
- 176 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
About this book
Complete, referenced information in an easy-to-use formatMany of the monographs in the European Pharmacopiea, the industry standard test for certain groups of ingredients and excipients, do not describe the tests in full, but reference general methods based on test-tube chemistry. When a test fails, you need to know what went wrong, how it can be f
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Part I
Identifications
chapter one
Precipitation in identifications
In qualitative tests based on selective precipitation, the identity of the substance in question is verified by ensuring that it gives a precipitate under the defined conditions. In many cases it is a more or less explicit demand of the test that the precipitate have a defined appearance, such as for example a dense gelatinous or crystalline appearance. Whether a substance gives a precipitate in the conditions the tests specify depends on its solubility under these exact conditions and also on the kinetics of the given precipitation. The appearance of a precipitate, apart from the color, is largely determined by its particle size distribution and by its affinity toward the water molecules and ions of the surrounding solution.
Precipitate selectivity
A substance precipitates from a given solution when its solubility is exceeded. In the case of ionic substances, this means that the factor Q, which is the product of the molar concentrations of the salt forming ions, is higher than its solubility product. The solubility product is a constant defined as the product of the molar concentrations of the individual salt forming ions, in a saturated solution of the salt in question. The solubility product formula for the general salt AaBb is
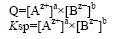
In many cases, the negative logarithm to the solubility product is used to make the vast span of solubility products encountered easier to manage.
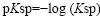
The important thing to have in mind is that precipitation is facilitated whenever the solubility product is exceeded, also in the cases where the ratios of individual ion concentrations are very different from the ones found in a saturated solution of the salt in question. This means, when referring to the general formula, that precipitation will take place even in cases of a very low A concentration compared to the ones seen in a saturated solution, if only B concentration is high enough. This is interesting especially in salts where protons (H+) or hydroxide (OH-) participates, since test solution pH then influences Q directly. A deviation in test solution pH of 1 or 2 units perhaps does not seem very much in a simple test such as an identification, but the pH scale being logarithmic means that [H+] or [OH-] is actually 10 or 100 times from the desired value. Depending on the stoichiometry of the salt in question, this has a large effect on Q.
It also means that if ions that participate in a given salt are added to the solution from unintended sources, this will lower the solubility of the salt by raising the test solution ion product. This is the so-called common ion effect.
It should also be noticed that the stoichiometric coefficients end up as exponents in the formula for the solubility product. The ion product Q is therefore more sensitive toward changes in concentrations of ions present in the salt formula in a stoichiometric ratio higher than 1.
Finally, it should be remembered that the solubility product in the form presented above is a simplification of the real formula, which uses ion activity a instead of ion concentration:
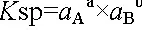
These are obtained by multiplying the molar concentration by activity coefficients. The activity coefficients are symbolized by γ:

The value of γ is given by the Debye-Huckel equation that incorporates, among other parameters, both the ionic strength of the solution and the valence of the ion. It is a number between 0 and 1, where 1 applies for very dilute solutions. The effect of a high ionic strength in a solution is that γ declines and deviates from 1. When multiplied by the concentration it gives a lower activity and ultimately a lower Q than would have been the case if molar concentration had been used, so that the concentration needed to reach the solubility product is higher.
This means that salts will display a higher solubility in a solution of high ionic strength than predicted by experiments in weaker ionic solutions or pure water. All the dissolved ionic compounds contribute to the solution’s ionic strength, especially the ones of a higher valence than 1. So the unintended presence of ionic substances brought there as sample preparation residues or by simple contamination might prevent a precipitation or disturb the basis of a selective precipitation.
The kinetics of a precipitation is dependent on the actual salt in question and on the degree of supersaturation in the solution. This means that even though the ion product might not have been lowered by ionic strength to an extent that precipitation is eliminated, it might have lowered the degree of supersaturation sufficiently to slow down the onset of precipitation. It again might alter the appearance of the precipitate.
The exact way a selective precipitation is performed has a strong influence on the selectivity obtained. One could, for example, identify a cation forming amphoteric hydroxide by first precipitating it with sodium hydroxide and then redissolving it in excess sodium hydroxide. In principle, this procedure can be designed in two different ways. One could slowly add sodium hydroxide until precipitation occurs and then further add sodium hydroxide until the precipitate redissolves. Or one could first add a defined amount of sodium hydroxide that knowingly should precipitate the actual cation and then further add a defined amount of sodium hydroxide that knowingly should redissolve the actual cation in question.
In terms of judging the ruggedness and selectivity of the test, these two principles are quite different. In the first case a high degree of ruggedness is achieved. Even if some contaminants in the test solution, or the counter-ion in the substance to be examined, delay precipitation, eventually, it will take place as further sodium hydroxide is added. This situation could entirely prevent precipitation from occurring as a consequence of adding the defined amount of sodium hydroxide dictated in the second case, thereby giving a false positive reaction. The selectivity, on the contrary, is more limited in the first procedure. All cation-forming amphorteric cations will give a positive reaction, regardless of the amount of sodium hydroxide needed to precipitate and redissolve the various cations.
In general, when discriminating inorganic ions by their solubility as different salts and in different chemical environments, one should recognize that seemingly small variations in test conditions can change the selectivity and robustness of the test. Variations in sample concentration, pH, and ionic strength of the test solution and presence of a counterion can alter the situation from what is intended.
Precipitate appearance
Precipitates have often been placed in one of three different categories based on their macroscopic appearance. A precipitate can be colloidal, crystalline, or curdy gelatinous. The kind of precipitate given by a specific salt has no correlation to its degree of insolubility but is determined by the particle size distribution of the precipitate, and its affinity toward the water molecules and ions of the surrounding solutio...
Table of contents
- Cover Page
- Title Page
- Copyright Page
- Preface
- Acknowledgment
- About the Author
- Part I: Identifications
- Part II: Limit Tests
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Pharmaceutical Chemical Analysis by Ole Pedersen in PDF and/or ePUB format, as well as other popular books in Technology & Engineering & Pharmacology. We have over 1.5 million books available in our catalogue for you to explore.