![]()
1
A Brief History of Translational Neuroscience
Edgar Garcia-Rill
SOME RECENT HISTORY
According to an Institute of Medicine (IoM) report released on July 17, 2003, translational research and interdisciplinary approaches to care must be more strongly supported by both academic health centers and federal funding agencies [1]. “Academic Health Centers: Leading Change in the 21st Century” strongly advocated increased attention to translational research. The report pointed out that, although “the various forms of research are interrelated, they are typically conducted by different scientists and funded separately.” This approach will have to change, stated the IoM Committee on the Roles of Academic Health Centers in the twenty-first century. “Increased coordination and collaboration will be required to meet growing demands for rapid improvements in health care and for a greater focus on the types of research that answer questions about what does and does not work.” Interestingly, the impression among congressional leaders has been that the justification for doubling the National Institutes of Health (NIH) budget was tied to increased support for translational and clinical research. Related to the need for translational research is a disturbing national trend showing that MDs holding R01 awards decreased from 20% in 1982 to only 4% in 2002. R01 awards are individual research grants to support a discrete projects and is the most common grant mechanism at the NIH. We researchers, both basic and clinical, stand to lose legislative and public support for research if the current trend continues.
Congressional leaders, policy-makers, and the public at large are increasingly concerned that the scientific discoveries of the past are failing to be translated into tangible benefits to public health. The response has been a series of initiatives making translational research a priority. However, two blocks to translational research have been identified, a lack of translation of basic science discoveries into clinical studies (T1) and from clinical studies into medical practice (T2) [2,3]. The definitions of T1 and T2 research are actually that (a) T1 research addresses the translation of basic science breakthroughs into clinical trials, mainly on human subjects, while (b) T2 research attempts to implement those clinical trial findings into everyday clinical practice, thereby optimizing current treatments, for example, deciding between two equivalent therapies that may differ in cost-effectiveness, or developing novel therapies based on the results of well-drafted clinical trials. In fact, there has been a call to emphasize T1 and T2 research in proportion to its ability to improve health [4]. Additional blocks have been identified, blocks to T3 research foil attempts to move evidence-based guidelines into health practice, through delivery, dissemination, and diffusion of research, and blocks to T4 research impair the evaluation of the “real-world” health outcomes of a T1 application in practice. The latter require improved outreach programs, with considerable activity using telemedicine and other community-based research approaches.
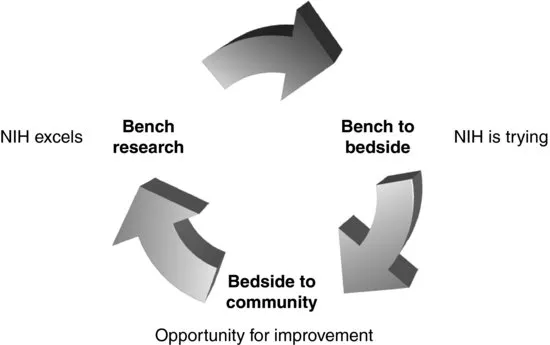
Typical T1 blocks to translational research include lack of willing participants, regulatory burdens, fragmented infrastructure, incompatible databases, and lack of qualified investigators [3]. Among the T2 blocks to translational research are career disincentives, practice limitations, high research costs, and lack of funding [3]. These issues will be addressed throughout this book, but, before going further, a common misconception is that translational research must proceed on a linear basis. There is considerable precedent to suggest that the linear approach to translational research, that is, proceeding from basic research on animals to clinical studies on humans, followed by clinical trials, and then applied studies, is not necessarily optimal. The lack of translation from animal research to clinical trials, the so-called T1 obstacle, suggests that a bottleneck exists at the transition between the huge amount of knowledge from basic studies to the trickle of clinically oriented research at present. However, this linear concept has been questioned, and one of the leaders in suggesting that we should consider this process as cyclical is Bill Crowley at Massachusetts General Hospital in Boston, MA. He has developed convincing examples of bedside to bench research, in which it is the genetic testing of individuals with genetic disorders that can drive the design and development of animal models on which can be tested novel therapeutic avenues, which can then be carried back to the bedside [5]. A better model for the progression from basic to clinical research and back is thus a cyclical model in which research can begin at various points in the cycle (Figure 1.1). Given the fact that performing translational research is indeed open-ended, the NIH has been careful to leave definitions open to interpretation. This is a wise position, allowing the field to employ brainpower and imagination to forge the future of translational research. The lack of pigeon holing of the meaning of translational research should be viewed as an opportunity rather than a limitation.
