![]()
Part 1 Basics
![]()
Basic science
The human nervous system is one of the most intricate of all bodily organs and new insights into its structure and function over recent decades have gone only part of the way towards unravelling the mysteries of this complex computer. The system is easiest to understand when broken down into manageable parts. Both the central and peripheral nervous systems are composed of nerve cells or neurons, which are organised in networks and serve various functions.
The processes underpinning these functions are complex, and can go wrong in lots of different ways. Much of the functioning, and malfunctioning, of the brain remains closed to us, and research continues apace. However, we are now gaining insights across a range of neurological diseases of the fundamental mechanisms that cause those diseases, and these are reviewed here. Of course, as information marches on some of what follows may prove to be at best an approximation of biological truth; but it is a better, more complete approximation than has been possible at any time in the past.
Neurons and synapses
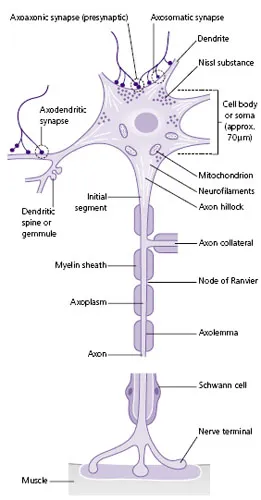
Neurons are shaped like a tree, with a cell body (the central part of the tree), an axon (the trunk) which conveys information from the cell body to the next nerve or muscle cell in the network, and dendrites (the roots) which receive inputs from other cells (Fig. A). The connection between two nerve cells is called a synapse, and information is transmitted between cells across synapses by chemicals called neurotransmitters. Neurons in the central nervous system are very small, but in the peripheral nervous system can transmit information across long distances; a lumbar anterior horn motor neuron axon (travelling from the lumbar region of the spinal cord to the feet) can be as long as 1 m, but only 10 µm wide. This is the equivalent of a drinking straw (5 mm wide) that is 500 m long! A sensory nerve from the big toe ending in the post-central sensory strip of the brain could be equivalent to a drinking straw of 1 km in length.
Like cardiac myocytes, neurons are excitable cells, meaning that they rely on electrical impulses to transmit information. The electric charge of a neuron is created and maintained by a delicate balance between positively and negatively charged ions, which enter and leave the cell through channels (such as sodium and potassium channels), the opening of which is usually regulated by pumps (such as sodium–potassium ATPase) in the cell membrane. When a neuron fires, rapid changes in ions within the cell (and their associated electric charges) cause depolarisation and repolarisation of the cell.
The action potential
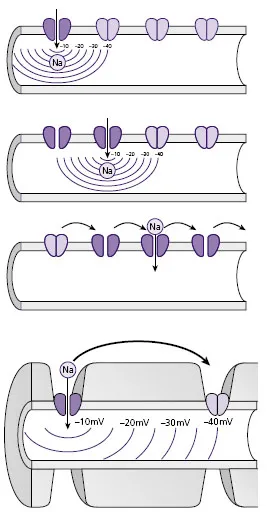
The various inputs to any given neuron lead to changes in the transmembrane potential in the region of the axon hillock. When the depolarisation raises the potential from the resting value (around –90 mV) to around –40 mV, specialised voltage gated sodium channels open, allowing the influx of sodium ions down their concentration gradient, with depolarisation and reversal of transmembrane potential to around +40 mV. This leads to changes in the transmembrane potential further down the neuron, and when sodium channels there sense that this has reached –40 mV they too open, and so the depolarisation is propagated down the neuron into the axon itself. In time the sodium channels close, and the resting balance of ions across the membrane is restored by sodium–potassium ATPase, a pump that consumes around two-thirds of neuronal energy expenditure.
