Hydrostatic Testing, Corrosion, and Microbiologically Influenced Corrosion: A Field Manual for Control and Prevention teaches industry professionals, managers, and researchers how to combat corrosion failure associated with hydrotesting. It discusses how a test liquid must be selected, how corrosion by bacteria should be controlled, and how to eliminate the risk of leakage. Rather than teaching how hydrotests should be conducted, it helps the reader evaluate the quality of a hydrotest that's already been conducted in terms of oxygen scavenger use, biocide testing, inhibitor addition, and water quality and explains the tasks that top and middle management must ensure are taken with respect to corrosion assessment of hydrotesting. The manual also discusses microbiologically influenced corrosion (MIC) as the main corrosion mechanism related to post-hydrotesting and offers essential knowledge on combating this corrosion process. In addition to being a manual for top and middle management on how to deal with corrosion, this book also:

eBook - ePub
Hydrostatic Testing, Corrosion, and Microbiologically Influenced Corrosion
A Field Manual for Control and Prevention
- 74 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Hydrostatic Testing, Corrosion, and Microbiologically Influenced Corrosion
A Field Manual for Control and Prevention
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Subtopic
MicrobiologiaChapter 1
A Review of the Essentials of Corrosion Needed to Assess Hydrostatic Testing
1.1 “Parallel” and “Series” Corrosion Scenarios
As we can all remember from our school days, and especially from the study of electricity, current can flow in one of two manners depending on whether resistors have been placed one after the other or in parallel with one another. The former, called a “series arrangement,” will show an overall resistance that is the sum of all the resistances, whereas in the latter, the “parallel arrangement,” the overall reciprocal of the resistance is equal to the sum of the reciprocals of the individual resistances.
In dealing with corrosion processes we can use the same analogy. Sometimes, corrosive processes can take place electrochemically in a series arrangement, where each will enhance the impact of the other—a synergistic impact. On the other hand, the corrosive processes can also act in a parallel manner, seemingly independently of one another, as shown schematically in Figure 1.1.
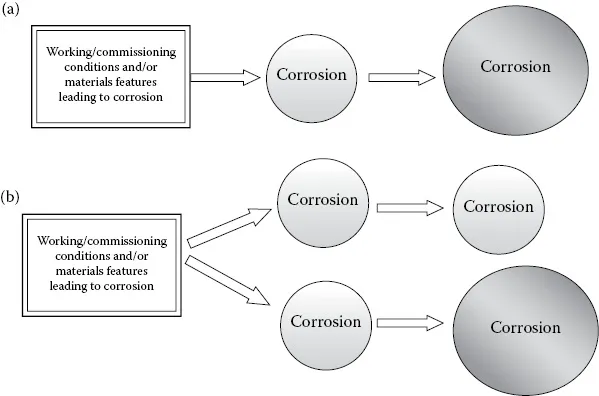
Figure 1.1 Corrosion processes arranged in series (a) and in parallel (b). The relative sizes reflect the severity of corrosion.
Figure 1.1 shows that a series corrosion process may simply originate from a corrosion problem that started because of the initial conditions (the chemistry of the system, materials selection, hydrostatic testing [HYD], and so on). This corrosion problem may be aggravated and cause more severe corrosion problems. In a parallel corrosion scenario, however, corrosion processes can evolve independently of each other, and the severity of corrosion (measured in terms of the corrosion rate) may remain the same (upper branch) or become worse (lower branch).
Understanding the concepts of series and parallel corrosion is of vital importance in analyzing the corrosion processes that can become involved during or after HYD is completed.
1.2 Corrosion Scenarios Likely to Develop as a Result of Hydrostatic Testing (HYD) Poor Practice
Corrosion is a “thermodynamically favorable” process, which simply means that the process will happen no matter what the conditions or how perfect the prevention/control measures. Corrosion will happen anyway, and all we can do is control it to make it as slow as possible. This means that the corrosion rates must be so low that, practically speaking, corrosion can be said to have been “under control.”
Anything that assists the “thermodynamically favorable” process of corrosion is undesirable. Factors such as poor welding (in welded parts), the use of maltreated water,* inadequate materials selection,† and so on could enhance the likelihood of corrosion occurring.
At present, we can address four corrosion scenarios most likely associated with poor HYD (of which microbial corrosion is the most probable) [2]:
▪Microbial corrosion (MIC)
▪Formation of electrochemical cells (oxygen concentration counts)
▪Galvanic corrosion
▪Underdeposit corrosion
We will describe MIC in Chapter 2, but here we will lightly touch on three points that may seem obvious, but are, in fact, quite confusing.
▪Oxygen is an integral part of any electrochemical corrosion. For this reason, oxygen scavengers are added when necessary (see Chapter 3) to exclude oxygen and thus control corrosion.
▪“Dissimilar metals corrosion” is an incorrect terminology and instead of that, “Galvanic corrosion” must be preferred to be used. Galvanic corrosion can occur on the very same metal if conditions allow. A typical example is when a segment of an old pipe is replaced with a new pipe made of the same material. If the pipes have not been taken care of adequately, the new pipe can become the anode and will corrode, despite being the same material as the rest of the pipe.
▪It is possible that the galvanic cell formed as a result of an underdeposit corrosion mechanism such as formation of underbiofilm differential aeration cells will form the required driving force for corrosion. This example alone should serve to show the complexity that may be involved in multiple corrosion reactions, setting some to each other as series and some as parallels. Another example of such is given below.
As shown in Figure 1.1, the four corrosion reactions described above can take place in series or in parallel. If, through poor corrosion management, too much deposit is formed inside a pipe, then underdeposit corrosion is inevitable. If, in addition to that, the water used for HYD is maltreated, then by increasing the likelihood of MIC we can face an unfortunate combination of both microbial and non-microbial corrosion taking place inside the pipe. Figure 1.2 shows a situation where the deposits within a pipeline have accumulated due to poor corrosion management and have formed a suitable location at which corrosion can easily proceed.*

