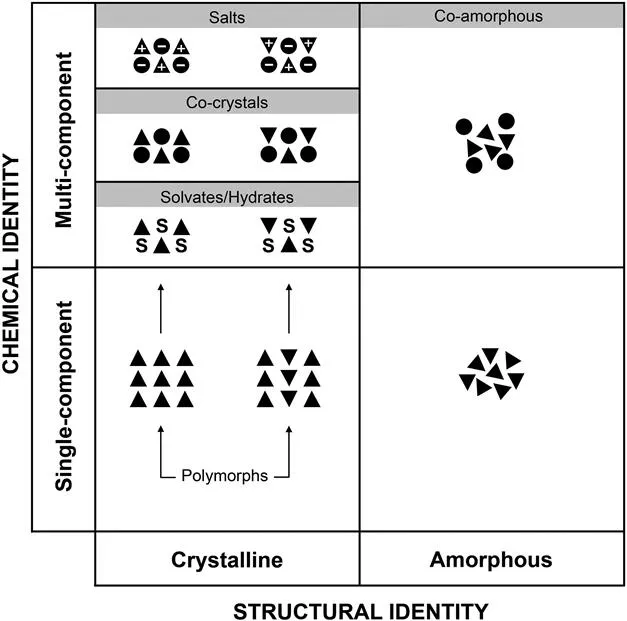
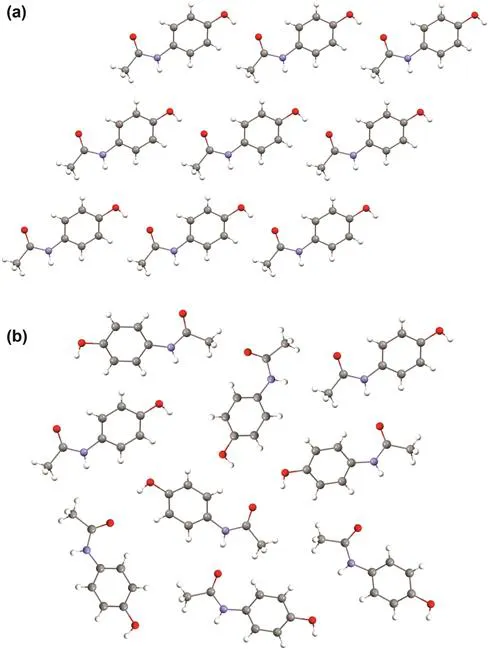
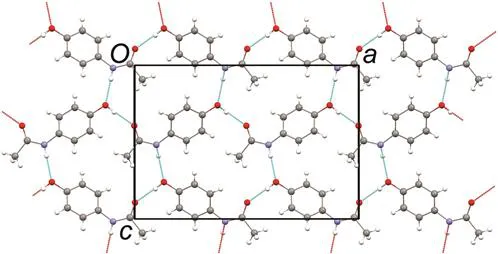
The pharmaceutical industry has become acutely aware of the importance of the solid state, but pharmaceutical scientists often lack specific training in topics related to solid-state structure and crystallography. This book provides needed support in this topical area.
Taking an intuitive and informal approach to solid-state structure and crystallographic concepts, this book is written for anyone who needs a clear understanding of modern crystallography, with specific reference to small-molecule pharmaceutical solids. The author describes molecular crystals and crystal structures, symmetry, space groups, single-crystal and powder X-ray diffraction techniques and the analysis and interpretation of crystallographic data. Useful technical details are presented where necessary and case studies from the pharmaceutical literature put theory into a practical context.
Written by an internationally leading figure and with its focus on molecular crystals, this book is equally applicable to chemists with a need to understand and apply X-ray crystal-structure determination.