
- 538 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Efficient Methods for Preparing Silicon Compounds
About this book
Efficient Methods for Preparing Silicon Compounds is a unique and valuable handbook for chemists and students involved in advanced studies of preparative chemistry in academia and industry. Organized by the various coordination numbers (from two to six) of the central silicon atom of the reported compounds, this book provides researchers with a handy and immediate reference for any compound or properties needed in the area.
Edited by a renowned expert in the field, each chapter explores a different type of compound, thoroughly illustrated with useful schemes and supplemented by additional references. Knowledgeable contributors report on a broad range of compounds on which they have published and which are already used on a broad scale or have the potential to be used in the very near future to develop a new field of research or application in silicon chemistry.
- Includes contributions and edits from leading experts in the field
- Includes detailed chemical schemes and useful references for each preparative method
- Organized by the coordination numbers of the central silicon atom for each compound for easy navigation
- Serves as a go-to primer for researchers in novel compositions of silicon matter
Trusted by 375,005 students
Access to over 1 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
Physical SciencesSubtopic
Inorganic Chemistry1
Arylsilanes as Precursors of Cyclohexa-2,5-dienylsilanes
Y. Landais University of Bordeaux, Institute of Molecular Sciences, Talence, France
Abstract
Cyclohexa-2,5-dienylsilanes are prepared in good yields through Birch reduction of the corresponding arylsilanes. The methodology may be applied to multigram-scale synthesis. An alternative electrochemical approach is also available using an aluminum sacrificial anode, thus, avoiding the use of large quantities of ammonia.
Keywords
Arylsilanes; Birch reduction; Cyclohexadienes; Electrochemistry; HMPA; Sacrificial anodeThe Birch reduction of arenes is a well-known method to access functionalized cyclohexadienes using lithium, sodium, or potassium in ammonia as a reducing medium (1). This process has been applied to a large number of arenes and polyarenes, including aromatic compounds substituted with a silicon group (2). Eaborn first applied the Birch reduction to simple trialkylarylsilanes, obtaining the desired silyl-substituted cyclohexa-2,5-dienes (3,4). We extended to arylchlorosilanes this Birch reduction (5). The resulting silanol, which is obtained, may be manipulated further, for instance, through the formation of a siloxane, allowing further intramolecular hydrosilylation (6) or may be oxidized into a hydroxy group following the Tamao–Kumada–Fleming process (7–9). The method can be extended to other chlorosilanes, but steric hindrance around the silicon center is detrimental to the yield in silanol.
Preparation of such silyl-substituted cyclohexa-2,5-dienes may also be carried out as described by Woerpel through the metallation of the parent cyclohexa-2,5-diene with t-BuLi followed by the silylation of the resulting pentadienyl anion with the suitable chlorosilane (10). We have also developed an alternative electrochemical method (vide infra) using a sacrificial aluminum anode (4,5,11).
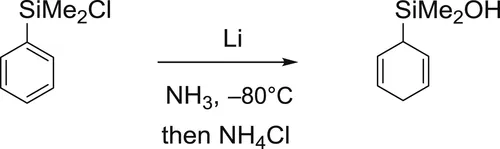
Preparation of cyclohexa-2,5-dienyldimethylsilanol
Apparatus
A dry 250-mL three-necked flask equipped with a magnetic stirrer, an inlet for argon, a low-temperature thermometer, a gas condenser cooled with liquid nitrogen, safety glasses, laboratory coat, and protective gloves.
Chemicals
Ammonia gas cylinder, lithium powder. PhMe2SiCl is commercially available but may also be prepared on 200–300 g scale from bromobenzene and Me2SiCl2 (12).
Experimental procedure
In a dry 250-mL three-necked flask, equipped with a magnetic stirrer, an inlet for argon, and a thermometer, was condensed NH3 (80 mL) at −80°C under argon. The phenyldimethylchlorosilane (1 mL, 6 mmol) was then slowly added and a white precipitate appeared. After 5 min, lithium powder (0.3 g, 42 mmol) was introduced and the solution turned immediately blue. This solution was then stirred at −80°C for 45 min and anhydrous NH4Cl was added until the blue coloration disappears. Ether (30 mL) and water (20 mL) were then added successively and ammonia was evaporated at room temperature. The aqueous layer was extracted with ether. The combined extracts were washed with water (2×) then with a saturated NaCl solution, dried over MgSO4, and the solvents were evaporated in vacuo. The residue was then purified by Kugelrohr distillation (70°C, 0.4 mbar) or by flash chromatography through Florisil (petroleum ether/EtOAc 95:5) to give the cyclohexadienylsilanol as a colorless oil (0.72 g, 77%).
Apparatus
A 100-mL one-compartment cell fitted with a sacrificial anode of aluminum and a cylindrical stainless grid.
Chemicals
LiCl, t-BuOH, hexamethylphosphoramide (HMPA).
Attention!
This experiment can only be done in a well-ventilated hood as HMPA is known as a carcinogenic solvent.