Figure 1.2 Deposits removed after pigging through a 300 km pipe (a) and a close-up of the overall texture of the deposits (b). (Courtesy of Reza Javaherdashti.)
When welding is done poorly, poor welding and post-welding treatment (PWT) will lead to the formation of platforms upon which corrosion can initiate (with or without HYD). If the conditions are made worse by applying poor HYD using maltreated water for a long period of time, the corrosion that may have happened as a result of the poor welding will be enhanced by an unfortunate combination with microbial corrosion. Figure 1.3 shows welding defects formed during HYD.

Figure 1.3 Example of a failed line pipe manufactured by low-frequency electric resistance welding (LF-ERW) after HYD. (From J.P. Sinha, C.P. Varghese, Assessment of seam integrity of an aging petroleum pipeline constructed with low frequency ERW line pipes, 6th Pipeline Technology Conference 2011, Germany, 2011, http://www.pipeline-conference.com/abstracts/assessment-seam-integrity-aging-petroleum-pipeline-constructed-low-frequency-erw-line. With permission.)
Figure 1.4 presents yet another example of how poor welding practice can result in a post-HYD failure.

Figure 1.4 Brittle fracture during a pipeline hydrotest due to a discontinuity in a submerged arc welding (SAW) longitudinal weld. (From Massimo Benedetto. With permission.)
Therefore, it is essential to realize that what we do can enhance an already existing corrosion problem or will assist in enhancing its detrimental effect on the asset and equipment.
The outcome ...
Table of contents
- Cover
- Half Title
- Title Page
- Copyright Page
- Dedication
- Contents
- Author
- Introduction
- Chapter 1: A Review of the Essentials of Corrosion Needed to Assess Hydrostatic Testing
- Chapter 2: Microbiology of Corrosion, Microbiologically Influenced Corrosion (MIC), and Its Role in HYD
- Chapter 3: Assessment of HYD
- Annex 1: Quantification of MIC Risk Factors Based on Post- and Pre-HYD
- Annex 2: A List of Biocides and Their Pros and Cons
- Annex 3: Corrosion Knowledge Management
- References
- Abbreviations
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Hydrostatic Testing, Corrosion, and Microbiologically Influenced Corrosion by Reza Javaherdashti,Farzaneh Akvan in PDF and/or ePUB format, as well as other popular books in Tecnologia e ingegneria & Microbiologia. We have over 1.5 million books available in our catalogue for you to explore.