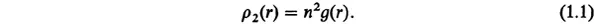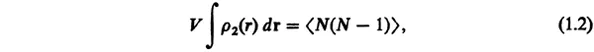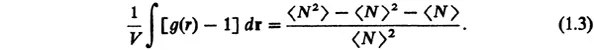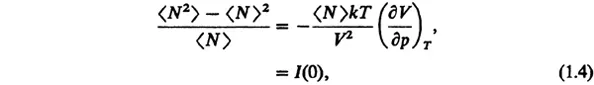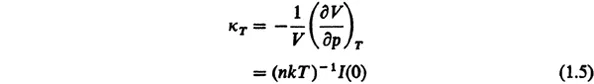![]()
1
SIMPLE LIQUIDS
One could well argue that it is more difficult to treat theoretically a liquid than a gas with a random distribution of molecules or a crystal with a regular structure. Nevertheless, its description is a good place to begin discussing condensed matter physics since the liquid state is no stranger to us and yet has strong molecular correlations. Theoretical techniques developed for a liquid are often applicable to other condensed matter systems.
1.1. Pair distribution function
The liquid state is similar to the solid state in having a free surface, low compressibility, and high density. In fact, its density is only about 5% lower than that of a solid, although there are exceptional cases such as water. However, its fluidity and molecular order are unique. Molecules in liquids can change position continuously, fill out a vessel with a formation of a meniscus, and do not have the long-range order of solids but only short-range order. Both liquids and gases are called fluids but these two are clearly distinguishable except near the critical point.
Since the molecular distribution of a liquid differs from that of a gas or a crystal, its study is important. This study can be performed effectively using two experimental methods: X-ray or electromagnetic wave diffraction and neutron scattering. The former method probes a static distribution that is quite different from those of gases and crystals. The latter provides information on the dynamic correlation of molecules, which is also distinctive. We shall discuss the former in this section and the latter in the next section.
X-ray scattering from a liquid depends not only on the molecular distribution but also on the molecular species. Therefore, we consider only a simple liquid (without any complex molecular structure), homogeneous and in equilibrium [1].
X-ray studies probe the radial distribution function g(r), which is connected with the pair distribution function ρ2(r) such that
The pair distribution function ρ2(r) represents the probability of finding two molecules at a relative distance r. For homogeneous and isotropic systems, this probability depends only on the magnitude of r and approaches n2 as r → ∞, where n is the number density. Hence, the radial distribution function g(r) has the asymptotic property:
g(r) → 1, (r → ∞).
Its deviation from this asymptotic value represents the molecular correlations. For a given distance r, ng(r)4πr2 dr is the number of molecules in the spherical shell of width dr at r.
The pair distribution function satisfies the normalization:
where V is the volume and N is the number of molecules in the liquid. The average 〈…〉 is taken if this total number is fluctuating. In such cases, ρ2(r) is defined in a grand ensemble where 〈…〉 represents a grand-ensemble average.
From Eqs. (1.1) and (1.2), we learn that the radial distribution function satisfies
For large 〈N〉, the right-hand side represents the square average fluctuations of the total number N about its average 〈N〉. One can show statistically [2] that
where I(0) is proportional to the intensity of scattered radiation at zero scattering angle and p is the pressure. Note that the quantity
is the isothermal compressibility. Light scattering will be discussed shortly.
Generally, g(r) stays 0 near the origin at r = 0 due to the strong short-range repulsion of molecules, reaches a maximum at a point where nearest-neighbor molecules gather, and then shows weaker maxima and gradually approaches its asymptotic value of 1. Its schematic curve is illustrated in Fi...