
eBook - ePub
Pharmaceutical Biocatalysis
Fundamentals, Enzyme Inhibitors, and Enzymes in Health and Diseases
- 774 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Pharmaceutical Biocatalysis
Fundamentals, Enzyme Inhibitors, and Enzymes in Health and Diseases
About this book
This book provides an overview of the world market of therapeutic enzymes and enzyme inhibitors, rare diseases, orphan drugs, the costs of drug development and therapies, and enzymes in downstream processing of pharmaceuticals. It discusses carbonic anhydrase inhibitors and their multiple drug interactions, carboxylesterase inhibitors for pharmaceutical applications, employment of inhibitors for the treatment of neurodegenerative diseases, use of engineered proteins, bioactive peptides, and fibrinolytic enzymes for thrombolytic therapy, and enzymes important for the design and development of new drugs/drug metabolites such as aldehyde oxidases and cytochrome P450 enzymes and the role the latter play in vascular biology and pathophysiology. The treatment of cancer is explored in connection with enzymatic amino acid deprivation therapies and new drugs that act as chemical degraders of oncogenic proteins. The book also introduces the resistance mechanisms of cancer. Furthermore, it provides an insight into the relationship between pathological conditions of cardiovascular disease and oxidative stress. The text also focuses on the potential use of nanoparticles as carriers for enzymes with medical relevance, computer-aided drug design for the identification of multi-target directed ligands, and the development of improved therapeutics through a glycan-"designer" approach. It concludes with an introduction to the chemoenzymatic synthesis of drugs.
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
MedicineSubtopic
Biotechnology in MedicineChapter 1
Pharmaceuticals: Some General Aspects
Department of Physical Chemistry, University of Hamburg, D-20146 Hamburg, Germany
1.1 Introduction
In this chapter, apart from brief general remarks, some aspects of enzymes and enzyme inhibitors employed for therapeutic applications will be discussed. The topics are, among others, the following:
• The world market of these drugs
• Regulatory authorities (mainly the US FDA and the EMA)
• Quality standards
• Rare diseases and orphan drugs
• Therapeutic enzymes and enzyme inhibitors
• Cost of drug development and therapies
• Future developments
The term “drug” is used here in connection with recombinant therapeutic proteins (for the world market of therapeutic proteins, see Research and Markets, 2016) as well as chemically synthesized small molecules that have been approved for the treatment and prevention of diseases. Not included are diagnostic imaging agents as, e.g., Netspot (detection of rare neuroendocrine tumors) or Axumin (detection of recurrent prostate cancer) both approved by the FDA in 2016, and other compounds used for diagnostic purposes. The targets of small molecule drugs are proteins to which they bind to exert therapeutic efficacy; enzymes involved in drug metabolism are not defined here as drug target. The WHO Collaborating Centre for Drug Statistics Methodology classifies drugs by The Anatomical Therapeutic Chemical (ATC) Classification System (ATC and ATCvet) with respect to the organ or system, on which they act, their therapeutic, pharmacological, and chemical properties, etc., including, e.g., defined daily drug doses (WHOATC 2018).
1.2 General Remarks
Enzyme inhibitor drugs are used to treat chronic diseases and disorders such as cardiovascular diseases (angina and myocardial infarction, heart failure, hypertensive heart and rheumatic heart disease, etc.), gastrointestinal diseases, Diabetes Type 2, pain, fever, inflammation, neurodegenerative diseases such as Parkinson’s and Alzheimer’s, asthma and COPD, psoriasis and psoriatic arthritis, different infectious diseases (trypanosomiasis, leishmaniasis, tuberculosis, malaria, acquired immunodeficiency syndrome, etc.), a variety of different types of cancers (see below), some of the rare lysosomal storage diseases, life style related conditions (erectile dysfunction, benign prostatic hyperplasia, alopecia) and many others.
Pharmaceutical enzymes may be categorized as enzymes in replacement therapy, enzymes in cancer treatment, enzymes for fibrinolysis, enzymes to cure digestive disorders and enzymes that are used topically for various treatments (Yari et al., 2017). Modern DNA technology together with other approaches such as PEGlation or glycoengineering generates enzymes with extended half-life and reduced or no immunogenicity.
The Food and Drug Administration (FDA or USFDA) a federal agency of the United States Department of Health and Human Services is—together with several FDA organizations among them the Center for Drug Evaluation and Research (CDER)—responsible for controlling apart from cosmetics, animal foods and feed, tobacco products, dietary supplements and veterinary products, new drug application (new molecular entities, NMEs) and prescription, over-the-counter pharmaceutical drugs, generic drugs, vaccines, biopharmaceuticals, etc. An NME is a drug with an active moiety that is approved by the FDA as a single ingredient drug or as part of a combination product or marketed in the US for the first time; hence, treatment with an NME is often tantamount to a new therapy for patients.
