by taking 32,000 kJ·nm−3 as the lower heating value of natural gas. The lower heating value is relevant if the heat of condensation of the water vapor in the combustion gases is not recovered. In the UK, an industrial price of €0.30 is typical:
These calculations illustrate that drying is an expensive means of accomplishing a liquid–solid separation; as a rule of thumb, 2 to 3 kg of steam is required for the evaporation of each kilogram of water. In a four-effect evaporation plant, approximately 4 kg of water can be evaporated with 1 kg of steam. Furthermore, performing a solid–liquid separation by means of a centrifuge or filter is usually much cheaper than using a dryer. Calculations concerning the energy required by the drying process begin with an assessment of the enthalpy difference between the process flows leaving the dryer and the process flow entering the dryer. Enthalpy differences are heat effects at constant pressure. In convective drying processes, the drying gas should be excluded from these calculations. Thus, the net heat is arrived at.
The heat required for drying can be supplied by the fundamentally different mechanisms of convection, conduction, and radiation:
1. Convection. A carrier gas (usually, air) supplies the heat for the evaporation of the liquid by the conversion of sensible heat into latent heat. The carrier gas subsequently entrains the volatile matter.
2. Conduction. The heat is supplied indirectly and the carrier gas serves only to remove the evaporated liquid. Typically, the airflow is approximately 10% of the airflow used in a convective process. Conduction of heat is the heat transport mechanism at contact drying.
3. Radiation. This type of drying can, in principle, be nonpenetrating, such as the drying of paint by infrared radiation, or penetrating, such as the drying of food or pharmaceuticals by dielectric drying. Dielectric drying (radio-frequency drying and microwave drying) is the only process in which heat is developed in the material being dried rather than having heat diffused into the material. Again a carrier gas is required to remove the evaporated liquid.
A combination of two mechanisms may be encountered in some dryer types.
The situation in the United States was analyzed by Strumillo and Lopez-Cacicedo [2], who found that 99% of dryer energy consumption could be attributed to six dryer types. In order of importance:
- Flash dryer
- Spray dryer
- Cylinder dryer for paper
- Convective rotary dryer
- Contact rotary dryer
- Fluid-bed dryer
This list illustrates that in terms of tonnage, convective drying is more important than conduction (contact) drying.
Dryer Types
A great variety of dryer types is commercially available. The reasons are as follows:
- Different products have very different drying times.
- The product quality often requires a certain dryer type or mode.
- It is often necessary to transport particulate material through a dryer.
A distinction should be made between free and bound moisture. Initially, free water is evaporated until the critical moisture content is reached. Free water's latent heat of evaporation is essentially equal to that of water on evaporating from a pool, with the heat transfer being the rate-determining step. Evaporation occurs at a constant rat...