In September of 2003, Elias A. Zerhouni, MD, the then new Director of NIH, presented his “roadmap” for medical research. “The purpose is to identify major opportunities and gaps in biomedical research that no single institute at NIH could tackle alone but that the agency as a whole must address to make the biggest impact on the progress of medical research.” In reengineering the clinical research enterprise, “the exciting basic science discoveries currently being made demand that clinical research continue and even expand.” “Translational research has proven to be a powerful process that primes the entire clinical research engine. Key to building a strong infrastructure will be to increase interactions between basic and clinical scientists, and ease the movement of powerful new tools from the laboratory into the clinic.”
FUNDING TRANSLATIONAL RESEARCH
Academic health centers have been very good at making enormous strides in basic scientific research. In the coming years, this is likely to continue, but they will also need to begin refining the evidence base for health care. The general framework is that of discovery, which relies on basic research, followed by testing and application, which rely on clinical research, and then evaluation, which relies on applied research. Results from applied research are presumed to feed information to the formulation of further discovery. In reality, the process should begin at any point in the cycle. Academic health centers will begin to explore this cyclic continuum, with those that redesign and plan properly being more successful at garnering NIH, and public, support. A number of obstacles exist to the transition toward this continuum of research activities. First, there is a low supply of clinical researchers; second, there is a lack of institutional organization to support translational research; and third, there are inadequate funding levels to support such research. The first obstacle will be addressed in the next chapter on mentoring of clinician scientists and how to set up a career development program. The second obstacle will be addressed in the last chapter on how academic health centers can reshape themselves to not only meet the challenges of translational research, but also take advantage of the wide-open field of possibilities available for performing translational research.
LACK OF FUNDING
The third obstacle is being met on one front with the development of the Clinical Translational Science Award (CTSA) program under the National Center for Research Resources (NCRR). Even before the General Clinical Research Center (GCRC) program at NCRR was revamped into the CTSA, NIH-wide initiatives were implemented. You may recall that the GCRC program was intended as an institutional facility for inpatient and outpatient research. That model was critically flawed in terms of being unable to facilitate research for young investigators and failed to provide sufficient training to increase the pipeline of clinical scientists. These deficiencies have been addressed in the design of the CTSA program. But, even before these changes, there were concerns about the low funding levels of clinically oriented research. For example, in the review of NIH applications, informal surveys at NIH determined that those applications that used animals tended to score on average 10 percentile points better than those that used humans. That is, simply the fact that the “human subject” instead of the “animal research” box on the face of the application was checked meant that, on average, these applications were scored at a lower level of enthusiasm. Of course, research on human subjects is in many ways more difficult to control, and more fraught with variability and technological difficulties, so that it is not hard to understand this attitude. In response, the review criteria of standard research grant applications were changed at all levels and institutes at NIH. The following are now typical review criteria, with the phrases in bold being the new ones added to accommodate the new emphasis on clinically oriented research. “Significance: Does this study address an important problem? If the aims of the application are achieved, how will scientific knowledge or clinical practice be advanced? What will be the effect of these studies on the concepts, methods, technologies, treatments, services, or preventative interventions that drive this field? Approach: Are the conceptual or clinical framework, design, methods, and analyses adequately developed, well integrated, well reasoned, and appropriate to the aims of the project? Does the applicant acknowledge potential problem areas and consider alternative tactics? Innovation: Is the project original and innovative? For example: Does the project challenge existing paradigms or clinical practice; address an innovative hypothesis or critical barrier to progress in the field? Does the project develop or employ novel concepts, approaches, methodologies, tools, or technologies for this area? Investigators: Are the investigators appropriately trained and well suited to carry out this work? Is the work proposed appropriate to the experience level of the principal investigator and other researchers? Does the investigative team bring complementary and integrated expertise to the project (if applicable)? Environment: Does the scientific environment in which the work will be done contribute to the probability of success? Do the proposed studies benefit from unique features of the scientific environment, or subject populations, or employ useful collaborative arrangements? Is there evidence of institutional support?”
Launched in 2006 and led by the NCRR, the CTSA program is working at institutional, regional, and national levels to create a discipline of clinical and translational science. Its primary mission is to more efficiently translate the rapidly evolving knowledge developed in basic biomedical research into treatments to improve human health. From 2006 to 2008, 38 academic health centers and research institutions in 23 states became part of the consortium. In 2010, the consortium consisted of 55 member institutions. When fully implemented, approximately 60 institutions will be linked in a way that is intended to energize the discipline of clinical and translational science with >$500 million per year of NIH funding. Diversity in the size, scope, and geographic location of participating institutions has been mandated because such diversity is thought to strengthen the CTSA consortium and enhance its impact.
