
- 288 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Reactive Separation Processes
About this book
This book summarizes the available information in six known areas of reactive separation: reaction/distillation, reaction/extraction, reaction/absorption, reaction/adsorption, reaction/membrane, and reaction/crystallization.
Trusted by 375,005 students
Access to over 1 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Chapter 1
REACTIVE SEPARATION PROCESSES
1.1 Introduction
Reactive separation processes are unique in that they couple chemical reactions and physical separations into a single unit operation. This fusion of reaction and separation operations into one combined operation is prized for the simplicity and novelty this approach brings to the process flowsheet. These reactive separations are also coveted for the investment and operating cost savings garnered on successful scale-up to commercial operations. Reactive separation processes as a whole are not a new concept. Numerous applications have been commercialized for traditional separation methods over the course of six decades. However, the academic and industrial communities have taken renewed interest in the development and commercialization of reactive separations in recent years in response to economic opportunities and pressures exerted, which have caused the emergence of new industries and decline of existing industries, the emergence of new separation and process technologies, the demand for higher building-block purities for food, pharmaceutical, polymer, and electronics products, changes in the availability and pricing of key resources, and the growing concern for protecting our environment. This interest is reflected in the increased volume of literature issued on the development and design of reactive separations and the increasing number of technical conferences devoted to the subject.
The concept of the reactive separation process is schematically presented in Figure 1.1. In traditional process design (the sequential reaction/separation process in Figure 1.1), a chemical reactor is typically sequenced with a downstream separator. In this way, feed is first converted into valued products which are then isolated and recovered in the separator. To optimize the product yield and purity, the operating conditions of the reactor and the separator are varied to achieve optimal economic performance subject to prevailing constraints. In many cases, recycle streams are incorporated into the process to reprocess unreacted feed or intermediate products back through the reactor and separator to increase overall process yields. In contrast, the design of a reactive separation process (the integrated reaction/separation process in Figure 1.1) focuses on integrating the reactor and separator operations into a single process operation with simultaneous reaction and separation. In combining sequential processing steps into an integrated processing approach, additional processing benefits may be achieved. Quite often, these benefits include the elimination of one or more recycle streams which are associated with optimizing performance of the original sequential process configuration. More excitingly, the integration may lead to the design of a separation process which cannot be achieved with separate reactor and separator process flow elements.
The applications of reactive separation process design are numerous and span a broad range of process operations. The development and application of reactive separations is not new. For example, reactive absorption has long been practiced for the removal of acid gases in the petroleum production and refining industries, and acid/base extractions and reactive distillations have long been practiced in the purification of chemicals. However, mention the phrase “reactive separations” and chemists and engineers will likely point to the more recent commercialization of reactive distillation technologies for the esterification of acetic acid to methyl acetate and the etherification of methanol with isobutylene to methyl tertiary butyl ether (MTBE) as examples of the art of reactive separation design.
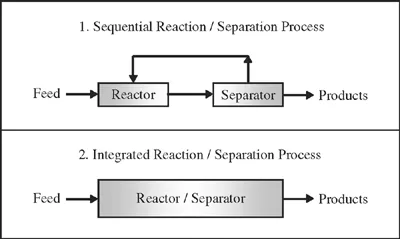
Figure 1.1: Principle of combined reaction and separation system.
These latter developments herald a renewal of interest in the development and commercialization of reactive separations. This trend is reflected in the rate at which the academic and industrial communities continue to expend effort to develop and apply reactive separations technology. A quick and rough measure of this trend is provided by scanning the number of citations issued each year on this subject in Chemical Abstracts using a keyword search (Table 1.1). For example, a quick citation search was conducted online for the subject of reactive distillation using keywords such as reactive distillation, distillation with reaction, simultaneous distillation with reaction, etc. This search yielded 263 publication citations and 123 patent citations from 1968 through the end of 1997. Breakdown of these listings by year in which the publications and patents were issued gives us a qualitative trend of the activity devoted to developments in this field as a function of time.
Figure 1.2 graphically presents the results of such a search for reactive separations in general for the 30-year time span from 1968 through 1997. The graph plots the cumulative number of citations at five-year intervals starting in 1972 for publications which specifically address reactive separations versus the year for which the citations were collected, on a semilog plot. The results are further segregated by type of process operations as follows: (1) reactive distillation, (2) extraction with reaction, (3) absorption with reaction, (4) adsorption with reaction, (5) reactive membrane separation, and (6) reactive crystallization. All six of these classes of reactive separations exhibit one to two orders of magnitude increase in the rate at which articles have been published in the literature over time.
