
- 239 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
About this book
Natural gas represents nearly one-quarter of the world's energy resources. More than half of American homes rely on it as their main heating fuel. It serves as the raw material necessary in everyday paints, plastics, medicines and explosives. It produces the cleanest of all fossil fuels. It is natural gas—and everybody should acquire a basic understanding of it. This valuable easy-to-use reference supplies all the basics that every person should know about the natural gas industry. Introductory engineers, managers and analysts will benefit from this informative, practical handbook. Natural gas remains a vital component of all energy sources, and with an increasing demand for information on this useful energy source, Natural Gas: A Basic Handbook is an essential tool for anyone involved in the energy industry.
Trusted by 375,005 students
Access to over 1 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Subtopic
Combustibles fossilesPart I
Origin and Properties
CHAPTER 1
History and Uses
1.1 Introduction
Natural gas (also called marsh gas and swamp gas in older texts and more recently landfill gas) is a gaseous fossil fuel found in oil fields, natural gas fields, and coal beds. As one of the cleanest, safest, and most useful of all energy sources, it is a vital component of the world’s supply of energy. While it is commonly grouped in with other fossil fuels and sources of energy, there are many characteristics of natural gas that make it unique.
Natural gas is the result of the decay of animal remains and plant remains (organic debris) that has occurred over millions of years. Over time, the mud and soil that covered the organic debris changed to rock and trapped the debris beneath the newly-formed rock sediments. Pressure and, to some extent, heat (as yet undefined) changed some of the organic material into coal, some into oil (petroleum), and some into natural gas. Whether or not the debris formed coal, petroleum, or gas depended upon the nature of the debris and the localized conditions under which the changes occurred.
Natural gas is found in reservoirs beneath the surface of the earth (see “Composition and Properties”) and is often associated with petroleum, although gas that is not associated with petroleum is also known. Production companies use sophisticated, expensive technology to find and drill into these reservoirs. Once brought from underground, the natural gas is refined to remove impurities such as water, other gases, sand, and other compounds. Some hydrocarbons, such as propane and butane, are removed and sold separately. Other impurities are also removed, such as hydrogen sulfide (the refining of which can produce sulfur, which is then also sold separately). After refining (Chapter 7), the clean natural gas is transmitted through a network of pipelines that deliver natural gas to its point of use.
Two new and possibly large sources of methane that may extend the availability of natural gas are methane hydrates (also called gas hydrates) and coal-bed methane (Berecz and Balla-Achs, 1983; Sloan, 1997; Gudmundsson et al., 1998; Max, 2000; Sloan, 2000). Their production technologies have only recently been developed, and these sources are now becoming economically competitive.
Methane-rich gases are also produced by the anaerobic decay of non-fossil organic material and are referred to as biogas. Sources of biogas include swamps, which produce swamp gas; marshes, which produce marsh gas; landfills, which produce landfill gas, as well as sewage sludge and manure, by way of anaerobic digesters, in addition to enteric fermentation, particularly in cattle.
Although natural gas is a vital component of the world’s supply of energy and one of the most useful of all energy sources, it must be understood that the word gas has a variety of different uses, and meanings. Fuel for automobiles is also called gas (being a shortened version of gasoline), but that is a totally different fuel. The gas used in a barbecue grill is actually propane (C3H8), which, while closely associated with and commonly found in natural gas and petroleum, is not really natural gas.
Natural gas is the flammable gaseous mixture that occurs alone or with petroleum in reservoirs and is predominantly methane (CH4) and some of the higher molecular weight paraffins (CnH2n+2) generally containing up to five carbon atoms (Table 1-1). Briefly, methane is the simplest member of the hydrocarbon series and has one carbon atom and four hydrogen atoms (Figure 1-1).
Table 1-1
Range of Composition of Natural Gas
| Gas | Composition | Range |
| Methane | CH6 | 70–90% |
| Ethane | C2H4 | |
| Propane | C3H8 | 0–20% |
| Butane | C4H10 | |
| Pentane and higher hydrocarbons | C5H12 | 0–10% |
| Carbon dioxide | CO2 | 0–8% |
| Oxygen | O2 | 0–0.2% |
| Nitrogen | N2 | 0–5% |
| Hydrogen sulfide, carbonyl sulfide | H2S, COS | 0–5% |
| Rare gases: Argon, Helium, Neon, Xenon | A, He, Ne, Xe | trace |
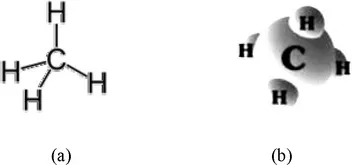
Figure 1-1 Simplified representation of methane as (a) a two-dimensional formula and (b) a three-dimensional formula.
