Based on a California Institute of Technology course, this outstanding introduction to formal quantum mechanics is geared toward advanced undergraduates in applied physics. The text addresses not only the basic formalism and related phenomena but also takes students a step further to a consideration of generic and important applications. The treatment's exploration of a wide range of topics culminates in two eminently practical subjects, the semiconductor transistor and the laser. Subjects include operators, Eigenvalue problems, the harmonic oscillator, angular momentum, matrix formulation of quantum mechanics, perturbation theory, the interaction of electromagnetic radiation with atomic systems, and absorption and dispersion of radiation in atomic media.
Additional topics include laser oscillation, quantum statistics, applications of the statistical distribution laws, the interaction of electrons and nuclei with magnetic fields, and charge transport in semiconductors. Each chapter concludes with a set of problems.

- 320 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
An Introduction to Theory and Applications of Quantum Mechanics
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
Physical SciencesSubtopic
Quantum TheoryCHAPTER ONE
Why Quantum
Mechanics?
Mechanics?
In the late 1800s and early 1900s it was becoming clear that the science of physics was due for a major revision. An increasing number of phenomena and observations failed to be adequately, or even approximately, described by the laws of physics as they were then formulated. Problems arose especially in attempts to provide an explanation for phenomena involving “small” particles such as electrons and atoms and their interaction with electromagnetic fields.
At first these deficiencies in physics were patched by ad hoc hypotheses and postulates. However, as their number grew it became clear that physics needed a complete reformulation, especially the physics of small systems. The result was quantum mechanics—one of the towering intellectual achievements of humankind.
In this first chapter we will place the formal development of quantum mechanics in context by outlining some of the basic results of classical physics. We will then recount some of the phenomena that, historically, defied explanation by classical physics.
1.1 NEWTONIAN MECHANICS AND CLASSICAL ELECTROMAGNETISM
Newtonian Mechanics
In classical nonrelativistic physics particles are assumed to move under the influence of forces. The law describing the motion is

where m is the mass of the particle, F the force, and v the velocity. This law, together with the law of gravitational attraction, for example, proved adequate for describing the motion of heavenly bodies and for predicting accurately the orbits of artillery shells. One important aspect of Newtonian mechanics was its determinism. Once the position and velocity of a particle were specified at some instant of time, and if the forces acting on it were known, then its behavior at all other times was exactly determined.
Electromagnetism
The electric and magnetic fields, E(r, t) and B(r, t), are described in classical electromagnetism by Maxwell’s equations, which in free space and in MKS units take the form1


where c = velocity of light. Using the vector identity ∇ × (∇ × A)=– ∇2A + ∇∇·A plus the fact that in free space ∇ · E = 0, we obtain from (1.2a) and (1.2b)

and a similar equation for B. Equation (1.3) admits solutions of the form

provided

The field (1.4) describes a plane wave propagating with a velocity c along k. A stationary observer will observe the field to oscillate at a frequency v = ω/2π. (The unit frequency is 1 Hz = l cycle/sec.) The wavelength is given by

Classical physics thus provides two formalisms with which to describe natural phenomena. The first—mechanics—deals with particles; the second —electromagnetic theory—deals with radiation waves. The two classes of phenomena were assumed to be distinct but coupled through the Lorentz force equation
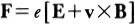
which gives the force F exerted on a particle of charge e moving with velocity v in fields E,B.
1.2 BLACK BODY RADIATION
One of the major unsolved problems occupying physicists around the late 1800s and the early 1900s was that of black body radiation. An idealized “black body” is a material that absorbs perfectly at all wavelengths. Many common materials—lampblack, for example—are excellent absorbers over large spectral regions. General thermodynamic arguments have shown that the spectral intensity (watts/m2 per unit frequency interval) I(v) of emitted radiation should be the same for all black bodies at a given temperature. This indeed was found to hold true, and experimental measurements of I(v) yielded the curves shown in Fig. 1.1. The intensity reaches a maximum at some frequency vm while dropping to zero on either side of vm. The frequency vm, as well as the height of the peak, increase with temperature.
Theoretical attempts to predict the behavior of the black body spectral intensity from the then known first principles were unsuccessful until 1900. The application of statistical thermodynamics and the ordinary laws of mechanics and electromagnetic theory led to the so-called Rayleigh-Jeans formula

where k = 1.3807 × 10–23 joule/k is the Boltzmann constant. This law is plotted in Fig. 1.1 and, except for very low frequencies, is in total disagreement with experimental results. The Rayleigh–Jeans law predict...
Table of contents
- Cover
- Title Page
- Copyright Page
- Preface
- Contents
- Chapter 1 - Why Quantum Mechanics?
- Chapter 2 - Operators
- Chapter 3 - The Basic Postulates of Quantum Mechanics
- Chapter 4 - One-Dimensional Energy Eigenvalue Problems
- Chapter 5 - The Harmonic Oscillator
- Chapter 6 - The Quantum Mechanics of Angular Momentum
- Chapter 7 - Particles in Spherical Symmetric Potential Fields and the Hydrogen Atom
- Chapter 8 - Systems of Identical Particles
- Chapter 9 - Matrix Formulation of Quantum Mechanics
- Chapter 10 - The Time-Dependent Schrödinger Equation
- Chapter 11 - Perturbation Theory
- Chapter 12 - The Interaction of Electromagnetic Radiation with Atomic Systems
- Chapter 13 - Absorption and Dispersion of Radiation in Atomic Media
- Chapter 14 - Laser Oscillation
- Chapter 15 - Quantum Statistics
- Chapter 16 - Some Specific Applications of the Statistical Distribution Laws
- Chapter 17 - The Band Theory of Electrons in Crystals
- Chapter 18 - The Interaction of Electrons and Nuclei with Magnetic Fields. Magnetic Resonance. The Maser
- Chapter 19 - Charge Transport in Semiconductors
- Chapter 20 - The p-n Semiconductor Junction. The p-n-p Junction Transistor.
- Chapter 21 - The Semiconductor Injection Laser
- Bibliography
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access An Introduction to Theory and Applications of Quantum Mechanics by Amnon Yariv in PDF and/or ePUB format, as well as other popular books in Physical Sciences & Quantum Theory. We have over 1.5 million books available in our catalogue for you to explore.