
eBook - ePub
Pvp
A Critical Review of the Kinetics and Toxicology of Polyvinylpyrrolidone (Povidone)
- 232 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Pvp
A Critical Review of the Kinetics and Toxicology of Polyvinylpyrrolidone (Povidone)
About this book
Polyvinylpyrrolidone is widely used in medicine, pharmaceuticals, cosmetics, foods, printing inks, textiles, and many more diverse applications. This book describes the 50 years of research, published and unpublished, on the absorption, distribution, storage, and excretion of PVP. The toxicology of PVP is critically evaluated.
The author's involvement in the recent reevaluation of PVP by the Joint Expert Committee on Food Additives of the World Health Organization (WHO) and the Food and Agriculture Organization (FOA) led them to undertake this comprehensive review of all the information on the subject. This book will be invaluable for anyone who is involved with polyvinylpyrrolidone. Included is a broad review of the toxicological studies performed on PVP, including acute, subchronic, chronic, reproductive, mutagenicity, and carcinogenicity studies. There is also an appendix listing the key studies, with references, on the absorption, renal elimination, distribution, acute toxicity, subchronic toxicity, chronic toxicity, teratogenicity, mutagenicity, and carcinogenicity of PVP.
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
MedicineSubtopic
Toxicology1
Introduction
Polyvinylpyrrolidone (PVP, povidone) is the generic name for the water-soluble homopolymer of N-vinyl-2-pyrrolidone (NVP), and in this review it will be referred to simply as PVP. Polyvinylpolypyrrolidone (PVPP, crospovidone) is the generic name of the cross-linked insoluble homopolymer of NVP.
First developed in Germany at I.G. Farben by Professor Walter Reppe and his colleagues during the 1930s, PVP was subsequently widely used as a blood-plasma substitute and extender during World War II. It has the advantages of being nonantigenic, requiring no cross-matching and avoiding the dangers of infectious diseases inherent in blood. In later years, it has been superseded by other materials. However, the issue concerning the effectiveness of 3.5% PVP (K-30) in an isotonic saline solution for parenteral administration was part of the U.S. Drug Efficacy Study Implementation (DESI) review program, and was reported to be effective for the correction of low blood volume in the treatment of shock (Fed. Reg., 1971).
PVP now finds a large number of other uses. It derives its commercial success from its biological compatibility, low toxicity, film-forming and adhesive characteristics, unusual complexing ability, relatively inert behavior toward salts and acids, and its resistance to thermal degradation in solution. Because of these diverse properties, PVP finds important applications in a number of industries, especially the pharmaceutical, food, beverage, cosmetic, toiletry and photographic industries (Hort and Gasman, 1983).
PHARMACEUTICAL USE
PVP, because it becomes sticky when wetted by water and many solvents, is used extensively as a tablet binder. Tablets bound with PVP exhibit reliable rates of drug dissolution, which can also permit its use in sustained-release preparations. PVP is used as a tablet coating aid, and has found use in the formulation of antibiotics, hormones and analgesics for parenteral administration, and also of ophthalmic and topical preparations.
The formulation of poorly water-soluble drugs has challenged the pharmaceutical industry during recent years, and PVP has been found to be useful in the preparation of solids, solutions, and suspensions where enhanced solubility has increased bioavailability. Very recently, it has found use as a carrier to create a gel-like matrix for the transdermal application of drugs.
Iodine complexed with PVP (povidone iodine) is widely used as a germicide in antimicrobial soaps, surgical hand scrubs, patient preoperative skin cleansers, antiseptics and skin wound cleansers.
FOODS AND BEVERAGES
The WHO Joint Expert Committee on Food Additives (JECFA) has granted an Acceptable Daily Intake (ADI) of 0–50 mg/kg/day for PVP (WHO, 1986). Under United States law PVP has a number of permitted uses in foodstuffs, including use as a binder for vitamin and mineral concentrate tablets and as a binder for synthetic sweetener tablets. It is also used as a stabilizer for liquid vitamin and mineral concentrates and to prevent crystallization of liquid synthetic sweetener preparations.
It is also used as a diluent and dispersant for food colors. PVP is permitted as a coating for fresh citrus fruit and as a component in the packaging materials which are in contact with aqueous, fatty and dry foods.
PVP has been used in the chill-proofing of beer and the color stabilization of beverages such as white wines, fruit juices and vinegar. PVP has found use because of its ability to complex and form insoluble precipitates with certain polyphenolic compounds, such as the anthocyanogens in beer and wine. PVP has been replaced in this application by the insoluble PVPP.
COSMETICS AND TOILETRIES
PVP and its copolymers are used as a thickener, dispersing agent, lubricant and binder in the cosmetics industry. The polymers are particularly suitable adjuvants for skin cleansing and protection preparations, and hair tints and dressings. Many creams, either greasy or nongreasy, can be prepared from PVP. It acts as a stiffener in hair setting lotions and improves the consistency of shampoos and household detergents to protect the skin. In contrast to anionic colloids, PVP can be combined with cationic substances, e.g., disinfectants or basic dyes, but allowance must be made for the fact that it has a marked affinity for dyes.
PHOTOGRAPHIC PRODUCTS
PVP acts as a protective colloid and silver halide – suspending agent; it is used as a processing aid in the development of silver halide film and to eliminate the occurrence of dichroic stain. As a coating aid, PVP in silver halide emulsions reduces viscosity and increases the covering power of the developed image.
