Nanotechnology is one of the growing areas of this century, also opening new horizons for tuning optical properties. This book introduces basic tuning schemes, including those on a single quantum object level, with an emphasis on surface and interface manipulation of semiconducting and metallic quantum dots. There are two opposing demands in current forefront applications of quantum dots as optical labels, namely high luminescence stability (suppression of luminescence intermittency) and controllable intermittency and bleaching on a single-particle level to facilitate super-resolution optical microscopy (for which Eric Betzig, Stefan W. Hell, and William E. Moerner were awarded the 2014 Nobel Prize in Chemistry). The book discusses these contradictory demands with respect to both an understanding of the basic processes and applications. The chapters are a combination of scholarly presentation and comprehensive review and include case studies from the authors' research, including unpublished results. Special emphasis is on a detailed understanding of spectroscopic and dynamic properties of semiconducting quantum dots. The book is suitable for senior undergraduates and researchers in the fields of optical nanoscience, materials science, and nanotechnology.

eBook - ePub
Tuning Semiconducting and Metallic Quantum Dots
Spectroscopy and Dynamics
- 406 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Tuning Semiconducting and Metallic Quantum Dots
Spectroscopy and Dynamics
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
Physical SciencesSubtopic
BiologyChapter 1
Size Matters: Optical Properties of Nanoparticles
Christian von Borczyskowskia and Eduard Zenkevichb
a Institute of Physics, Center for Nanostructured Materials and Analytics, Technische Universität Chemnitz, Reichenhainer Str. 70, D-09107 Chemnitz, Germany
b Department of Information Technologies and Robototechnique, National Technical University of Belarus, Prospect Nezavisimosti 65, 220013 Minsk, Belarus
1.1 Introduction
One of the fascinations about nanoparticles or nanocrystals (NCs) built from a few to several thousand atoms is the fact that they are from a quantum mechanical point of view a class of materials between atoms and solids showing properties of both of them. Though a deep understanding of their properties emerged only during the last few decades, the related versatility has been used already since centuries. Probably the most obvious feature is the dependence of properties, like the color, on the size of NCs, both for semiconducting and for metal particles. A lot of features can nowadays be understood in terms of quantum confinement resembling in its most simple approximation, the “particle in a box” model taught in undergraduate courses on quantum mechanics. NCs falling into this category are thought of as 1D quantum objects and have been also named “quantum dots” (QDs). We will use this name, or QDs, throughout the book. The fundamental optical excitations in a semiconducting QD depend on electrons, holes, and excitons in a 3D (confining) potential. Systematic research began in the early 1980s with the identification of quantum confinement in small semiconductor NCs. Different from scaling down nanostructures lithographically as is the present standard in micro-and nanotechnology, QDs are in most cases formed via bottom-up approaches in glasses or polymers or from solutions. In general, semiconductor nanostructures show an amazing variety of interesting properties different from conventional solid state materials. A recent comprehensive review outlines the prospects of nanoscience with NCs [Kovalenko et al., 2015].
Though quantum confinement and its consequences already open an enormously wide field with respect to both fundamental research and applications, QDs are not only in between atoms and solids but depend to a large extent on surface properties. This can be considered not as a disadvantage but just the opposite since elementary processes such as charge accumulation or catalysis depend essentially on interface properties. In this book, we will pay special attention from an empirical point of view to surface or interface features. Especially we will discuss how they can be tuned by various means.
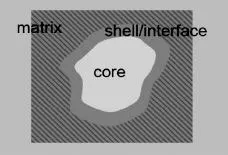
Figure 1.1 shows schematically what has to be taken into account when analyzing a QD, including influences outside the intrinsically confining potential. We have indicated that a crystal structure may not be perfect under realistic conditions. In any case a crystal structure includes facets and surface domains, depending on crystal symmetry. Besides the QD core (e.g., CdSe as a II–VI semiconductor), QDs may contain shells from other semiconductor materials (e.g., ZnS) and/or organic surfactants (ligands). The latter are essential when QDs are synthesized from solution to stop further growth or to protect from aggregation (Oswald ripening). Such a structure already constitutes two to three interfaces. In general, QDs are embedded in a matrix (liquid, polymer, glass). Moreover, in the case of thin films, a supporting substrate might also have to be taken into account. All these subsystems have to be followed since though the electronic wavefunctions are confined in a potential, they might tunnel through confining barriers, “exploring” the corresponding environment on a nanoscale. These considerations are essential, especially with respect to applications.
