![]()
1
Electroluminescence in Small Molecules
TETSUO TSUTSUI
CONTENTS
1.1 Introduction
1.2 Recombination and Emission in Bulk
1.3 Multilayer Structures
1.4 Design of Hole Transport, Electron Transport, and Emissive Molecules
1.5 EL Quantum Efficiency and Power Conversion Efficiency
1.6 Concluding Remarks
References
1.1 INTRODUCTION
Small-size color displays (e.g., mobile phones) based on organic light-emitting diodes (OLEDs) have recently penetrated the commercial market. The attraction with this new technology is based on the bright, full-color light emission from OLEDs, which originates from the radiative relaxation of the electronic excited states of π-conjugated molecular systems. The size of the OLED displays is expected to expand in the near future and will include large TV screens, other information displays, and even solid state lighting.
In the last century, much effort has been devoted to developing a variety of artificial lighting sources such as gas lamps, electric light bulbs, fluorescent lamps, neon lamps, cathode-ray tubes, inorganic light-emitting diodes, and semiconductor lasers. Every artificial light source is based on simple mechanisms. For example, incandescence is based on short-wavelength edge emission of blackbody radiation from high temperature substances. Light emission from excited states of atoms or inorganic solids is another mechanism. In contrast, a variety of colors of living species are based on reflection or transmission, which are due to optical transitions between the excited and ground electronic states of molecules. There are animals and plants, such as moonlight mushrooms, luminous bacteria, lantern fish, sea firefly, firefly squid, firefly, and railroad worm that have the ability to continuously emit light and thus are quite visible in the dark. Even the advanced technologies achieved in the 20th century have not been successful in imitating the lighting mechanisms that take place in such living species. Fortunately, however, just at the end of the 20th century, the technology on OLEDs, the artificial version of living light, has been successfully established.
The mechanism for charge injection electroluminescence (EL) in covalently bonded molecular materials is common and is found among many molecular systems, including mainchain π-conjugated polymers. In this chapter, we describe charge injection EL in molecular glass films composed of small molecules. In the case of thin solid films composed of small molecules with unsaturated bonds, there is no need to describe their electronic structure using an extended π-electron system. Their electronic properties can simply be given in terms of their molecular orbitals, in particular the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO). Thus, charge transport across the molecular solid can be described as a hopping process among localized π-electron systems, where the location for opposite charge recombination and light emission is assumed to be on specific molecules. This simple picture for charge-injection EL serves as a useful guide. It is helpful when using different molecular design concepts and provides a reasonable basis for device efficiency considerations.
1.2 RECOMBINATION AND EMISSION IN BULK
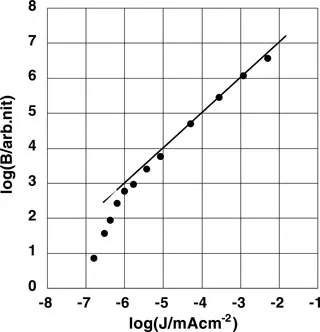
It is useful to look at the early work (1960s) on electroluminescence (EL) in organic single crystals to understand the high performance exhibited by OLEDs based on very thin films. Helfrich and Schneider reported bright EL from a single-crystal anthracene, where they used anthracene cation and anion containing electrolyte solutions as anode and cathode, respectively.1,2 Figure 1.1 shows the data replotted from this study where the emission intensity scales linearly with the injected current density over more than three decades. This result is quite valuable and it provides clear-cut evidence for a charge-injection process, which is the basis for determining the EL quantum efficiency defined as the ratio of photons emitted per injected charges. Helfrich and Schneider1,2 reported that the emission arose predominantly from a region near the positive electrode (anode), indicating that electrons are injected from the cathode and transported through the crystal to meet the holes entering from the opposite electrode. This result clearly shows that bulk-controlled charge transport and recombination govern the process of charge injection EL.
Figure 1.1 The relationship between the brightness and the current density in an OLED made of an anthracene single crystal. (Data from figure 2 in Reference 1 is re-plotted.) The thickness and size of electrodes are 5 mm and 0.2 cm2, respectively.
Following this early work, numerous reports on electroluminescence from various organic single crystals have appeared.3, 4, 5, 6, 7, 8 It is worthwhile noticing that the reported EL quantum efficiencies decreased as the troublesome and unstable liquid contacts were replaced with their more stable but less efficient solid counterparts. The development of stable solid state contacts with good injection characteristics for both electrons and holes is an important area deserving further attention. Further improvements will not only lead to better EL efficiency but also to overall improved device stability. In molecular systems, which have very low carrier densities, the injected positive and negative charges (referred to as electrons and holes for simplicity) are expected to exist as space charges with no local charge neutrality. This case is clearly different from inorganic semiconductor LEDs, in which minority charge carrier injection at a p–n junction determines the charge recombination process.
OLEDs based on single crystals (µm) are not useful for practical applications. The high voltages, the small light-emitting areas, and the difficulty of single crystal processing are some of the shortfalls that would prevent their use as pixel elements in displays and solid state lighting. This led to the exploration of means to fabricate thin film (nm) structures, and charge injection EL was reported in Langmuir-Blodgett films, vacuum-sublimed polycrystalline films, and vacuum-sublimed amorphous glassy films.9, 10, 11, 12, 13, 14 Both the EL quantum efficiencies and stabilities of those thin-film devices remained low until the breakthrough work of Tang and VanSlyke.15 In 1987, they reported the fabrication of high-performance OLEDs using a double-layer vacuum-sublimed heterostructure based on organic dyes.15 Following this demonstration, Adachi and co-workers extended and generalized the concept to multilayer structures,16, 17, 18, 19, 20, 21 and developed high-efficiency devices. It should be stressed, however, that charge recombination and light emission still originate from molecules in the bulk.
1.3 MULTILAYER STRUCTURES
The structure of OLEDs containing two organic layers consists of a transparent indium-tin-oxide (ITO) anode, an organic hole transpo...