DYEING APPLICATIONS AND INKS
PVP improves the solubility of dye-based inks to give a greater color value per weight of dye. In pigmented inks it increases tinctorial strength, dispersion stability, viscosity and gloss. It is used as a pigment dispersant in paper. It enhances dye receptivity in paper coatings, and hydrophobic fibers such as polyacrylonitrile, polyesters, nylon and cellulosics.
DETERGENTS
PVP functions as a builder in clear, liquid, heavy-duty detergent preparations. It improves soil-suspending capacity.
SUSPENSIONS, DISPERSIONS, AND EMULSIONS
Additions of PVP stabilize aqueous suspensions, dispersions and emulsions. They are adsorbed in a thin layer on the surface of the colloidal particles and thus prevent contact with other colloidal particles.
PRODUCTION OF PLASTICS
In the production of solid polymers, PVP is used as a protective colloid and polymerization stabilizer.
ADHESIVES
In view of its good water solubility, PVP is a useful component in adhesives for rewettable bonding.
PAINTS AND COATINGS
In emulsion paint and other dispersions for coating, PVP finds use as a protective colloid and dispersing agent to influence the rheology of the dispersions.
PAPER AUXILIARIES
In view of its good water solubility, PVP is useful in coated paper that will be rewetted at a later stage. In this application the PVP prevents curling and imparts a smooth surface to the paper.
TABLETTING AUXILIARY
PVP is a binder for all types of tablets for nonpharmaceutical applications, e.g., cleaning tablets for false teeth or paint tablets for children’s paint boxes.
OTHER INDUSTRIAL USES
PVP, because of its unique properties, for example, stability over a wide pH range, is useful in various areas of oil recovery; as an additive to cement formulations to increase viscosity and setting time while decreasing fluid loss; in the plastics industry as a particle size regulator, suspending agent and viscosity modifier, and in many other areas of the chemical industry.
2
Synthesis and Properties of PVP
SYNTHESIS OF PVP
PVP is readily prepared by the polymerization of N-vinyl-2-pyrrolidone (N-vinyl-2-pyrrolidinone, NVP). Traditionally, the synthesis of N-vinyl-2-pyrrolidone and the polymerization of the homopolymer have been based on the Reppe acetylene chemistry represented by the following series of reactions:
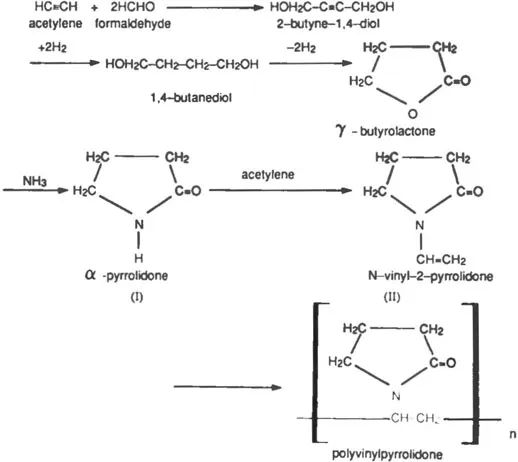
Figure 1. Reppe synthesis of PVP.
The reaction of α-pyrrolidone (I) with acetylene under pressure in the presence of a catalyst yields N-vinyl-2-pyrrolidone (II). A simplified version of polymerization may be visualized as a coupling of the unsaturated −CH = CH2 groups of several N-vinyl-2-pyrrolidone molecules to form a chain of carbon atoms, to which pyrrolidone rings are attached through their nitrogen atoms:
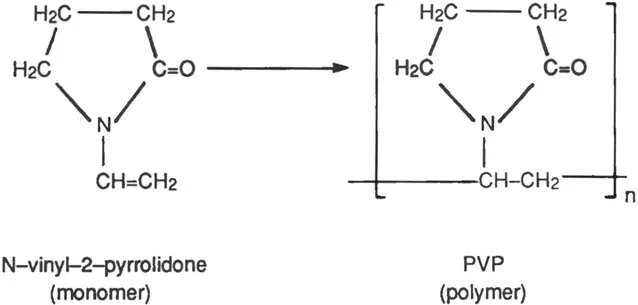
The relationship between N-vinyl-2-pyrrolidone (monomer) and PVP (polymer) may be represented more concisely as follows:
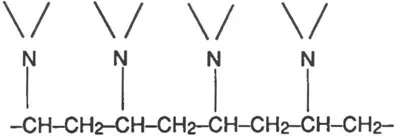
where n stands for the number of N-vinyl-2-pyrrolidone (monomer) molecules which have united to form a molecule of PVP (polymer), or in other words, the degree of polymerization of the product.
Currently there are two basic approaches being used commercially for the polymerization of PVP. Most commonly, PVP is polymerized in water as described by Reppe (1949). Used to a lesser extent, but being practiced commercially, is polymerization in isopropyl alcohol.
Both methods yield polymers in solution form. In the isopropyl alcohol method th...
Table of contents
- Cover
- Half Title
- Title Page
- Copyright Page
- Table of Contents
- 1 Introduction
- 2 Synthesis and Properties of PVP
- 3 Movement of Large Molecules Across Membranes – General Considerations
- 4 Absorption of PVP by Various Routes of Administration
- 5 Excretion and Metabolism of PVP
- 6 Distribution and Storage of PVP
- 7 Storage of PVP in Humans
- 8 Functional Consequences of PVP Uptake by Body Tissues, with Particular Reference to the Reticuloendothelial System (RES) and the Immune System
- 9 Toxicological Studies on PVP
- 10 Overall Summary and Conclusions
- References
- Appendix
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Pvp by Wolfgang Schwarz in PDF and/or ePUB format, as well as other popular books in Medicine & Toxicology. We have over 1.5 million books available in our catalogue for you to explore.