
eBook - ePub
Fundamentals of Aluminium Metallurgy
Production, Processing and Applications
- 864 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
About this book
Aluminium is an important metal in manufacturing, due to its versatile properties and the many applications of both the processed metal and its alloys in different industries. Fundamentals of aluminium metallurgy provides a comprehensive overview of the production, properties and processing of aluminium, and its applications in manufacturing industries.Part one discusses different methods of producing and casting aluminium, covering areas such as casting of alloys, quality issues and specific production methods such as high-pressure diecasting. The metallurgical properties of aluminium and its alloys are reviewed in Part two, with chapters on such topics as hardening, precipitation processes and solute partitioning and clustering, as well as properties such as fracture resistance. Finally, Part three includes chapters on joining, laser sintering and other methods of processing aluminium, and its applications in particular areas of industry such as aerospace.With its distinguished editor and team of expert contributors, Fundamentals of aluminium metallurgy is a standard reference for researchers in metallurgy, as well as all those involved in the manufacture and use of aluminium products.
- Provides a comprehensive overview of the production, properties and processing of aluminium, and its applications in manufacturing industries
- Considers many issues of central importance in aluminium production and utilization considering quality issues and design for fatigue growth resistance
- Metallurgical properties of aluminium and its alloys are further explored with particular reference to work hardening and applications of industrial alloys
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
1
Introduction to aluminium metallurgy
R.N. Lumley, CSIRO, Light Metals Flagship, Australia
Abstract:
This chapter provides the introduction to the book and gives a brief overview of the history and growth of the global aluminium industry up to the present day, including some of the significant achievements, breakthroughs and challenges. It also discusses the future avenues of growth and where the global industry may be headed over the next 20 years.
Key words
aluminium industry
aluminium alloys
primary aluminium
secondary aluminium
1.1 Aluminium as an engineering material
Aluminium is the third most abundant element in the earth’s crust, and the most abundant metallic element. For the last 50 years, it has been second only to iron in its industrial use. Aluminium does not occur in an elemental state; rather it is always combined in a chemical compound. Its existence was not recognized until 1808, when an impure form was identified by Sir Humphry Davy in Britain. In 1825, Hans Christian Oersted of Denmark produced minute quantities of the metal,and two years later Freidrich Wöhler of Germany described a process for producing aluminium as a powder by reacting potassium with anhydrous aluminium chloride. In 1845, he determined its specific gravity thereby establishing it as a low-density metal.Thefirst commercial process for producing aluminium was developed by Henri Sainte-Claire Deville in 1854, using an improved version of Wöhler’s process. The metal was, however, still very expensive, and during the next 30–40 years numerous attempts were made to develop more economic processes whereby costs could be reduced. Developments during this period are described in an interesting book by Aldophe Minet (1902, 1905) published first in German in 1902 and then English in 1905.
The development of the present electrolytic process occurred almost simultaneously in 1886 by Charles Martin Hall in the United States and Paul L.T Héroult of France (i.e. the Hall–Héroult process). This represented a major advance, since it opened the way for the economical production of aluminium, which began in about 1890. One essential stage in the process was the extraction of alumina (Al2O3) from the ore bauxite, and an efficient method to do this was developed by Karl Joseph Bayer in 1888. A further essential factor was the availability since the early 1870 s of generators capable of supplying electricity on the large scales required for electrolysis. To this day, the Bayer and the Hall–Héroult processes have remained the most economical methods for the production of commercial quantities of aluminium, and are the mainstay of the primary aluminium industry.
1.2 The development of aluminium alloys
It is interesting to note that the potential for aluminium alloys as engineering materials was recognized well before it became an industrial metal. In early 1886, it had been stated that: ‘It has been generally held that the most useful field for this metal would be found in its alloys, many of which are possessed of valuable properties; but we incline to the opinion that the metal itself, should it become possible to produce it cheaply enough to enable it to compete in price with tin or copper for example, would find innumerable applications’ (The Manufacturer and Builder, 1886). Due to its very high cost, the early applications of aluminium were often limited to small- or high-value items made of alloys such as Al-Ag which were used in the manufacture of instruments for marine observations (such as sextants) and balance beams where light weight was highly advantageous. Various other high-value domestic items were also produced such as tablespoons, which were reported in 1879 to cost (US) $20/dozen or close to a month’s wages for a labourer of the time (The Manufacturer and Builder, 1879).
