![]()
CHAPTER 1
The Theory of Electricity
“Electricity is really just organized lightning.”
George Carlin
WHAT IS ELECTRICITY?
For thousands of years, lightning (electricity) puzzled, mystified, and terrified people. Over the past few hundred years, scientists such as Benjamin Franklin, André-Marie Ampère, Alessandro Volta, Michael Faraday, and many others pieced together an understanding of electricity and helped to unlock its secrets. Step by step, bit by bit, they built a plausible model of electricity that fit mathematical formulas and provided a real-world explanation of the phenomenon Even after they had a basic understanding of the key relationships and the fundamentals of electricity, early pioneers such as Joseph Swan, Thomas Edison, Nikola Tesla, and George Westinghouse still struggled to harness its power for daily use in a safe and efficient manner.
During that time—the late 1800s and early 1900s—one of the first practical uses of electricity was to illuminate common areas such as city streets and town squares. New York City quickly became entangled—quite literally—in electrical wires and electricity. Several times, horrified bystanders witnessed the accidental electrocution of workers in the naked light of day, and electricity gained a reputation for being both mysterious and dangerous. Thomas Edison played on the fear of the public to protect his economic interests by promoting DC power distribution as being safer than AC power distribution, while George Westinghouse grew his business on the principles of AC and its inherent economic advantages over DC The ensuing controversy did nothing to ease the public’s apprehension about electricity, nor did it help to clarify its nature or promote its understanding.
To this day, many people have little understanding of the nature of electricity. Some of us still have difficulty answering the question, “What is electricity?” After all, we can’t see it, hear it, or smell it. And we certainly don’t want to taste it or feel it.
An electrician might understand how to hook up a power distribution system but may not fully understand exactly how electricity behaves. By studying the fundamentals of electricity we can better understand how to use electricity safely, effectively, and within the confines of codes and regulations, which will help us excel at our jobs in the entertainment industry while keeping us safer.
ELECTRONS IN MOTION
The short answer to the question “What is electricity?” is that it is the transfer of energy through the motion of charge-carrying electrons. Lightning is an example of electricity and of electrons—lots and lots of them—in motion Electricians are generally concerned with a much more controlled situation where electricity flows through a given path in a safe, predictable manner. But the electricity we use in shows is no different than that of a lightning strike, a static discharge, or a flashlight battery supplying power to an LED. Each is an example of the transfer of energy through the motion of electrons.
But from where do these electrons come? The answer can be found in one of the most basic building blocks of the universe, the atom.
THE ATOM
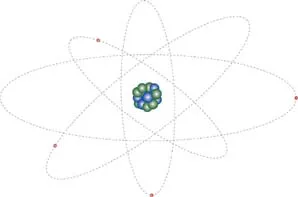
The word “atom” comes from the Greek word atomos, meaning indivisible. It is the smallest particle that still retains the properties of the element from which it comes. When you were in school, you were probably taught the classic model of an atom with a nucleus at the center and electrons orbiting around it. The nucleus is made up of a number of protons and neutrons, and they are held together by nuclear forces. The specific number of neutrons, protons, and electrons depends on the element. For example, copper atoms normally have 29 protons, 35 neutrons, and 29 electrons.
FIGURE 1.1
The classic model of an atom showing the nucleus at the center and electrons in orbit around the nucleus. (Not to scale.)
If you took your side cutters and cut a small strand of copper from a cable, you would have billions of copper atoms. If you then cut that piece in half, and then in half again, and over and over until you got down to the single piece that still looked and acted like copper, then you would have an atom But you would have to be pretty good with those cutters. One atom of copper is approximately 10−12 meters in diameter. Put another way, it takes about 254 billion copper atoms placed side by side to make 2.5 centimeters (1 inch). Good luck with that.
FIGURE 1.2
It would take approximately 254 billion copper atoms to span a 2.5-centimeter (1-inch) width of copper wire.
Atoms are literally everywhere. They make up the air you breathe, the water you drink, the clothes you wear, and the food you eat. They are the building blocks of the universe.
SUBATOMIC PARTICLES
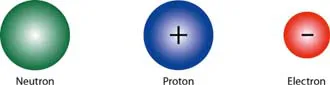
Despite what the early Greeks thought, atoms can be divided. It turns out that they are made up of even smaller subatomic particles called electrons, neutrons, and protons. These subatomic particles are very important to the understanding of electricity. Electrons carry a negative charge, protons have a positive charge, and neutrons have no charge at all. It’s the interaction of these charges that causes the phenomenon we call electricity.
FIGURE 1.3
Atoms are made up of particles call neutrons, protons, and electrons. Neutrons carry no charge, protons are positively charged, and electrons are negatively charged. (Not to scale. Color was added by the author to distinguish between the different particles.)
ELECTROSTATIC ATTRACTION AND REPULSION
Notice that the copper atom has the same number of electrons as it has protons. The charge on a single electron is the same strength as a single proton but its opposite in polarity. In most atoms, the number of electrons is the same as the number of protons, so the charges cancel each other resulting in an overall charge of zero. Sometimes an atom can lose an electron or two because some other force external to the atom, such as a battery, can pull it away. In that case, the charges in the atom are no longer balanced and it becomes positively charged.
One of the fundamental laws of nature is that opposite charges attract and ...