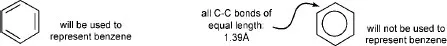![]()
1
Heterocyclic Nomenclature
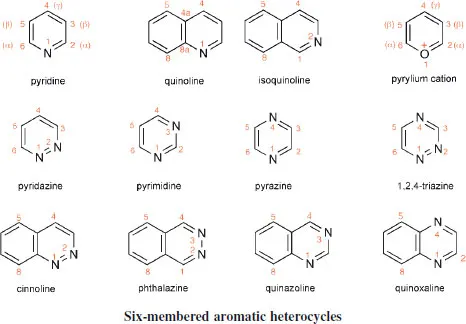
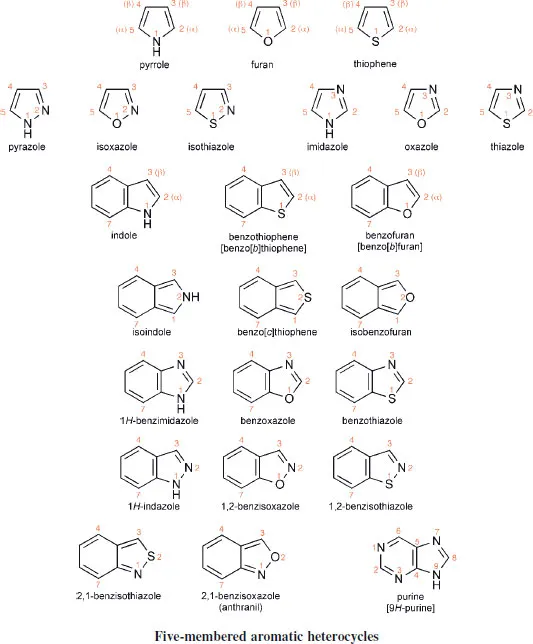
A selection of the structures, names and standard numbering of the more common heteroaromatic systems and some common non-aromatic heterocycles are given here as a necessary prelude to the discussions which follow in subsequent chapters. The aromatic heterocycles have been grouped into those with six-membered rings and those with five-membered rings. The names of six-membered aromatic heterocycles that contain nitrogen generally end in ‘ine’, though note that ‘purine’ is the name for a very important bicyclic system which has both a six- and a five-membered nitrogen-containing heterocycle. Five-membered heterocycles containing nitrogen general end with ‘ole’. Note the use of italic ‘H’ in a name such as ‘9H-purine’ to designate the location of an N-hydrogen in a system in which, by tautomerism, the hydrogen could reside on another nitrogen (e.g. N-7 in the case of purine). Names such ‘pyridine’, ‘pyrrole’, ‘thiophene’, originally trivial, are now the standard, systematic names for these heterocycles; names such as ‘1,2,4-riazine’ for a six-membered ring with three nitrogens located as indicated by the numbers, are more logically systematic.
A device that is useful, especially in discussions of reactivity, is the designation of positions as ‘α’, ‘β’, or ‘γ’. For example, the 2- and the 6-positions in pyridine are equivalent in reactivity terms, so to make discussion of such reactivity clearer, each of these positions is referred to as an ‘α-position’. Comparable use of α and β is made in describing reactivity in five-membered systems. These useful designations are shown on some of the structures. Note that carbons at angular positions do not have a separate number, but are designated using the number of the preceding atom followed by ‘a’-as illustrated (only) for quino-line. For historical reasons purine does not follow this rule.
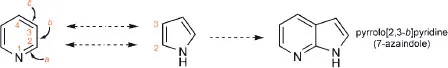
A detailed discussion of the systematic rules for naming polycyclic systems in which several aromatic or heteroaromatic rings are fused together is beyond the scope of this book, however, a simple example will serve to illustrate the principle. In the name ‘pyrrolo[2,3-b]pyridine’, the numbers signify the positions of the first-named heterocycle, numbered as if it were a separate entity, which are the points of ring fusion; the italic letter, ‘b’ in this case, designates the side of the second-named heterocycle to which the other ring is fused, the lettering deriving from the numbering of that heterocycle as a separate entity, i.e. side a is between atoms 1 and 2, side b is between atoms 2 and 3, etc. Actually, this particular heterocycle is more often referred to as ‘7-azaindole’-note the use of the prefix ‘aza’ to denote the replacement of a ring carbon by nitrogen, i.e. of C-7-H of indole by N.
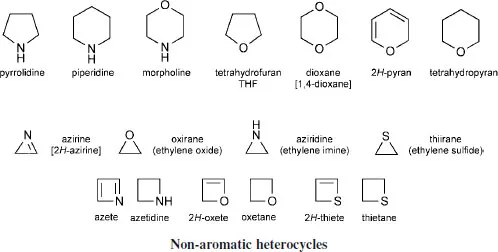
The main thrust of this book concerns the aromatic heterocycles, exemplified above, however Chapter 30 explores briefly the chemistry of saturated or partially unsaturated systems, including three- and four-membered heterocycles.
