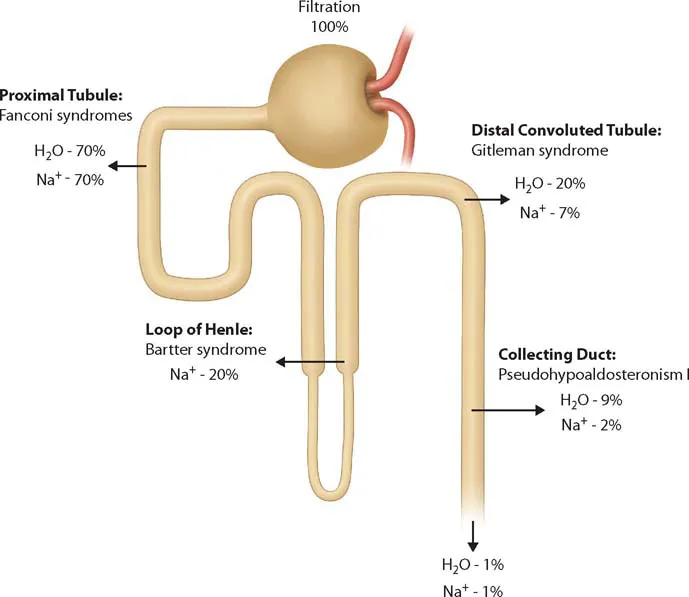
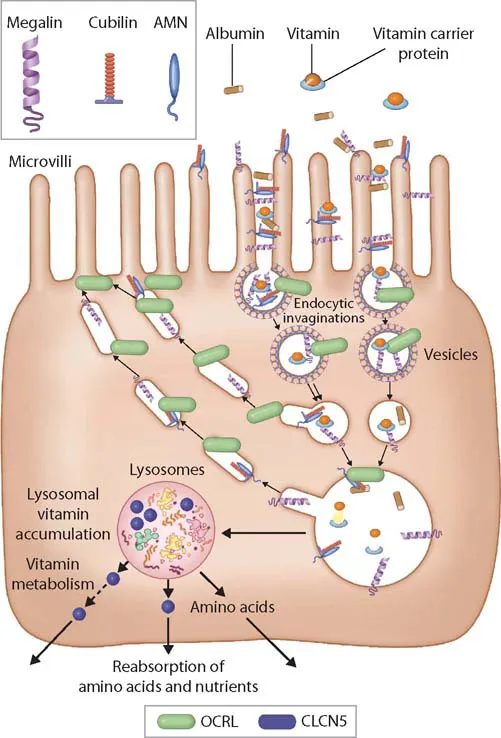
The third edition of a bestseller, this book provides insight from a wide array of international contributors in the field of pediatric nephrology. Copiously illustrated with photomicrographs and clinical diagrams, the third edition reflects current advances in the field.
Each chapter contains a set of questions, directed at helping fellows succeed at the American Pediatric Nephrology Board examination. New information for this edition includes changes in treatment options for hyponatremia, and updates on hyperparathyroidism and transplantation. Text boxes highlight important "take home points" throughout the chapters.
Clinical Pediatric Nephrology, Third Edition will be a valuable reference for clinicians in nephrology, pediatrics and urology, and any professional involved in the care of children with renal diseases seeking a reliable contemporary text.