Solids—Anything that holds together on its own to form a particular mass or shape. Bowling balls, ice, and skin are all considered solids. Solids always retain the same volume, and don’t change shape unless a force is applied to them.
Gases—A substance that expands and spreads out to take the shape of its container and fill it up as much as possible, and changes volume as the container changes volume. Examples of gases are steam, oxygen, air, and propane gas.
Composition of Matter
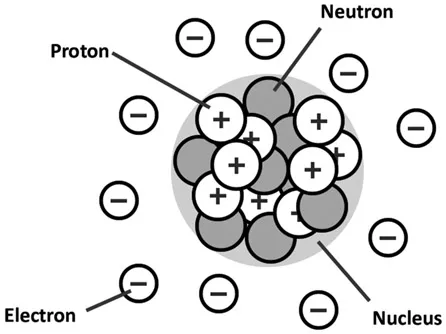
Physics tells us that all matter, whether solid, liquid, or gas, is made up of basic units called atoms. An atom is, theoretically, a tiny blob of matter surrounded by other blobs of matter that float around it. All the blobs have a sort of electrical charge associated with them that binds them together and keeps them from flying apart.
Because atoms are so tiny, we can’t see them the way we normally see objects, where light reflects offthe object and into our eyes. Only recently have we found ways to “see” atoms by using electric current to detect their movement patterns.
To start off understanding atoms, you can think of an atom as a balloon with foam packing peanuts stuck to it. If you rub a balloon on your clothes, you cause a little bit of electrical charge to form on the balloon. If you move the balloon near some packing peanuts, the peanuts will lightly stick to the balloon due to electrical attraction.
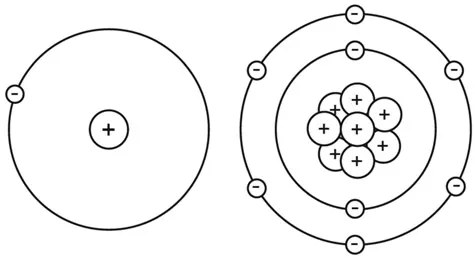
Each basic element on Earth has a unique blob configuration. Hydrogen, for example, has just one inner and outer blob. Oxygen has eight inner and outer blobs.
Configuration of hydrogen (left) and oxygen (right).
No one really understands why electrical attraction works the way it does. We only know that it works. While the quantum physicists are hard at work trying to figure this out, no one knows for sure. But electrical attraction explains a bunch of other stuffthat scientists study, so they roll with it.
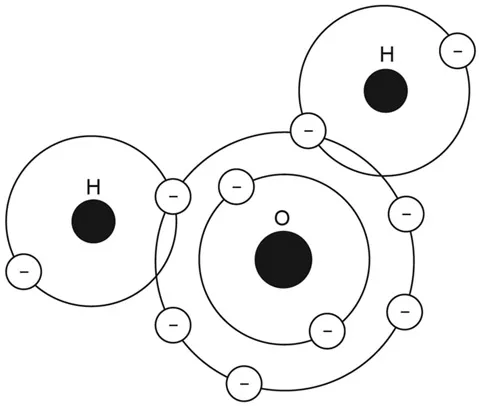
All substances are made up of different combinations of atoms. When the atoms get close together they share their outer blobs, which binds them together. For example, if you throw together one oxygen atom and two hydrogen atoms, you get pure liquid water, or H2O.
H2 means two hydrogen atoms, single O means one oxygen.
When two or more atoms join together in a stable state, this is called a molecule. Two hydrogen atoms combine with an oxygen atom to form a water molecule.
Now for some terminology. In an atom, the central blob is called a nucleus. The blobs floating around it are negatively charged electrons. The nucleus contains of a bunch of positively charged blobs called protons, which attract the negatively charged electrons. The nucleus also contains neutrons, which have no charge at all, but which work as a sort of atomic glue to keep the protons stuck to the nucleus.
It’s not vital to memorize these terms, as long as you have an overall understanding of this most basic level of matter (and you know which page in this book to come back to should you need to refresh your memory). Electrons in particular are at the root of a lot of visually interesting phenomena like lightning and electricity. In this book, we go over electrons’ roles in more detail in pertinent chapters.
Atoms and Binding
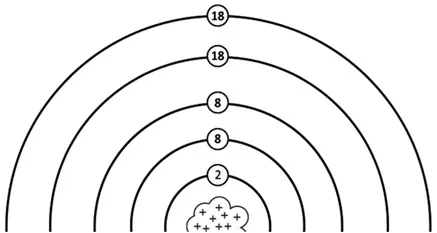
Atoms like to have their electrons arranged a certain way. In the innermost ring around the nucleus, they like to have two electrons. More electrons after that are arranged in another ring or shell outside the first, with at most eight electrons. The third level has at most eight electrons. Any levels outside that like to have at most 18 electrons.
Most atoms are neutral, meaning they have the same number of protons as electrons. The number of neutrons can vary in neutral atoms, and is not always the same as the number of protons.
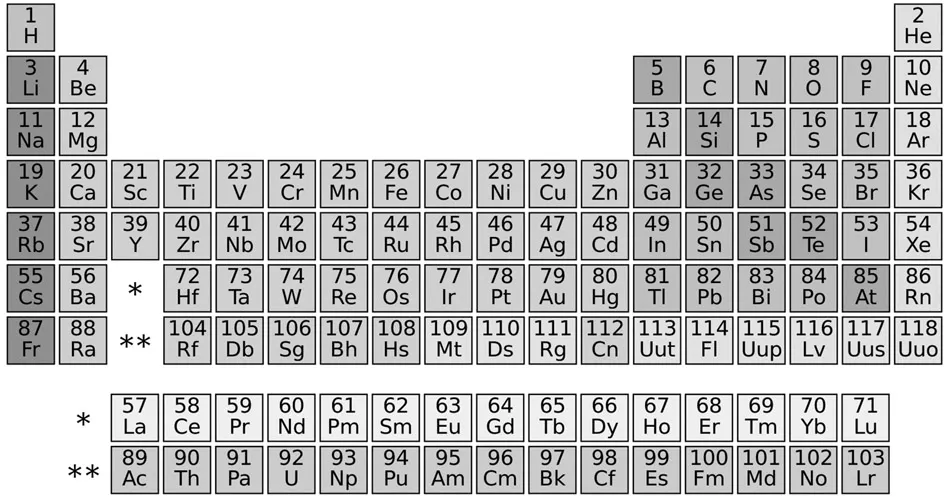
The Periodic Table of Elements organizes the elements in our universe according to how many protons each one has, and how many shells it has.
Periodic Table of Elements.
If an atom’s outermost shell isn’t filled with the number of electrons it likes to have, it likes to find other atoms a...