In its purest form, the natural gas delivered to the consumer is almost pure methane, and the remaining hydrocarbons and non-hydrocarbons have been removed though refining. The non-hydrocarbon constituents include, but are not limited to, carbon dioxide (CO2), hydrogen sulfide (H2S), nitrogen (N2), and helium (He).
All of the hydrocarbon constituents of natural gas are combustible, but non-flammable non-hydrocarbon components (carbon dioxide, hydrogen sulfide, nitrogen, and helium) detract slightly from the heating value of natural gas. However, they are valuable, and in certain natural gases where their concentrations are relatively high, they may be extracted commercially.
Natural gas is colorless, shapeless, and odorless in its pure form. Quite uninteresting except that natural gas is combustible, and when burned it gives off a great deal of energy. Unlike other fossil fuels, however, natural gas is clean burning and emits lower levels of potentially harmful by-products into the air. It is the environmental aspect of natural gas use that is currently elevating its use as an important energy source.
There are several general definitions that have been applied to natural gas science and technology. For example, lean gas contains methane as the major constituent. On the other hand, wet gas contains considerable amounts of the higher molecular weight hydrocarbons. Natural gas is considered dry when it is almost pure methane, having had most of the other commonly associated hydrocarbons removed. When other hydrocarbons are present, the natural gas is wet. To further define the terms dry and wet in quantitative measures, the term dry natural gas indicates that there is less than 0.1 gallon (1 gallon, US, = 264.2 m3) of gasoline vapor (higher molecular weight paraffins) per 1,000 ft3 (1 ft3 = 0.028 m3). The term wet natural gas indicates that there are such paraffins present in the gas, in fact more than 0.1 gal/1,000 ft3.
Sour gas contains hydrogen sulfide, whereas sweet gas contains very little, if any, hydrogen sulfide. Residue gas is natural gas from which the higher molecular weight hydrocarbons have been extracted and casing-head gas is derived from petroleum but is separated at the separation facility at the wellhead.
Natural gas is considered as an environmentally friendly clean fuel, offering important environmental benefits when compared to other fossil fuels. The superior environmental qualities over coal or crude oil are that emissions of sulfur dioxide are negligible, and nitrous oxide and carbon dioxide emissions are lower. This helps to reduce problems of acid rain, ozone layer, or greenhouse gases. Natural gas is also a very safe source of energy when transported, stored, and used.
Natural gas is produced in many countries around the world and most of those countries produce both oil and natural gas; a few produce only natural gas. The ten largest natural gas producing countries are: United States, Canada, Russia, United Kingdom, Algeria, Netherlands, Iran, Indonesia, Norway, and Uzbekistan (EIA, 2004). Because transportation costs add to the cost of natural gas, in most countries natural gas is consumed within the country or exported to a neighboring country by pipeline. Technology for liquefying natural gas so that it can be transported in tankers is improving (Chapter 4). As technology continues to expand the options for gas transportation, demand for natural gas is expected to grow.
A common misconception about natural gas is that resources are being depleted at an alarming rate and the supplies are quickly running out. In fact, there is a vast amount of natural gas estimated still to be retrieved from a variety of reservoirs (Chapter 2). However, many proponents of the depletion theory believe that price spikes indicate that natural gas resources are depleted beyond the point of no return. However, price spikes of any commodity are not always caused by waning resources but can be the outcome of other forces at work in the marketplace.
1.2 History
By definition, natural gas is a naturally occurring gaseous fossil fuel that is found in oil fields, and natural gas fields, and coal beds. For clarification, natural gas is not the same as town gas, although the history of natural gas cleaning has its roots in town gas cleaning (Chapter 5).
Town gas is a generic term referring to manufactured gas produced for sale to consumers and municipalities. The terms coal gas, manufactured gas, producer gas, and syngas (synthetic natural gas, SNG) are also used for gas produced from coal. ...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- List of Figures
- List of Tables
- Preface
- Part I: Origin and Properties
- Part II: Gas Processing
- Conversion Factors
- Glossary
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.4M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Natural Gas by James G. Speight in PDF and/or ePUB format, as well as other popular books in Technologie et ingénierie & Combustibles fossiles. We have over one million books available in our catalogue for you to explore.