Well before the Hall–Héroult process was known publicly, the compositions of several engineering alloys had been investigated, including those of aluminium with varying contents of Fe, Cu, Zn, Sn, Au and Ag (e.g. The Manufacturer and Builder, 1877). As early as the 1870 s, alloys containing between five per cent and ten per cent Cu were reported as being suitable for castings as well as for working into sheet, wire and other wrought products (e.g. Lange, 1873). One particular composition within this range, which was known as alloy 12 (Al-8Cu), was used extensively at least until the late 1930s for castings in automotive, consumer and aerospace applications, including the engine of the Wright Flyer 1 in 1903 (for a photograph of Wright engine #17 from 1910, see Armistej, 2006). By the time the book by Minet (1902, 1905) was published, the mechanical properties of a range of aluminium alloys and processes employed for working them had been examined in greater detail. Even at this early stage, their potential use for castings and wrought body structures for the transport industry had been recognized. For example, the Paris exposition of 1900 featured a wide range of aluminium alloy castings including a number produced for the then fledgling automotive industry (Minet, 1902, 1905). Wrought sheet of an alloy Al-3Cu-0.8Sn, known as ‘partinium’ (Minet, 1902, 1905), had also been used extensively in the electric vehicle named ‘La Jamais Contente’ built by Rothschild Coach Builders. Driven by Camille Jenatzy of Belgium, this vehicle attained the first world speed record greater than 100 km/h (105.9 km/h) in 1899 (e.g. Furet, 2008).
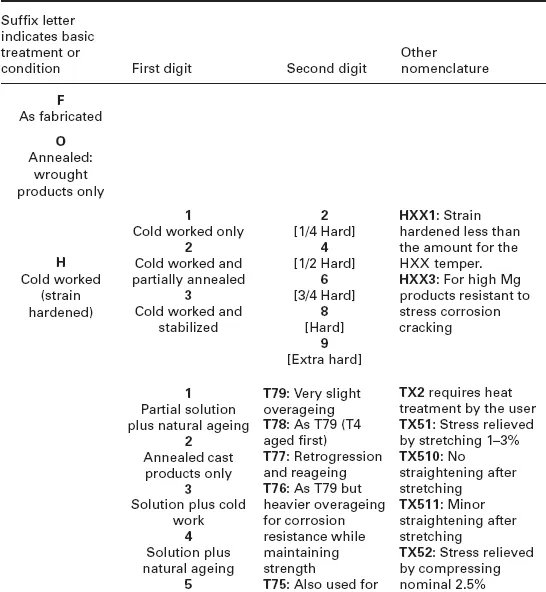
In 1906, four years after the publication of Minet’s book, Alfred Wilm of Germany discovered the basic procedure by which some aluminium alloys (i.e. the Al-3.5Cu-0.5 Mg-0.5Mn alloy that became known as Duralumin) could be heat treated. These techniques which involved precipitation (age) hardening are still used today, for strengthening a wide range of automotive and aerospace components (Cahn, 1984; Wilm, 1911). The basic technique developed by Wilm (a high-temperature solution treatment followed by quenching) was similar to that used previously for aluminium bronzes (Cu with 5–10% Al), as a way of softening the alloy so that it was easier to work at ambient temperature (The Manufacturer and Builder, 1881). The discovery of age hardening by Wilm is reported to be an extension of earlier patents related to the heating and quenching of aluminium (Cahn, 1984). Duralumin was instrumental in the development of the airship and aircraft industries and related Al-Cu-Mg alloys are still produced today. Examples of current wrought and cast aluminium alloys, and whether or not they are heat treatable, are shown in Table 1.1. Similarly, a wide array of procedures for heat treatment and thermomechanical processing has been developed, which are summarized in Table 1.2.
Table 1.1
The Aluminum Association alloy designation system*
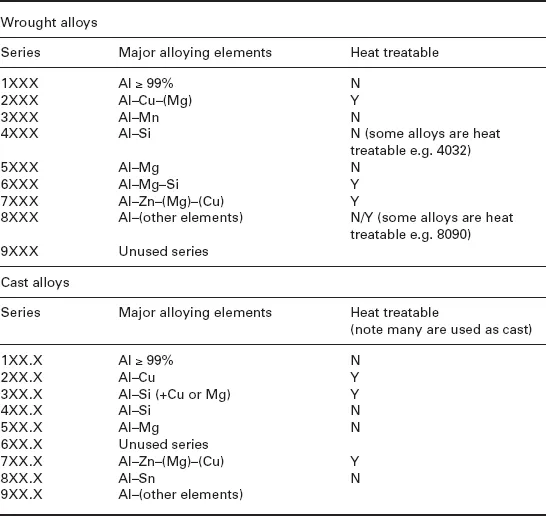
*While the Aluminum Association wrought alloy designations are very widely used, there are no universal casting alloy designations in wide use. However, for any casting alloy, there are usually equivalent alloy designations from each different region.
Table 1.2
Registered temper designations for aluminium alloys

1.3 Cast aluminium alloys
For many years, the casting of aluminium presented problems. Although it was relatively easy to produce useful shapes, it was rare for the castings to be free of visual porosity or other defects. By 1892, it had been recognized that: ‘It requires some experience and expertness on the part of the founder to master the...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- Contributor contact details
- Chapter 1: Introduction to aluminium metallurgy
- Part I: Production and casting of aluminium and its alloys
- Part II: Metallurgical properties of aluminium and its alloys
- Part III: Processing and applications of aluminium and its alloys
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Fundamentals of Aluminium Metallurgy by Roger Lumley in PDF and/or ePUB format, as well as other popular books in Technology & Engineering & Mining Engineering. We have over 1.5 million books available in our catalogue for you to explore.