
- 293 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Pyrite Oxidation and Its Control
About this book
Pyrite Oxidation and its Control is the single available text on the market that presents the latest findings on pyrite oxidation and acid mine drainage (AMD). This new information is an indispensable reference for generating new concepts and technologies for controlling pyrite oxidation.
This book focuses on pyrite oxidation theory, experimental findings on oxidation mechanisms, as well as applications and limitations of amelioration technologies. The text also includes discussions on the theory and potential application of novel pyrite microencapsulation technologies for controlling pyrite oxidation currently under investigation in the author's laboratory.
Trusted by 375,005 students
Access to over 1 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
REVIEW OF BASIC CONCEPTS OF SURFACE CHEMISTRY AND CHEMISTRY AND CONTROL OF ACID DRAINAGE
Chapter 1
A REVIEW ON SOLUTION/MINERAL CHEMISTRY
1.1 Ion Activity versus Concentration
It is generally agreed that chemical reactions in nature (e.g., mineral dissolution/precipitation) take place because of electrical and chemical gradients. With regard to the former, there is disagreement as to how one may measure these gradients since they are not directly related to concentration (Amacher, 1984; Sposito, 1984; Sposito and Mattigod, 1979). Aided by computer technology, approaches have been introduced that allow estimation of single-ion activity (a direct reflection of the chemical potential of ionic species) in mixed electrolyte systems (Davies, 1962; Adams, 1971; Sposito, 1984). Experimental data (Pavan and Bingham, 1982; Sposito, 1984) support the argument that single-ion activity best describes/predicts chemical reactions.
Single-ion activity in solution is estimated by employing the equation of effective ionic strength (I)
(1.1) |
where Zj and mj are the charge and effective molar concentration of ionic species (Sposito, 1984). Based on I, activity coefficients of ionic species can be calculated using the Davies equation (Davies, 1962):
(1.2) |
where γ is the activity coefficient of ionic species j, and A is a constant equal to 0.512. Single ion activity (αj) can then be estimated by:
(1.3) |
The above calculations necessitate the use of ion association models to make estimates of concentration of ionic species (mj) using as inputs elemental concentrations (see section 1.1.3, Ion pairing) (Adams, 1971; Sposito and Mattigod, 1979; Sposito, 1984; Evangelou, 1986). However, direct measurement of single-ion activity in mixed electrolyte systems for the purpose of verifying ion association models cannot be made (Amacher, 1984; Sposito, 1984); validity of ion-association models can only be demonstrated indirectly.
In order to gain an appreciation of the importance of single-ion activity in describing/predicting chemical reactions as opposed to elemental concentration, one needs to look into the effect of ionic strength and ion valence on single-ion activity coefficients (γ) (Equation 1.2). This is demonstrated in Figure 1.1. It shows that at an ionic strength of 0.04 M, the γ value for say Ca2+, is approximately 0.5, while that of a monovalent ion is approximately 0.8.
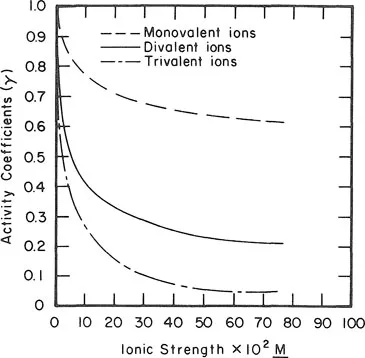
Figure 1.1. Relationship between solution ionic strength and activity coefficients of ions of different valencies. Calculated utilizing the Davies equation (Davies, 1962). (From Evangelou and Sobek, 1988. With permission.)
Examples of practical implications of single-ion activity with respect to pH and mineral solubility are given in sections 1.1.1 and 1.1.2.
1.1.1 Solution pH
Most reactions in soil/geologic systems are pH driven because of the hydrogen’s (H+) great potential to react with solution chemical species and/or mineral surfaces due to its small size and high electron accepting potential.
Technically, pH is the negative logarithm of the hydrogen ion activity (Equations 1.4)
(1.4) |
The pH scale ranges from 0 to 14, with 7 being the neutral point. Values greater than 7 are basic or alkaline, while values less than 7 are acidic. For example, if the hydrogen ion activity of a water sample is 10−7 mol L−1, its pH is 7, since 7 is the negative logarithm of 10−7.
Water emanating from surface mine sites for example can vary in ionic strength (I) anywhere from 0.001 to 0.1. Values greater than 0.1 can be observed in acid mine drainages (Lekhakul, 1981). For a solution with an I of 0.008 M, the γ value for H+ will be 0.9. On the other hand, a solution with an I value of 0.04 M will exhibit a γ value for H+ of 0.8. Since pH represents H+ activity, the same pH in the above systems would represent a different H+ concentration. For example, at pH of 3 and at an I value of 0.008 M:
(1.5) |
while at an I of 0.04 M
(1.6) |
Therefore, at an ionic strength of 0.008 M the H+ activity represents 90% of its concentration, while at an ionic strength of 0.04 M, the H+ activity represents 80% of its concentration.
The above example demonstrates that even in the case of a monovalent ion...
Table of contents
- Cover
- Title Page
- Copyright Page
- Table of Contents
- INTRODUCTION
- PART I REVIEW OF BASIC CONCEPTS OF SURFACE CHEMISTRY AND CHEMISTRY AND CONTROL OF ACID DRAINAGE
- PART II MOLECULAR CHEMISTRY OF PYRITE OXIDATION KINETICS AND PYRITE REDOX CHEMISTRY
- PART III ROLE OF BACTERIA ON PYRITE OXIDATION
- PART IV ADVANCES ON PYRITE MICROENCAPSULATION TECHNOLOGIES
- PART V SUMMARY
- REFERENCES
- SUBJECT INDEX
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.4M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Pyrite Oxidation and Its Control by V. P. Evangelou in PDF and/or ePUB format, as well as other popular books in Physical Sciences & Chemistry. We have over one million books available in our catalogue for you to explore.