![]()
Chapter 1 Electrical fundamentals
This chapter will provide you with an introduction to the essential electrical theory that underpins the rest of this book. It has been designed to help fill any gaps in your knowledge and bring you quickly up to speed. You will find this chapter particularly useful if you have not previously studied electrical principles. However, if you are in any doubt as to whether or not you should work through this chapter you can always turn to Section 1.9 on page 37 and see how you get on with the multiple choice questions at the end of this chapter.
1.1 Electron theory
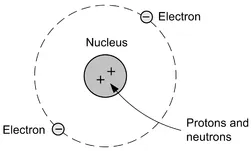
All matter is made up of atoms or groups of atoms (molecules) bonded together in a particular way. In order to understand something about the nature of electrical charge we need to consider a simple model of the atom. This model, known as the Bohr model (see Fig. 1.1), shows a single atom consisting of a central nucleus with orbiting electrons.
Within the nucleus there are protons which are positively charged and neutrons which, as their name implies, are electrically neutral and have no charge. Orbiting the nucleus are electrons that have a negative charge, equal in magnitude (size) to the charge on the proton. These electrons are approximately two thousand times lighter than the protons and neutrons in the nucleus.
In a stable atom the number of protons and electrons are equal, so that overall, the atom is neutral and has no charge. However, if we rub two particular materials together, electrons may be transferred from one to another. This alters the stability of the atom, leaving it with a net positive or negative charge. When an atom within a material loses electrons it becomes positively charged and is known as a positive ion, when an atom gains an electron it has a surplus negative charge and so is referred to as a negative ion. These differences in charge can cause electrostatic effects. For example, combing your hair with a nylon comb may result in a difference in charge between your hair and the rest of your body, resulting in your hair standing on end when your hand or some other differently charged body is brought close to it.
Figure 1.1 The Bohr model of the atom
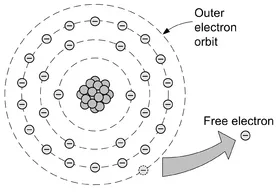
Figure 1.2 A material with a loosely bound electron in its outer shell
The number of electrons occupying a given orbit within an atom is predictable and is based on the position of the element within the periodic table. The electrons in all atoms sit in a particular orbit, or shell, dependent on their energy level. Each of these shells within the atom is filled by electrons from the nucleus outwards, as shown in Fig. 1.2. The first, innermost, of these shells can have up to two electrons, the second shell can have up to eight and the third up to 18.
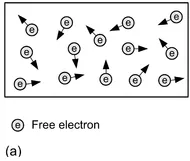
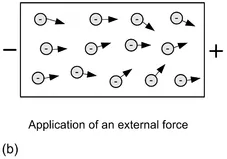
Figure 1.3 Free electrons and the application of an external force
All electrons and protons carry an electrostatic charge but its value is so small that a more convenient unit of charge is needed for practical use which we call...