Neuronal axons are covered in an insulating substance called myelin, which is a component of Schwann cells (in the peripheral nervous system) and oligodendrocytes (in the central nervous system). The effect of the myelin sheath is to provide insulation and allow faster conduction along the axon, as well as providing metabolic
support for the cell. Along the course of the axon there are tiny gaps between myelinating cells called nodes of Ranvier (which are up to 2 mm apart). The action potential jumps between these nodes by a process called saltatory conduction, which speeds up the process of conduction (Fig. B).
Disorders affecting the myelin sheath are usually immune-mediated and include multiple sclerosis and Guillain–Barré syndrome. When the myelin sheath is damaged, conduction velocity along the axon is reduced and the cell may die, producing neuronal atrophy. Multiple sclerosis (MS) is a disorder of cell-mediated immunity caused by recurrent attacks on oligoden-drocytes, and results in sustained impairment because of incomplete remyelination and secondary axonal damage. Neuromyelitis optica (NMO) is a related condition with recurrent episodes of optic neuritis and spinal cord inflammation. NMO is a disorder of humoral immunity caused by antibodies to the astocyte water channel aquaporin, and the prognosis is worse than that of MS.
Guillain–Barré syndrome is a disorder of humoral immunity resulting from a (usually monophasic) attack on Schwann cells causing demyelination. Antibodies raised in response to infection such as Campylobacter cross react with gangliosides expressed on the surface of Schwann cells and invoke an inflammatory response. The principal effect on nerve conduction is reduced conduction velocity rather than the reduced compound action potential seen in axonal neuropathies. This reflects the fact that while the axons are usually stripped of their myelin sheath, the underlying axons seldom die off, so that conduction is slower but the number of cells contributing to the compound action potential remains relatively constant.
Synaptic transmission
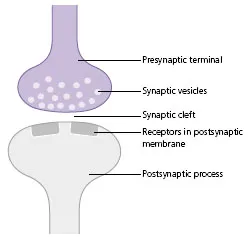
Neurons generally use chemical signalling to communicate with each other and with muscles at specialised structures called synapses (or, in the case of muscles, the neuromuscular junction). When a neuron depolarises, the wave of depolarisation spreads along the axon and reaches the presynaptic terminal at the end of the axon. The resultant change in membrane potential is sensed by voltage gated calcium channels which then open, leading to the influx of calcium. The resulting increase in intracellular calcium triggers the binding and fusion of presynaptic vesicles, which contain the neurotransmitters to the presynaptic membrane, leading to the release of transmitter to the synaptic cleft. The neurotransmitters diffuse across the synaptic cleft to reach the postsynaptic membrane on the next neuron in the network (Fig. C).
There follows a rapid increase in transmitter concentration at the postsynaptic membrane. Some of this transmitter binds to receptors on the postsynaptic membrane, some undergoes re-uptake by the presynaptic nerve terminal and some is metabolised by enzymes (such as acetylcholinesterase or catechol-O-methyl transferase).
Postsynaptic receptor binding
When it reaches the postsynaptic membrane, the neurotransmitter binds to receptors which are usually proteins. There are two main types of receptor: ion
channelassociated receptors and G-protein-coupled receptors. The latter are associated with G-proteins that result in the activation or inhibition of GTPases. The former are associated with ion channels (ligand gated) so that binding of the neurotransmitter to the receptor causes opening of the channel allowing influx or efflux of ions along their concentration gradient. Opening of a single ion channel could never cause sufficiention flows to raise the intracellular potential enough to result in neuronal depolarisation, but if the summation of inputs from different ion channels at different postsynaptic membranes is sufficient, depolarisation will occur, with opening of voltage gated channels and thereby initiation of an action potential.
These same receptors are the site of action of many of the drugs used in the treatment both of neurological diseases and of other conditions. For instance, many of the clinical manifestations of Parkinson’s disease are caused by degeneration of neuronal pathways between the substantia nigra and the striatum (so-called nigrostratal pathways). The neurons that are involved in abnormal degeneration in Parkinson’s disease have their cell bodies in the substantia nigra (‘nigra’, o...