This growth of interest in the subject of reactive separation processes is also reflected in the number of patents issued in each of the above areas. Using the same quick keyword search that was used to generate Figure 1.2, the growing interest can be roughly trended by examining the cumulative patent citation count for the 1968–1997 time frame. This data is plotted by class of separation in Figure 1.3 alongside the cumulative count for the number of article citations cited in Figure 1.2. The trends roughly match those observed for the open literature. The greatest number of patents was issued for reactive absorption technology development. The development of proprietary interests for the other five classes through the issue of patents lags the activity in the reactive absorption field by one to two orders of magnitude. Among these latter five classes, reactive membrane development followed by reactive distillation shows the greatest interest in terms of number of patents issued in these fields.
Table 1.1: Summary of Keywords Used to Conduct Chemical Abstracts Citation Search for Chapter 1 Figures.
| Subject | Search Keywords | Search Results |
| Reactive distillation | Catalytic distillation or distillation, catalytic | 263 Publications from 1967–1998 123 Patents from 1967–1998 |
| Catalysis with distillation or distillation with catalysis | ||
| Reactive distillation or distillation, reactive | ||
| Simultaneous (I)a reaction and (I) distillation | ||
| Extraction with reaction | Extraction with reaction or extraction with chemical reaction | 331 Publications from 1967–1998 32 Patents from 1967–1998 |
| Reactive extraction | ||
| Simultaneous (I) extraction and (I) reaction | ||
| Combined (I) extraction and (I) reaction | ||
| Extractive alkylation or extractive methylation | ||
| Extraction, with reaction Catalysis (I) with extraction Reaction, with extraction | ||
| Absorption with reaction | Simultaneous absorption and reaction | 24,718 Publications from 1968–1998 |
| Gas treating or treatment or process | 22,425 Patents from 1968–1998 | |
| Treating gas(es) or treatment gas(es) or processing gas(es) | ||
| Absorptive reactive or absorptive reactivity | ||
| Absorption reaction or absorption reactivity | ||
| Absorption catalysis or catalytic or catalyst | ||
| Absorptive catalysis or catalytic or catalyst | ||
| Removal acid gas(es) | ||
| Adsorption with reaction | Adsorption reaction with or adsorption with reaction | 688 Publications from 1968–1998 40 Patents from 1968–1998 |
| Reaction with adsorption or reaction, adsorption with | ||
| Adsorption reaction Reaction, catalytic, adsorption (L)b (simultaneous or combined) | ||
| Reactive adsorption Adsorption (L) reaction (L) simultaneous | ||
| Simultaneous adsorption reaction | ||
| Catalytic adsorption/obi (in selected sections of Chemical Abstracts) | ||
| Reactive membrane separation | Membrane reactor Reactive membrane Simultaneous reaction and membrane | 1874 Publications from 1968–1998 262 Patents from 1968–1998 |
| Membrane separation with reaction | ||
| Reactive crystallization | Crystallization with reaction or reaction with crystallization | 182 Publications from 1968–1998 22 Patents from 1968–1998 |
| Simultaneous crystallization (I) and reaction (I) | ||
| Precipitation (I) reactive (but in geological chemistry section of Chemical Abstracts) | ||
| Crystallization (I) reactive and crystallization is an index heading |
a I = search indexing term.
b L = search indexing terms linked.
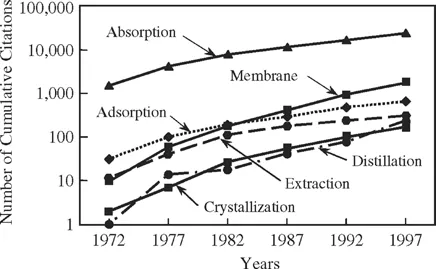
Figure 1.2: Number of Chemical Abstracts citations for publications on reactive separations from 1972 to 1997.
1.2 Advantages and Disadvantages
What is it that attracts the interest to develop these reactive separation processes? As difficult as it is to accurately trend the interest in the development of reactive separations, it is still more difficult to generalize the driving forces which lead to the development of these reactive separations. The simplest approach is to focus on the ben...
Table of contents
- Cover
- Half Title
- Title Page
- Copyright Page
- Table of Contents
- Preface
- Acknowledgments
- Contributors
- 1 Reactive Separation Processes
- 2 Reactive Distillation
- 3 Extraction with Reaction
- 4 Absorption with Reaction
- 5 Adsorption with Reaction
- 6 Reactive Membrane Separation
- 7 Reactive Crystallization
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.4M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Reactive Separation Processes by Kulprathipanja in PDF and/or ePUB format, as well as other popular books in Physical Sciences & Industrial & Technical Chemistry. We have over one million books available in our catalogue for you to explore.