
eBook - ePub
An Introduction to Pharmaceutical Sciences
Production, Chemistry, Techniques and Technology
- 446 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
An Introduction to Pharmaceutical Sciences
Production, Chemistry, Techniques and Technology
About this book
This textbook is written as a unified approach to various topics, ranging from drug discovery to manufacturing, techniques and technology, regulation and marketing. The key theme of the book is pharmaceuticals - what every student of pharmaceutical sciences should know: from the active pharmaceutical ingredients to the preparation of various dosage forms along with the relevant chemistry, this book makes pharmaceuticals relevant to undergraduate students of pharmacy and pharmaceutical sciences.This book explains how a particular drug was discovered and then converted from lab-scale to manufacturing scale, to the market. It explains the motivation for drug discovery, the reaction chemistry involved, experimental difficulties, various dosage forms and the reasoning behind them, mechanism of action, quality assurance and role of regulatory agencies. After having a course based on this book, the student will be able to understand: 1) the career prospects in the pharmaceutical industry, 2) the need for interdisciplinary teamwork in science, 3) the techniques and technology involved in making pharmaceuticals starting from bulk drugs, and 4) different dosage forms and critical factors in the development of pharmaceutical formulations in relation to the principles of chemistry.A few blockbuster drugs including atorvastatin, sildanefil, ranitidine, ciprofloxacin, amoxicillin, and the longest serving drugs such as aspirin and paracetamol are discussed in detail. Finally, the book also covers the important current pharmaceutical issues like quality control, safety, counterfeiting and abuse of drugs, and future prospects for pharmaceutical industry.
- Unified approach explaining drug discovery, bulk drug manufacturing, formulation of dosage forms, with pharmacological and therapeutic actions
- Manufacturing processes of representative active pharmaceutical ingredients and their chemistry plus formulation of dosage forms presented in this book are based on actual industrial processes
- Covers many aspects relevant to students of the pharmaceutical sciences or newly employed pharmaceutical researchers/employees. It contains summary information about regulatory agencies of different countries
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
MedicineSubtopic
Pharmacology1
Introduction
Learning objective
This introductory chapter provides students with a description of pharmaceutical science and its major components: pharmaceuticals and the pharmaceutical industry. There is a discussion of the gradual development of the industry, its multidisciplinary nature, its economics and technology, its impact on global health, and its ethical nature. Some pharmaceutical industries and their contributions to human life expectancy around the globe are briefly presented.
The mission of pharmaceutical science is to improve global health by discovering, developing, and producing quality medicines that are safe, appropriate, effective, affordable, and cost-effective, and widely distributing them around the world (Figure 1.1).
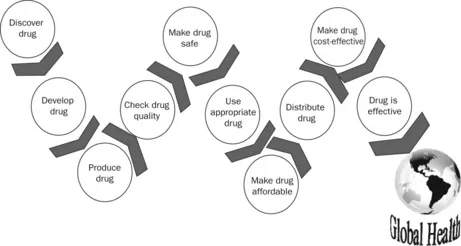
Figure 1.1 The mission of pharmaceutical science
Key concept terms
Absorption
movement of a drug from the site of entry into the blood circulation system
ACE inhibitor
angiotensin-converting enzyme
Administration
how a drug is taken
AIDS
Acquired Immune Deficiency Syndrome
API
active pharmaceutical ingredient
Apothecary
a pharmacist
Biopharmaceutical
biotechnology-based drugs, biologics
Blockbuster drug
a drug that has made more than US$1 billion/year
Bulk drug
API in bulk
Chronotherapy
administering a drug to work in coordination with a body’s biological clock
Distribution
the circulation of a drug in the body once absorbed
Dosage form
the physical form of a drug, such as tablet, capsule, ointment, and liquid injection
Elimination
removal of a drug from the body
Ethics
concepts of correct conduct
Excipient
non-active pharmaceutical ingredient
Formulated drug
dosage form of drugs made from APIs mixed with excipients
GMP
good manufacturing practice
HIV
Human Immunodeficiency Virus
IMS Health
Intercontinental Marketing Services, an information company
Injectable
intravenous or intramascular shot
Longevity
how long a person lives
Orange Guide
a book on GMP guidelines published by the Medicines and Healthcare Products Regulatory Agency
Parenteral
injectable
Pharmaceutical
synthetic chemical used as drugs
US FDA
US Food and Drug Administration
Pharmaceutical science is a multidisciplinary science combining many areas of the basic and applied sciences, such as chemistry, biochemistry, biology, mathematics, statistics, physics, medical sciences, and engineering.
1.1 The theme of the book
This book has been developed as a unified approach to topics ranging from bulk drugs to formulated medicines based on a course offered by the author as a special topic within the physical sciences at the Mississippi University for Women. The theme of the book is similar to the course theme: to explain what students should know about pharmaceuticals.
Staying in good health is important to most people, but many people become ill or have injuries at some time in their lives. Therefore, pharmaceuticals have a role in curing people or helping them return to good health. The importance of pharmaceuticals to the US population’s longevity cannot be understated.
Several books have been written on pharmaceutical topics, including pharmaceutical production, medicinal chemistry, drug design, dosage forms, pharmaceutical technology, quality assurance and control of pharmaceuticals, pharmaceutical research, and pharmaceutical marketing. These books are all specialized and very useful. This book is a little different as it is a biography of medicine. Beginning with the discovery of pharmaceutical applications, continuing through the manufacture of active pharmaceutical ingredients (APIs) and the preparation of various dosage forms, and finally marketing to patients, it explains pharmaceutical and medicinal ch...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- Dedication
- Preface
- Acknowledgements
- List of figures and tables
- Abbreviations
- About the author
- Chapter 1: Introduction
- Chapter 2: Career prospects in the pharmaceutical industry
- Chapter 3: Drugs, medicines, and regulatory authorities
- Chapter 4: Bulk drugs or active pharmaceutical ingredients
- Chapter 5: Formulated drugs 1
- Chapter 6: Formulated drugs 2
- Chapter 7: The stability of medicines
- Chapter 8: Quality assurance in medicines
- Chapter 9: Pharmacological concepts and drugs
- Chapter 10: The top five most common or long-selling drugs
- Chapter 11: Life-style drugs, statins, COX-2 drugs
- Chapter 12: Counterfeit drugs and drug abuse
- Chapter 13: New pharmaceutical technology and pharmaceuticals
- Chapter 14: Future prospects for the pharmaceutical industry
- Chapter 15: Pharmaceutical case studies
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access An Introduction to Pharmaceutical Sciences by Jiben Roy in PDF and/or ePUB format, as well as other popular books in Medicine & Pharmacology. We have over 1.5 million books available in our catalogue for you to explore.