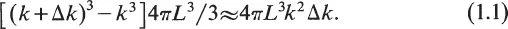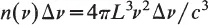![]()
1
The History of Quantum Mechanics
This chapter has a twofold purpose. It gives the reader a historical perspective, and introduces some of the important concepts of quantum mechanics. As the reader will notice, probability theory did not enter into quantum mechanics at the outset. The pioneers of the field had no reason or intention to make quantum mechanics a probabilistic subject. The stochastic nature of quantum mechanics was reluctantly accepted later when it proved to be an intrinsic, inescapable part of the field.
1.1. Early History
The idea of a quantum of energy was first introduced by Max Planck on December 14, 1900 at a meeting of the German Physical Society. He invented this concept to explain an empirical formula for the energy per unit volume E(v) of radiation of frequency v emitted by a body heated to a temperature T.
In the late 1800s, Rayleigh and Jeans derived a formula for E(v) using classical mechanics. Suppose heat radiation is emitted through a small door of a cubical furnace of length L which is heated to a temperature T. The energy of the radiation e(v) at frequency v is found by determining the number of waves n(v) with frequency v and multiplying n(v) by the energy associated with these waves. According to a time-honored principle of physics, the equipartition principle, each wave should carry KT units of energy on the average, where K is Boltzmann’s constant. Hence e(v) = n(v)KT. To find n(v), the waves are represented mathematically by plane waves exp[2πi(k·r – vt)] where k is a vector of length k = v/c (c is the speed of light) in the direction of wave propagation with components (n1/L, n2/L, n3/L), nl, n2, n3 being integers. The integers appear since we must have a whole number of wavelengths in the furnace cavity. We now find the number of waves n(v) Δv with frequency between v and v + Δv. Draw a three-dimensional lattice with dots at each point with integer coordinates and draw a spherical shell whose inner and outer radii are k and k + Δk, respectively. Then the number of waves whose wavenumber is between k and k + Δk is the number of dots in this shell. This number is approximately the volume of the shell divided by the volume surrounding an individual dot and thus equals
Hence
and
Formula (1.2), called the Rayleigh–Jeans law, agrees with experiment for low frequencies v. However, for high frequencies, (1.2) not only deviates substantially from experiment, it gives a contradiction called the ultraviolet catastrophy. According to Eq. (1.2), as the frequency increases so does the corresponding energy. The ultraviolet catastrophy concludes that the total energy per unit volume due to all frequencies is infinite.
The ultraviolet catastrophy was one of the first instances in which classical mechanics was found to be inadequate. Planck’s energy quantum, invented to derive the correct formula for E(v) and thus avoid this catastrophy, ushered in a new era, the era of quantum mechanics. Planck’s idea was so unusual and seemed so grotesque at the time, that he himself could hardly believe it. Planck suggested that instead of allowing the energy of radiation waves to have arbitrary values, we make the following postulate.
The energy of electromagnetic waves can exist only in the form of discrete packages, or quanta, the energy content of eac...