Its European counterpart is the European Medicines Agency (EMA), which recently moved from London to Amsterdam. The function of the EMA (2017, 2018) is to protect “public and animal health in 28 EU Member States, as well as the countries of the European Economic Area, by ensuring that all medicines available on the EU market are safe, effective and of high quality; the EMA serves a market of over 500 million people living in the EU.” A European medicines regulatory network aims among others on reducing the administrative burden through the centralized authorization procedure, translating into medicines reaching patients faster. The EMA has a number of committees for Medicinal Products for Human Use, Pharmacovigilance Risk Assessment, for Medicinal Products for Veterinary Use, for Orphan Medicinal Products, Herbal Medicinal Products, for Advanced Therapies, and a Paediatric Committee.
According to more recent developments, the FDA offers an Expedited Program for speeding up the availability of drugs that treat serious diseases: Fast track (the aim is to facilitate the development, and expedite the review of drugs to treat serious conditions, filling an unmet medical need), breakthrough therapy (substantial improvement over available therapy), priority review (FDA’s goal is to take action on an application within 6 months), and accelerated approval (allows drugs to be approved based on a surrogate endpoint) are the 4 ways applied singly or in conjunction in which the FDA cooperates with the industry (see Table 1.1) in developing drugs (FDA, 2018; 2018a). This resulted in an increase of the speed at which new drugs are and have been developed and reviewed, and is expressed by the fact that 78% and 87% of drug applications were approved by the FDA in the first review cycle in 2014 and 2015, respectively, compared with just 58% between 2008 and 2012 (Tworkoski, 2016).
API | Drug Name | FDA-approved use | Expedited program | Company |
|---|---|---|---|---|
Elbasvir & grazoprevir | Zepatier | Chronic hepatitis C virus | BT | MSD SHARP & DOHME GMBH |
Brivaracetam | Briviact | Partial onset seizures | UCB (Union Chimique Belge) | |
Obiltoxaximab | Anthim | Inhalational anthrax | Elusys Therapeutics, Inc. | |
Ixekizumab | Taltz | Plaque psoriasis | Eli Lilly & Company | |
Reslizumab | Cinqair | Severe asthma | Teva Pharmaceuticals | |
Defibrotide sodium | Defitelio | Hepatic veno-occlusive disease | PR, OD | Jazz Pharmaceuticals |
Venetoclax | Venclexta | Chronic lymphocytic leukemia | AA, BT, PR, OD | AbbVie Inc. |
Pimavanserin | Nuplazid | Hallucinations & delusions | BT, PR | Acadia Pharmaceuticals Inc. |
Atezolizumab | Tecentriq | Urothelial carcinoma | AA, BT, PR | Genentech |
Daclizumab | Zinbryta | Multiple sclerosis | Biogen, Inc. | |
Obeticholic acid | Ocaliva | Chronic liver disease | AA, FT, OD | Intercept Pharmaceuticals, Inc. |
Fluciclovine F 18; imaging agent | Axumin | To detect recur-rent prostate cancer | Blue Earth Diagnostics, Ltd. | |
Ga 68 dotatate; imaging agent | NETSPOT | To detect neuroendocrine tumors | PR, OD | Advanced Accelerator Applications USA, Inc. |
Sofosbuvir & velpatasvir | Epclusa | All six major forms of hepatitis C virus | PR | Gilead Sciences, Inc. |
Lifitegrast ophthalmic solution | Xiidra | Dry eye disease | Shire US Inc. | |
Lixisenatide | Adlyxin | Improve glycemic control | Sanofi-Aventis U.S. LLC | |
Eteplirsen | Exondys 51 | Duchenne muscular dystrophy | AA, FT, PR, OD | Sarepta Therapeutics |
Olaratumab | Lartruvo | Soft tissue sarcoma | AA, BT, FT, PR, OD | Eli Lilly & Company |
Crisaborole | Eucrisa | Atopic dermatitis | Anacor Pharmaceuticals, Inc. | |
Bezlotoxumab | Zinplava | Decrease the risk of Clostridium difficile infection | Merck Sharp & Dohme Corp. | |
Rucaparib | Rubraca | Ovarian cancer | AA, BT, PR, OD | Foundation Medicine, Inc. |
Nusinersen | Spinraza | Spinal muscular atrophy | FT, PR, OD | Ionis Pharmaceuticals |
Note: AA, Accelerated approval; PR, Priority review status, BT, Breakthrough therapy designation; FT, Fast track designation; OD, Orphan drug designation; API, Active Pharmaceutical ingredient. | ||||
The understanding of cancer pathogenesis has significantly improved in the recent past which has led to new therapeutic concepts such as targeted agents designed to inhibit molecular pathways that are crucial for tumor growth, or immunotherapeutic approaches (Vanneman and Dranoff, 2012; Gotwals et al., 2017; Banchereau and Palucka, 2018) including immune checkpoint inhibitors (Dine et al., 2017). Another recent example is Yescarta, a chimeric antigen receptor (CAR) T cell therapy, approved as the second gene therapy by the FDA for treating diffuse large B-cell lymphoma (DLBCL, the most common type of non-Hodgkin lymphoma; FDA, 2018c); Yescarta has received the orphan drug status (see next section). This has led to an improved survival of patients suffering from certain types of cancer and finds expression, too, in a trend towards an increased share of oncologic drugs as part of the number of drugs approved per year as shown in the following figure (see also Centerwatch, 2018).
