
- 432 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Handbook of Atmospheric Electrodynamics, Volume I
About this book
The participation of such diverse scientific and technical disciplines as meteorology, astronomy, atmospheric electricity, ionospheric and magnetospheric physics, electromagnetic wave propagation, and radio techniques in the research of atmospherics means that results are published in scientific papers widely spread throughout the literature. This Handbook collects the latest knowledge on atmospherics and presents it in two volumes. Each chapter is written by an expert in his or her field. Topics include the physics of thunderclouds, thunder, global atmospheric electric currents, biological aspects of sferics, and various space techniques for detecting lightning within our own atmosphere as well as in the atmospheres of other planets. Up-to-date applications and methodology are detailed. Volumes I and II offer a comprehensive discussion that together will serve as an important resource for practitioners, professionals, and students alike.
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
NaturwissenschaftenSubtopic
Kondensierte MaterieChapter 1
Ion Chemistry and Composition of the Atmosphere
CONTENTS
1. Introduction
2. Ionization Sources and Sinks
2.1. Sources
2.2. Sinks
3. Instrumentation
4. Ion Composition of the Atmosphere
4.1. Positive Ions
4.2. Negative Ions
5. Ionic Processes and Evolution
5.1. Ionic Processes
5.2. Positive Ions
5.3. Negative Ions
6. Trace Neutral Derivations from Ion Composition Measurements
References
1. INTRODUCTION
Atmospheric ions not only are important in controlling atmospheric electrical properties, but also play a role in aerosol processes and can be used to measure trace neutral concentrations very sensitively. Figure 1.1.1 shows the ion concentration of the atmosphere as a function of altitude (Arnold, 1980). The ion density varies between about 103 and 106 ions cm–3. The high densities are at high altitudes where the gas density is low. At altitudes below 100 km, the ionization density is relatively constant, varying by less than a factor of 10 around 103 ions cm−3. The neutral gas density changes by a factor of approximately 4 × 106 in this altitude range. The mixing ratio of ions to neutrals therefore decreases substantially from high altitude to low altitude. At ground level, there is only one ion for approximately 1016 neutral molecules.
Mass spectrometrie measurements of the ion composition are more difficult at lower altitudes due to the small relative concentration of ions and large neutral density. As a result, the ion composition of the upper atmosphere was measured before that of the lower atmosphere, even though the difficulty in using the various in situ measuring platforms increases with height. The various platforms are trailer, aircraft, balloon, sounding rocket, and satellite in order of increasing height range. The last portion of the atmosphere where in situ measurements have been made is the troposphere.
The purpose of this chapter is to give a survey of the various ionic processes that occur in the atmosphere. Emphasis will focus on the main processes, and the interested reader is referred to numerous sources for the details. The field of atmospheric chemistry has previously been the subject of numerous reviews (Reid, 1976; Arnold and Krankowsky, 1977; Ferguson et al., 1979; Arnold, 1980; Smith and Adams, 1980; Ferguson and Arnold, 1981; Arnold, 1982; Thomas, 1983; Arnold and Viggiano, 1986; Brasseur and De Baets, 1986; Brasseur and Solomon, 1986; Reid, 1989).
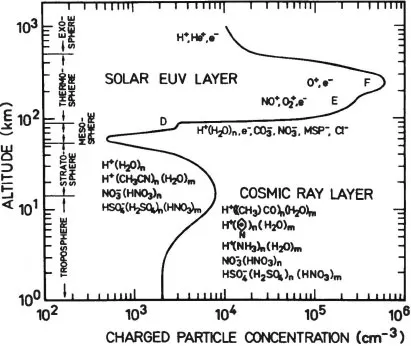
Figure 1.1.1 Typical ionization density profile of the atmosphere as a function of altitude. For each height regime, the major ions species are listed.