1.2 Quantum Confinement
1.2.1 Basic Concepts
A whole bunch of textbooks describes the treatment of excited states of (semiconductor) QDs, taking into account quantum confinement [Woggon, 1997; Gaponenko, 1998; Gaponenko, 2010; Rogach, 2008; Gavrilenko, 2011]. Therefore we will restrict ourselves here to the basic principles without going into detail. The band energy of a particle in a quasi-infinite bulk semiconductor is described by the dispersion relation
(1.1) |
with band-gap energy EG and the reciprocal effective mass 1/m* at k = 0 according to
(1.2) |
The band structure is characterized in Fig. 1.2 for II–VI semiconductors (e.g., CdSe or ZnS) with a direct band gap.
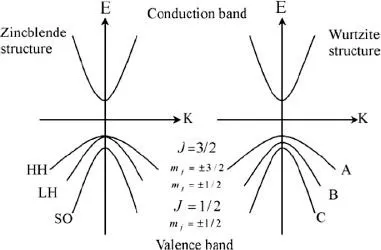
For semiconductors like CdS, CdSe, ZnS, or ZnSe, the conduction band is formed from s orbitals of the metal ions Cd or Zn, whereas the valence band corresponds to porbitals of the chalcogenides S or Se. While the conduction band can in most cases be approximated by parabolic potentials, the valence bands are partly degenerate and more complex, depending on the crystal structure (zinc-blende or wurtzite). We will describe the details after introducing quantum confinement.
Without quantum size effects the (optically generated) electron–hole pair is a hydrogen-like bound state, the so-called exciton. The interacting hole and electron (electron-hole pair) are described by the Hamiltonian in Eq. 1.3, which applies when the size of an NC is comparable to the critical length parameters, that is, the de Broglie wavelength λ and the exciton Bohr radius aB of the quasi-particles: electron, hole, and exciton. It is described as
(1.3) |
where is the effective mass of the electron (hole), respectively, and the static dielectric constant of the bulk semiconductor is ε2 (~9.5 for CdSe). The exciton is similarly, as a hydrogen atom, characterized by the exciton Bohr radius aB according to
(1.4) |
aB is in the range of 1–10 nm for typical II–VI semiconductors. In this range the QD is with respect to lattice constants of a macroscopic solid, but quantum size effects become important for the quasiparticles. Figure 1.3 shows schematically how the various scales (lattice constant, Bohr radius, crystal size) are related.
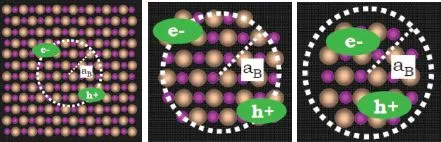
Table of contents
- Cover
- Half Title
- Title Page
- Copyright Page
- Dedication
- Table of Contents
- Preface
- Acknowledgments
- 1. Size Matters: Optical Properties of Nanoparticles
- 2. New Insights: Photophysics of CdSe Quantum Dots (Temperature-Related Approach)
- 3. Temperature-Dependent Exciton-Phonon Coupling in Quantum Dots
- 4. Optical Detection of Single Quantum Dots
- 5. Optical Spectroscopy of Single Ag Nanoclusters
- 6. Tuning Luminescence Intermittency
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Tuning Semiconducting and Metallic Quantum Dots by Christian von Borczyskowski, Eduard Zenkevich, Christian von Borczyskowski,Eduard Zenkevich in PDF and/or ePUB format, as well as other popular books in Physical Sciences & Biology. We have over 1.5 million books available in our catalogue for you to explore.