![]()
2
Structures and Spectroscopic Properties of Aromatic Heterocycles
This chapter describes the structures of aromatic heterocycles and gives a brief summary of some physical properties.1 The treatment we use is the valence-bond description, which we believe is appropriate for the understanding of all heterocyclic reactivity, perhaps save some very subtle effects, and is certainly sufficient for a general textbook on the subject. The more fundamental, molecular-orbital description of aromatic systems is less relevant to the day-to-day interpretation of heterocyclic reactivity, though it is necessary in some cases to utilise frontier orbital considerations,2 however such situations do not fall within the scope of this book.
2.1 Carbocyclic Aromatic Systems
2.1.1 Structures of Benzene and Naphthalene
The concept of aromaticity as represented by benzene is a familiar and relatively simple one. The difference between benzene on the one hand and alkenes on the other is well known: the latter react with electrophiles, such as bromine, easily by addition, whereas benzene reacts only under much more forcing conditions and then typically by substitution. The difference is due to the cyclic arrangement of six π-electrons in benzene: this forms a conjugated molecular-orbital system which is thermodynamically much more stable than a corresponding non-cyclically conjugated system. The additional stabilisation results in a diminished tendency to react by addition and a greater tendency to react by substitution for, in the latter manner, survival of the original cyclic conjugated system of electrons is ensured in the product. A general rule proposed by Hückel in 1931 states that aromaticity is observed in cyclically conjugated systems of 4n + 2 electrons, that is with 2, 6, 10, 14, etc., π-electrons; by far the majority of monocyclic aromatic and heteroaromatic systems are those with six π-electrons.
In this book we use the pictorial valence-bond resonance description of structure and reactivity. Even though this treatment is not rigorous, it is still the standard means for the understanding and learning of organic chemistry, which can at a more advanced level give way to the more complex, and mathematical, quantum-mechanical approach. We begin by recalling the structure of benzene in these terms.
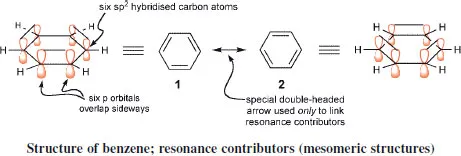
In benzene, the geometry of the ring, with angles of 120°, precisely fits the geometry of a planar trigonally hybridised carbon atom, and allows the assembly of a σ-skeleton of six sp2 hybridised carbon atoms in a strainless planar ring. Each carbon then has one extra electron which occupies an atomic p orbital orthogonal to the plane of the ring. The p orbitals interact to generate π-molecular orbitals associated with the aromatic system.
Benzene is described as a ‘resonance hybrid’ of the two extreme forms which correspond, in terms of orbital interactions, to the two possible spin-coupled pairings of adjacent p electrons: structures 1 and 2. These are known as ‘resonance contributors’, or ‘mesomeric structures’, have no existence in their own right, but serve to illustrate two extremes which contribute to the ‘real, structure of benzene. Note the standard use of a double-headed arrow to inter-relate resonance contributors. Such arrows must never be confused with the use of opposing straight ‘fish-hook’ arrows that are used to designate an equilibrium between two species. Resonance contributors have no separate existence; they are not in equilibrium one with the other.
Sometimes, benzenoid compounds (and also, occasionally six-and five-membered heterocyclic systems) are represented using a circle inside a hexagon (pentagon); although this emphasises their delocalised nature and the close similarity of the ring bond lengths (all exactly identical only in benzene itself), it is not helpful in interpreting reactions, or in writing ‘mechanisms’, and we do not use this method in this book.
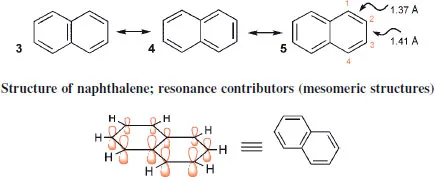
Treating naphthalene comparably reveals three resonance contributors, 3, 4 and 5. The valence-bond treatment predicts quite well the non-equivalence of the bond lengths in naphthalene: in two of the three contributing structures, C-1-C-2 is double and in one it is single, whereas C-2-C-3 is single in two and double in one. Statistically, then, the former may be looked on as 0.67 of a double bond and the latter as 0. 33 of a double bond: the measured bond lengths confirm that there indeed is this degree of bond fixation, with values closely consistent with statistical prediction.
2.1.2 Aromatic Resonance Energy3
The difference between the ground-state energy of benzene and that of hypothetical, non-aromatic, 1,3,5-cyclohexatriene corresponds to the degree of stabilisation conferred to benzene by the special cyclical interaction of the six π-electrons. This difference is known as aromatic resonance energy. Quantification depends on the assumptions made in estimating the energy of the ‘non-aromatic’ structure, and for this reason and others, a variety of values have been calculated for the various heteroaromatic systems; their absolute values are less important than their relative values. What one can say with certainty is that the resonance energy of bicyclic aromatic compounds, like naphthalene, is considerably less than twice that of the corresponding monocyclic system, implying a smaller loss of stabilisation energy on conversion to a reaction intermediate which still retains a complete benzene ring, for example during electrophilic substitution (see 3.2). The resonance energy of pyridine is of th...