![]()
A corrosion protection method is a technique used to minimize corrosion, such as the application of an anticorrosion coating, cathodic protection, or other methods that make metal resistant to corrosion. Corrosion protection methods are also known as corrosion control.
Corrosion causes hundreds of billions of dollars in losses each year. Corrosion is a very common yet serious problem across different industries and environments. Corrosion can be increased by the presence of high temperatures, high relative humidity, salt air, harsh chemicals, and even by mold, moss, and dirt deposits. A 2016 study by the National Association of Corrosion Engineers (NACE) pegged the global cost of corrosion at $2.5 trillion. Corrosion is not simply a sustainment concern; it needs to be addressed from program/system/equipment inception to disposal. Corrosion prevention and control at the early stages in a pipeline system development can result in a reduction of total ownership cost.
Five primary techniques used for limiting corrosion rates to practical levels are:
• Material selection
• Coatings
• Cathodic protection
• Chemical injection
• Proper anticorrosion design
The content of this book is developed from broad and in-depth experience regarding the protection of pipeline systems/equipment from corrosion and its effects. The book is arranged in the following sections:
• A summary of the main causes of corrosion and the requirements for protection of materials against corrosion.
• A summary of the selection of corrosion-resistant materials.
• A summary of coating materials commonly used for corrosion protection and the limitations to their use, their application, and repair.
• Guidelines for the design of cathodic protection systems and reviews of cathodic protection methods, materials, installation, and monitoring.
• Galvanic zinc application.
• Chemical corrosion inhibitor.
• Case studies and design example.
• A reference section detailing source documents and support documents which may be used in conjunction with this book.
It should be noted that guidance in this book is applicable to most locations around the world. However, there may be local constraints on corrosion coating selection, for example, which are not immediately evident if the design is performed from a distance.
This book is concerned with the protection of steels against corrosion in an aqueous environment, either immersed in seawater or buried. For most oil and gas applications, the steels are of the carbon or low alloy types, although in some cases, it is necessary to protect stainless steels.
![]()
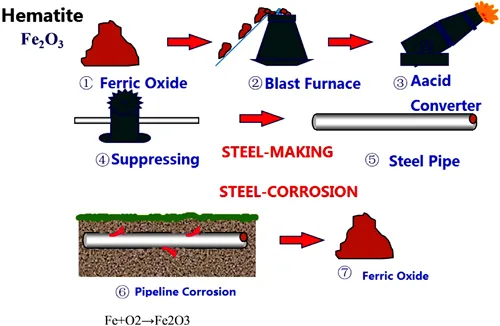
Corrosion is the deterioration of a material (usually metal) due to its interaction with the environment. Corrosion is a natural phenomenon, which should not surprise one, but rather should be expected to occur. Metals are high-energy materials which exist because heat energy was added to natural iron ores during the smelting process. Nature, by environmental contact, constantly attacks these high-energy materials and breaks them down to the natural elements from which they were derived.
The effect of corrosion on oil and gas pipelines can be catastrophic. The destruction of the metal eventually leads to leakages, which have the potential to cause massive disasters such as fires and explosions. High safety concerns and strict standards applicable to the oil and gas industry mean that constant monitoring is needed to identify the presence and extent of corrosion.
2.1 Basics of Aqueous Metallic Corrosion
A corroding system is driven by two spontaneous reactions that take place at the interface between the metal and an aqueous environment. The two simultaneous reactions are the oxidation (anodic) reaction and a reduction (or cathodic reaction). The first reaction is when the chemical species from the aqueous environment remove electrons from the metal; the other is a reaction in which metal surface atoms participate to replenish the electron deficiency.
Example of anodic (oxidation, electron donating) reaction:
| | |
Examples of cathodic (reduction, electron accepting) reactions:
| | |
An important effect is the electron exchange between the two reactions that constitutes an electronic current at the metal surface. This, in turn, imposes an electric potential on the metal surface of such a value that the supply and demand for electrons in the two coupled reactions are balanced.
The potential imposed on the metal is of much greater significance than simply to balance the complementary reactions which produce it. This is because it is one of the principal factors determining what the reactions will be.
Generally, at the anodic locations, there is corrosion damage (e.g., metal loss), while at the cathodic location, no corrosion damage occurs. The location of anodes and cathodes tends to move randomly over the surface of the metal for alloys that are subject to general corrosion. However, the location of an anode tends to become strongly localized for corrosion-resistant alloys, which are covered by a passive oxide film. This gives rise to localized corrosion damage such as pitting corrosion, stress corrosion cracking, and crevice corrosion (Figures 2.1–2.2).
FIGURE 2.1 Basics of corrosion. (Source: Tong, Shan. 2015. Cathodic protection. Training document, Ghana: Sinopec.)
2.2 Forms of Corros...