This chapter starts by discussing how charged particles are produced and lost in the atmosphere. A short discussion follows on how in situ measurements are made. Results of the in situ measurements are then presented. Instead of showing the results of individual measurements, a composite of the measurements is assembled where the main families of ions can be identified without distraction from minor species. A discussion of the ion chemistry of atmospheric ions follows. First, the general processes are discussed followed by details of the positive and negative ion chemistry. Emphasis is always placed on the main processes involved. The chapter ends with a discussion of how ion composition measurements are used to derive concentrations of atmospheric trace neutrals. This is an appropriate ending place because many of the recent advances in in situ ion composition measurements have been driven by the need for trace neutral detection.
2. IONIZATION SOURCES AND SINKS
2.1. SOURCES
There are numerous ionization sources in the atmosphere. The most abundant gases, N2 and O2, have ionization energies of 15.58 and 12.07 eV, respectively (Lias et al., 1988). Therefore, only energetic particles or electromagnetic radiation of very short wavelength are generally capable of producing ionization.
The simplest region of the atmosphere to understand is that below 60 to 70 km. Under normal circumstances, i.e., a quiet ionosphere, galactic cosmic rays are essentially the only source of ionization, except just above the earth’s surface. Figure 1.2.1 gives estimates of the ion pair production rate below 60 km (Thomas, 1974). Two curves are given for the cosmic ray production rate, one for solar maximum and one for solar minimum. There is also a latitude dependence because cosmic rays, which consist mainly of extraterrestrial protons and α particles, are affected by magnetic fields and therefore have better access into the earth’s poles. Because cosmic rays are very energetic, they ionize all gases with approximately the same efficiency. Thus, for practical purposes, one can assume that only the most abundant gases are ionized. The energetic nature of the particles means that not only and are produced, but also N+ and O+. Above 15 km, the cosmic ray flux is practically the same as it is at the top of the atmosphere and the ion production rate is proportional to the atmospheric number density. Below this height, the cosmic rays have been significantly absorbed by the atmosphere and the production rate decreases. The total variation in cosmic ray intensity is about a factor of 10. The variability depends mainly on the sunspot cycle and geomagnetic latitude.
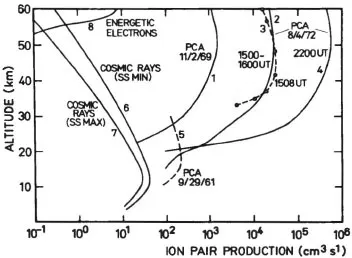
Figure 1.2.1 Ion production rate atmosphere as a function of altitude below 60 km. (After Thomas, L. [1974], Radio Sci., 9,121. Copyright 1974 by the American Geophysical Union. With permission.)
In the troposphere, two other ionization sources are important. At and near the earth’s surface, ionization from energetic particles occurs due to high energy particles from radioactive decay. The main source is radon. This creates a slight increase in the ion concentration confined to a boundary layer of approximately a kilometer. In addition, lightning will create local ionization.
Although cosmic rays are the main source of ionization for much of the time in the lower atmosphere, additional ionization is occasionally created by energetic particles reaching the lower atmosphere during tim...
Table of contents
- Cover
- Title Page
- Copyright Page
- Table of Contents
- 1 Ion Chemistry and Composition of the Atmosphere
- 2 Meteorological Aspects of Thunderstorms
- 3 Thunderstorm Electrification
- 4 Lightning Currents
- 5 Lightning Detection from Ground and Space
- 6 Artificially Triggered Lightning
- 7 Ball Lightning
- 8 Lightning and Atmospheric Chemistry: The Rate of Atmospheric NO Production
- 9 Lightning within Planetary Atmospheres
- 10 Quasistatic Electromagnetic Phenomena in the Atmosphere and Ionosphere
- 11 Schumann Resonances
- 12 Low-Frequency Radio Noise
- 13 Radio Noise Above 300 kHz Due to Natural Causes
- 14 Atmospheric Noise and Its Effects on Telecommunication System Performance
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Handbook of Atmospheric Electrodynamics, Volume I by Hans Volland in PDF and/or ePUB format, as well as other popular books in Naturwissenschaften & Kondensierte Materie. We have over 1.5 million books available in our catalogue for you to explore.