More recently, the NIH Scientific Management Review Board voted on December 7, 2010, to approve a recommendation to newly appointed NIH Director Francis Collins to create a new NIH center focused on translational medicine and therapeutics called the National Center for Advancing Translational Science (NCATS). The proposed center would house the currently NCRR-administered CTSA program along with the Cures Acceleration Network, Molecular Libraries Program, Therapeutics for Rare and Neglected Diseases, and Rapid Access to Interventional Development, as well as new NIH-FDA partnership activities.
These changes have generated considerable concern in the research community, and it is not yet clear whether congressional support will follow. For example, one of the mandates of NCATS will be the development of new drugs for therapeutic use. This is a response to the disturbing reduction in the pharmaceutical industry of spending on research and development, all in the face of a decline in the output of new drugs approved by the Food and Drug Administration (FDA). While Dr. Collins has been predicting that gene sequencing will lead to a host of new treatments, investments in the billions of dollars by the drug industry have failed to yield new gene-related therapies. While the NIH has historically been very good at supporting basic research, many wonder how good it will be at drug development, which requires a different set of skills. On the plus side, it may turn out that such an effort may generate a new type of researcher who can perform in both academic and drug company settings. On the minus side, this is the first time in the 80-year history of the NIH that an institute will be dismantled and the parts scattered across the rest of NIH. Historically, orphan programs tend to be phased out of existence by the “host” institute. These concerns are likely to persist for years, but it is hoped that unbiased and independent assessment of the success of the NCATS will tell us if the investment is worthwhile.
There are additional concerns. For example, the creation of the several components of the NCATS will require most, if not all, of the funding accorded the NCRR in order to support these new directions. This means that cuts to other programs could ensue. Among the most controversial consequences of eliminating the NCRR is the future of such programs in the NCRR portfolio as the Research Centers for Minority Institutions that supports centers as the name implies, and the Institutional Development Award (IDeA) program that supports such statewide infrastructure development incentives as the IDeA Networks of Biomedical Research Excellence (INBRE) program, and the Centers of Biomedical Research Excellence (COBRE) program that underwrite the creation of thematic, multidisciplinary centers, all in states with historically low levels of Federal funding. These programs are intended to provide diversity and correct the geographical inequalities in research support. These fairly small programs produce a huge return on investment, and add to the economic impact of academic health centers in small and medium-sized communities, which is in the order of >$3 billion for an average medical school [1]. The ramifications of this reorganization are likely to have considerable and lasting impact.
MAKING NIH FUNDING MORE EQUITABLE AND EFFICIENT
Most of the research in academic health centers in the United States is done under the auspices of the NIH. The NIH budget is currently around $31 billion, which is about 0.27% of the Gross Domestic Product (GDP), and about one half of what most developed countries spend on research. It can be argued that we do not spend enough on research. On the other hand, the United States spends twice as much for health care per capita as other developed countries, yet lags behind other wealthy nations in such measures as infant mortality and life expectancy. This can be interpreted to mean that the way we distribute our research dollar does not have sufficient impact on health care. Moreover, as mentioned in the preceding text, there is a regional inequality in the funding of biomedical research, being concentrated on the two coasts. The top 10 institutions are awarded about a third of all NIH extramural funding, while the next 40 institutions receive over one half of all the grant money. Institutions in states that account for over 20% of the population receive less than 10% of all awards. The NIH has instituted a number of measures to improve medical research. For example, during the Clinton administration, the NIH budget was doubled over a 10-year period. This had the effect of funding much new research and attracting three times the number of scientists into research, but it also made grants not twice, but three times more competitive. In the meantime, the disparity between the “haves” and the “have-nots” grew more severe. Such programs as INBRE and COBRE are essential to developing diversity in facilities throughout the country, increasing areas of research excellence, and serving the needs of all taxpayers. This is especially true when the issue is one of improving health for the public at large.
One massive cost that has not been addressed but could save millions of dollars is the establishment of a national indirect cost rate. Indirect costs are subsidies to the institution holding an NIH award for expenses incurred by the facilities related to the performance of the award such as heat and air, cleaning, purchasing, human resources, accounting, regulatory oversight, and so on. That is, an institution with a 50% indirect cost rate that is granted a $1 million award will actually receive $1.5 million, $1 million in direct costs and $0.5 million in indirect costs. Unfortunately, many institutions have negotiated rates as high as 100% or more. The same $1 million award at one of those institutions would cost NIH $2 million or more for performing the same research projec...