
- 141 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Food Engineering Laboratory Manual
About this book
FROM THE PREFACE
The purpose of this laboratory manual is to facilitate the understanding of the most relevant unit operations in food engineering. The first chapter presents information on how to approach laboratory experiments; topics covered include safety, preparing for a laboratory exercise, effectively performing an experiment, properly documenting data, and preparation of laboratory reports. The following eleven chapters cover unit operations centered on food applications: dehydration . . . . , thermal processing, friction losses in pipes, freezing, extrusion, evaporation, and physical separations. These chapters are systematically organized to include the most relevant theoretical background pertaining to each unit operation, the objectives of the laboratory exercise, materials and methods . . ., expected results, examples, questions, and references. The experiments presented have been designed for use with generic equipment to facilitate the adoption of this manual . . . .
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Subtopic
Science History & PracticeCHAPTER 1
Planning Experiments
1.1 INTRODUCTION
The principles governing the engineering aspects of food processing are the same as those applied in any engineering field in that engineers are educated to analyze, synthesize, design, and operate complex systems that manipulate mass, energy, and information to transform materials and energy into useful forms, which, in this case, are food products or food ingredients. This book is designed to give food engineering and/or food science students an understanding of the engineering principles and hands-on experiences involved in the processing of food products. With a clear understanding of the engineering basic principles of food processing, it is possible to develop new food processes and modify existing ones. Because an essential component of any laboratory exercise is to receive proper laboratory orientation, follow safety guidelines, and prepare laboratory reports, this chapter deals with fundamental engineering aspects related to specific laboratory exercises, how to get ready for an experiment, and how to report it.
1.2 MASS BALANCE
The law of conservation of mass states that mass cannot be created nor destroyed, so a mass balance in any process can be written as follows:
In a continuous process at steady state, the accumulation is zero. Therefore, a simple rule that “what goes in must come out” holds. For example, in the concentration process of milk, whole milk is fed into an evaporator. Under the law of conservation of mass, the total number of pounds of material (whole milk) entering the evaporator per unit time must equal the total number of pounds of concentrated milk and evaporated moisture that leave the evaporator. When solving the mass balance, four key steps must be followed:
(1) Select a system and draw a diagram representing the process (including all pertinent information on stream rate and compositions).
(2) Select an appropriate basis for calculation.
(3) Write the mass balance relationships for the various constituents in terms of the known and unknown quantities.
(4) Solve the resulting algebraic equations for the unknown quantities.
1.2.1 Example
A milk concentrate is to be made by evaporating water from whole milk. The whole milk contains 13% total solids (TS), and the concentrate should contain 49% TS. Calculate the amount of product and the water that needs to be evaporated.
1.2.1.1 SOLUTION
• Step 1: Select the evaporator to be a target system, and draw a diagram representing the process as follows:
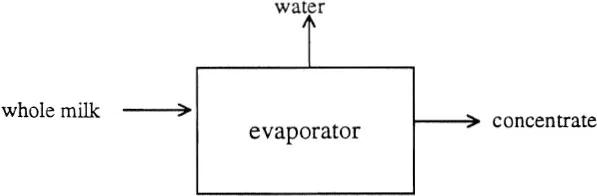
• Step 2: Select a basis for the calculation of 100 kg of incoming whole milk.
• Step 3: Write up the equations for the mass balance:
(1) The balance for the total material:
(1.1) |
(2) The balance of total solids (TS):
(1.2) |
• Step 4: Solve the equations. Note that Equation (1.2) has only one unknown and should therefore be solved first. The result is:
Substituting the C value into Equation (1.1), we get:
• Step 5: Appropriate answer: From every 100 kg of whole milk, we can manufacture 26.5 kg of the concentrated milk, and thus 73.5 kg of water must be evaporated.
1.3 ENERGY BALAN...
Table of contents
- Cover
- Half Title
- Title Page
- Copyright Page
- Table of Contents
- Preface
- Acknowledgements
- Chapter 1. Planning Experiments
- Chapter 2. Friction Losses Determination in a Pipe
- Chapter 3. Convective Heat Transfer Coefficient Determination
- Chapter 4. Thermal Processing of Foods: Part I. Heat Penetration
- Chapter 5. Thermal Processing of Foods: Part II. Lethality Determination
- Chapter 6. Freezing of Foods
- Chapter 7. Drying of Foods: Part I. Tray Drying
- Chapter 8. Drying of Foods: Part II. Spray Drying
- Chapter 9. Drying of Foods: Part III. Freeze Drying
- Chapter 10. Extrusion of Foods
- Chapter 11. Evaporation
- Chapter 12. Physical Separations
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Food Engineering Laboratory Manual by Gustavo V. Barbosa-Canovas,Li Ma,Blas J. Barletta in PDF and/or ePUB format, as well as other popular books in Technology & Engineering & Science History & Practice. We have over 1.5 million books available in our catalogue for you to explore.