![]()
1
Boron Hydrides and Related Structures
The boron hydrides, first described and characterized by Stock,299 form an unusual set of compounds. A systematic description of their chemistry is now possible, owing to the elucidation of their molecular structures, a valence-theory description, and many new studies of their chemical reactions. Just on the horizon is a detailed mechanistic description of their chemical reactions, and a predictive chemistry approaching that of carbon chemistry. In this monograph an attempt is made to systematize the molecular structure properties, the valence structures in terms of three-center bonds and molecular orbitals, the nuclear magnetic resonance (NMR) spectra, and, finally, the known chemical reactions.
1–1 The Hydrides
Tentative evidence, mostly of a physical type, places the number of presently identifiable distinct hydrides somewhere near thirty, but only ten have been isolated and well characterized. These known hydrides are B2H6, B4H10, B5H9, B5H11, B6H10, B9H15, B10H14, B10H16, B18H22, and iso-B18H22. The structural evidence for them is outlined below, and is followed by similar discussions concerning the boranates (boron hydride ions), the amine derivatives, the halides of boron, and the carboranes.
B2H6
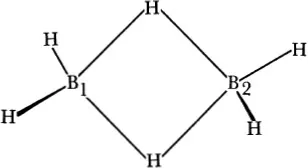
That the bridge type of structure is the correct one for diborane is generally recognized to have been established by Price242,243 on the basis of the infrared spectrum. Prior to this study, however, there did exist a general realization17,186,203,237,297,298 that the physical evidence was overwhelmingly in favor of this structure. The failure of general acceptance of, for example, the high barrier to internal rotation and the earlier infrared evidence297 as indicative of the bridge structure, must be attributed to the insistence8,10 that the early electron diffraction evidence favored the ethane type of structure. A later electron-diffraction study,105 however, confirmed the bridge structure and was in disagreement with the ethane type of structure. The structure is shown in Fig. 1–1.
The correct molecular geometry was first proposed by Dilthey.52 However, interpretations in terms of valence bonds or molecular orbitals were made only comparatively recently.203,237,317 Whether one thinks in terms of a “protonated double bond,” i.e., a four-atom bridge bond, in an ethylenic type of structure, or in terms of the interaction of two BH3 groups, is, in any complete analysis, merely a matter of taste, but analogy of bonding geometry and electronic structure with these properties of ethylene is very striking.
B4H10
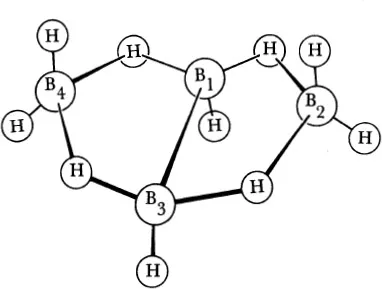
Both hydrogen and boron positions (Fig. 1–2) were established unambiguously by the X-ray diffraction study207,208 in which there were 616 observed reflections to determine 14 parameters counting the boron, scale, and temperature parameters. Hydrogen atoms, not included in the counting of parameters, appeared both in the presence of the boron atoms and when the boron atoms were subtracted out in the electron-density maps. This model was independently suggested in the recent electron-diffraction study139 on the basis of its plausible relation to the other boron hydrides and of its consistency with the electron-diffraction data, but these results are not unambiguous, for a slight modification of the model decided upon in the earlier diffraction study9 is equally consistent with the electron-diffraction data. Tests of other possible models were not made in this most recent electron-diffraction study, but the results of the earlier study were shown to be incorrect.
Figure 1–1 The structure of B2H6. The B1—B2 distance is 1.77 A, the B—H distance is 1.19 A, the B—Hµ (bridge) distance is 1.33 A, and the H—B—H angle is 121.5°.
Figure 1–2 The structure of B4H10. The B1—B3 distance is 1.71 A, and the other four close B · · · B distances are 1.84 A. The B—H distances average to 1.19 A, while the B—Hµ (bridge) distances are 1.33 A toward B1 and B3 and 1.43 A toward B2 and B4.
Comparison of the results of the X-ray and electron-diffraction study indicates the molecular parameters, B
1—B
2 = 1.84 A, B
1—B
3 = 1.71 A,
B
2B
1B
4 = 98°, B—H = 1.19 A, B
1—H
µ = 1.33 A, and B
2—H
µ = 1.43 A. This is a model obtained from the X-ray values of boron-boron distances and bond angles, including hydrogen angles, and from the electron-diffraction values of the B—H distances, but with the asymmetry of the hydrogen bridges reversed to agree with the X-ray results. This combination avoids the systematic errors introduced by the lack of complete convergence of the Fourier series in the hydrogen distances of the X-ray study and the very large uncertainties in the bond angles in the electron-diffraction study.
The boron arrangement can be considered as a fragment of either the icosahedron or the octahedron because the B2—B1—B4 bond angle is between 105° and 90°.
B5H9
The certainty with which this structure (Fig. 1–3) is established is based upon the crystal-structure study,58,59 in which, at the outset, molecules of C...