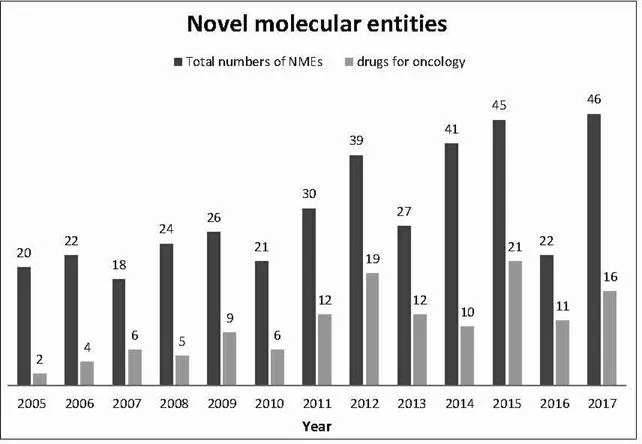
1.3 Orphan Drugs and Rare Diseases
According to the European Medicines Agency the number of rare/orphan diseases is between 5,000 and 8,000, and 27 million to 36 million patients in the EU are suffering from such diseases of which most (~80%) are of genetic origin. The NORD’s (National Organization for Rare Disorders, Inc.) database contained details for more than 1,200 rare diseases by 2018. The orphan drug status has been introduced in various...
Table of contents
- Cover
- Half Title
- Title Page
- Copyright Page
- Table of Contents
- Preface
- 1. Pharmaceuticals: Some General Aspects
- 2. Use of Enzymes in the Downstream Processing of Biopharmaceuticals
- 3. Therapeutic Use of Carbonic Anhydrase Inhibitors and Their Multiple Drug Interactions
- 4. Fibrinolytic Enzymes for Thrombolytic Therapy
- 5. Role of Engineered Proteins as Therapeutic Formulations
- 6. Synthesis of Bioactive Peptides for Pharmaceutical Applications
- 7. Cardiovascular Disease and Oxidative Stress
- 8. Enzymatic Amino Acid Deprivation Therapies Targeting Cancer
- 9. Carboxylesterase Inhibitors: Relevance for Pharmaceutical Applications 349
- 10. Molecular Aspects of the Activity and Inhibition of the FAD-Containing Monoamine Oxidases 397
- 11. Multi-Functional Monoamine Oxidase and Cholinesterase Inhibitors for the Treatment of Alzheimer’s Disease 427
- 12. The Neurodegenerative Characteristics of Alzheimer’s Disease and Related Multi-Target Drug Design Studies
- 13. Aldehyde Oxidases as Enzymes in Phase I Drug Metabolism
- 14. Cytochrome P450 Enzymes for the Synthesis of Novel and Known Drugs and Drug Metabolites
- 15. Cytochromes P450, Cardiovascular Homeostasis and Disease
- 16. Protein Degradation Inducers SNIPERs and Protacs against Oncogenic Proteins
- 17. Resistance Mechanisms of Tumor Cells
- 18. Biocatalytic Nanoreactors for Medical Purposes
- 19. Computer-Aided Drug Design for the Identification of Multi-Target Directed Ligands in Complex Diseases: An Overview
- 20. The Development of Improved Therapeutics through a Glycan-“Designer” Approach
- 21. On Biocatalysis as Resourceful Methodology for Complex Syntheses: Selective Catalysis, Cascades and Biosynthesis
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Pharmaceutical Biocatalysis by Peter Grunwald in PDF and/or ePUB format, as well as other popular books in Medicine & Biotechnology in Medicine. We have over 1.5 million books available in our catalogue for you to explore.