REA's Chemistry Super Review
Get all you need to know with Super Reviews!
2nd Edition
REA's Chemistry Super Review contains an in-depth review that explains everything high school and college students need to know about the subject. Written in an easy-to-read format, this study guide is an excellent refresher and helps students grasp the important elements quickly and effectively.Our Chemistry Super Review can be used as a companion to high school and college textbooks, or as a handy resource for anyone who wants to improve their chemistry skills and needs a fast review of the subject.Presented in a straightforward style, our review covers the material taught in a beginning-level chemistry course, including: atomic structure, bonding, chemical reactions, liquids, solids, gases, properties of solutions, chemical thermodynamics, and more.
The book contains questions and answers to help reinforce what students learned from the review. Quizzes on each topic help students increase their knowledge and understanding and target areas where they need extra review and practice.

- 240 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Chemistry Super Review - 2nd Ed.
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Publisher
Research & Education AssociationYear
2012Print ISBN
9780738611167
Edition
2eBook ISBN
9780738683867
CHAPTER 1
Introduction
1.1 Matter and Its Properties
1.1.1 Definition of Matter
Matter occupies space and possesses mass. Mass is an intrinsic property of matter.
Weight is the force, due to gravity, with which an object is attracted to the earth.
Force and mass are related to each other by Newton’s equation (Newton’s Law), F = ma, where F = force, m = mass, and a = acceleration. Weight and mass are related by the equation w = mg, where w = weight, m = mass, and g = acceleration due to gravity.
Note that the terms “mass” and “weight” are often (incorrectly) used interchangeably in most literature.
1.1.2 States of Matter
Matter occurs in three states or phases: solid, liquid, and gas. A solid has both a definite size and shape. A liquid has a definite volume but takes the shape of the container, and a gas has neither definite shape nor definite volume.
1.1.3 Composition of Matter
Matter is divided into two categories: distinct substances and mixtures. Distinct substances are either elements or compounds. An element is made up of only one kind of atom. A compound is composed of two or more kinds of atoms joined together in a definite composition.
Mixtures contain two or more distinct substances more or less intimately jumbled together. A mixture has no unique set of properties: it possesses the properties of the substances of which it is composed.
In a homogeneous mixture, the composition and physical properties are uniform throughout. Only a single phase is present. A homogeneous mixture can be gaseous, liquid, or solid. A heterogeneous mixture, such as oil and water, is not uniform and consists of two or more phases.
1.1.4 Properties of Matter
Extensive properties, such as mass and volume, depend on the size of the sample. Intensive properties, such as melting point, boiling point, and density, are independent of sample size.
Physical properties of matter are those properties that can be observed, usually with our senses. Examples of physical properties are physical state, color, and melting point.
Chemical properties of a substance are observed only in chemical reactions involving that substance.
Reactivity is a chemical property that refers to the tendency of a substance to undergo a particular chemical reaction.
Chemical changes are those that involve the breaking and/or forming of chemical bonds, as in a chemical reaction.
Physical changes do not result in the formation of new substances. Changes in state are physical changes.
1.2 Conservation of Matter
1.2.1 Law of Conservation of Matter
In a chemical change, matter is neither created nor destroyed, but only changed from one form to another. This law requires that “material balance” be maintained in chemical equations.
1.3 Laws of Definite and Multiple Proportions
1.3.1 Law of Definite Proportions
A pure compound is always composed of the same elements combined in a definite proportion by mass.
Problem Solving Example:

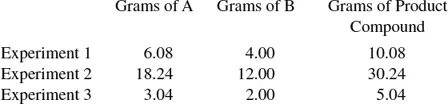
(a) Which two laws of chemical change are illustrated by the above data? (b) If 80 g of element B combines with 355 g of a third element C, what weight of A will combine with 71 g of element C? (c) If element B is oxygen, what is the equivalent weight of element C?

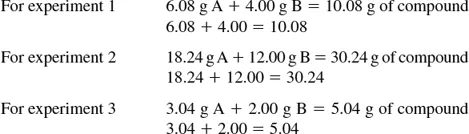
From these calculations one can see that the law of conservation of matter is shown.
Another important law of chemistry is the law of definite proportions. This law is stated: when elements combine to form a given compound, they do so in a fixed and invariable ratio by mass. One can check to see if this law is adhered to by calculating the ratio of the weight of A to the weight of B in the three experiments. If all of these ratios are equal, the law of definite proportions is shown.
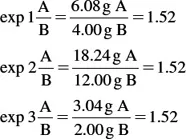
The law of definite proportions is illustrated here.
(b) From the law of definite proportions, one can find the number of grams of B that will combine with 71 g of ...
Table of contents
- Cover Page
- Title Page
- Copyright Page
- REA’s Chemistry Super Review®
- Available Super Review® Titles
- About Research & Education Association
- Contents
- Chapter 1 Introduction
- Chapter 2 Stoichiometry, Chemical Arithmetic
- Chapter 3 Atomic Structure and the Periodic Table
- Chapter 4 Bonding
- Chapter 5 Chemical Formulas
- Chapter 6 Types and Rates of Chemical Reactions
- Chapter 7 Gases
- Chapter 8 Liquids, Solids, and Phase Changes
- Chapter 9 Properties of Solutions
- Chapter 10 Acids and Bases
- Chapter 11 Acid-Base Equilibria in Aqueous Solutions
- Chapter 12 Chemical Equilibrium
- Chapter 13 Chemical Thermodynamics
- Chapter 14 Oxidation and Reduction
- Back Cover
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Chemistry Super Review - 2nd Ed. by Editors of REA in PDF and/or ePUB format, as well as other popular books in Physical Sciences & Chemistry. We have over 1.5 million books available in our catalogue for you to explore.