Experimental procedure
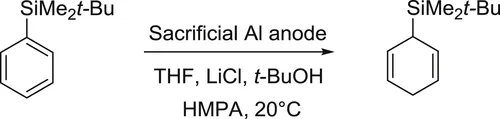
In a one-compartment cell fitted with a sacrificial anode of aluminum and a cylindrical stainless grid, was introduced under nitrogen, the supporting electrolyte LiCl (3.53 g, 83.3 mmol), t-BuOH (6 mL, 62.3 mmol), anhydrous THF (90 mL), HMPA (15 mL), and the t-butyldimethylphenylsilane (4 g, 20.8 mmol). Electrolysis (constant current 0.1 A) was then initiated and was maintained until the starting material has disappeared (≈17 h) (monitored by GC). A solution of HCl 10% (50 mL) and pentane (30 mL) was then added to the reaction mixture and the organic layer was decanted. The aqueous layer was extracted with pentane (3 × 20 mL) and the combined extracts were washed with brine, dried over MgSO4, and the solvents were evaporated in vacuo to afford the silyl...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- Dedication
- List of Contributors
- Preface
- 1. Arylsilanes as Precursors of Cyclohexa-2,5-dienylsilanes
- 2. Rhodium-Catalyzed Vinyldiazoesters Insertion Into SiH Bonds. Synthesis of Allylsilanes
- 3. Lewis Base–Stabilized Silyliums
- 4. Tetra(silyl)methane, (H3Si)4C, a Volatile Carbosilane for the Chemical Vapor Deposition of Amorphous Silicon Carbide Thin Films
- 5. Trimethylsilyl Perrhenate—A Nonionic Reagent Soluble in Organic Solvents for the Preparation of Perrhenates
- 6. Radicals, Anions, and Cations of Silicon and Silylenes
- 7. Multiple Bonding in Silicon Compounds
- 8. Silicon-Based Ligands for Transition Metal Coordination
- 9. Silylenes, Silylaminosilylene, Disilane, Silanimine, Silacyclohexadienones, Bis(silyl)-Alkenes, and Hydrosilanimine
- 10. Synthesis of Functionalized Silsesquioxanes as Molecular Templates for Hybrid Materials
- 11. Lithium Tris(2,4,6-triisopropylphenyl)disilenide: A Versatile Reagent for the Transfer of the Disilenyl Group
- 12. New Phosphine-Stabilized Si(II)-Complexes: Silicon(II)-Hydride and Silacyclopropylidene
- 13. (Monosodiumoxy)organoalkoxysilanes (Rebrov Salts)—Polyfunctional Monomers for Silicone Syntheses
- 14. Silicon(II) as a Synthon for the Access of Different Silicon(IV) and Silicon(II) Compounds
- 15. Silene, Silaimine, and Siletane Derivatives
- 16. Synthesis of a Zwitterionic 2,4-Disila-1,3-diphosphacyclobutadiene Compound
- 17. Silanetriols and Aluminosilicates
- 18. Synthesis of Silicon(II) Compounds and Their Reactions
- 19. Preparation of the NHC (L1,2) and Its Application for Synthesizing Lewis Base–Stabilized Dichlorosilylene L1,2SiCl2
- 20. Octaammonium POSS as a Building Block for Constructing Nanohybrid Materials
- 21. Tungsten- and Ruthenium-Silylene Complexes
- 22. 1,1-Di-tert-Butylsilacyclopropanes
- 23. Polysilanes, Polycarbosilanes, Dioxadisilacyclohexane, and Polysiloxanes
- 24. Synthesis of N-(Silylmethyl)amides of Carboxylic Acids and Related Compounds
- 25. Carbene Adducts of Silicon(IV) Chlorides: Versatile Reagents for Carbene Transfer and Sources for Cationic Silicon(IV) Complexes
- 26. Controlling n-Oligosilane Conformation by Stretching on a Staffane Rack
- 27. Bis-silyl Chelate Ligand Precursor XantsilH2 and Some Ruthenium Xantsil Complexes
- 28. Silyl(silylene) Complexes of Iron and Ruthenium
- 29. Cobalt-Methylidyne-Silanetriol as Precursor for Catalytic Hydroformylation in a Two-Phase System
- 30. Preparation of the SiCS Three- and the SiO2C2 Five-Membered Ring System
- 31. Preparation of SiF4(NH3)2 and Its Higher Ammoniate SiF4(NH3)2·2NH3
- 32. Silanols and Silsesquioxanes
- 33. Hydrido-Silyl Complexes of Chromium With Metal-Hydrogen-Silicon Three-Center Bonds
- 34. Sol-Gel Processing of Alkoxysilyl-Substituted Metal Complexes
- 35. Tertiary Alkyl Substituted Octasilsesquioxanes
- 36. o-(Dimesitylboryl)(dimethylsilyl)benzene: A System of Intramolecular SiH Bond Activation by o-Boryl Group
- 37. Organosilicon Synthesis for Construction of Organosilicon Clusters
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.4M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Efficient Methods for Preparing Silicon Compounds by Herbert W Roesky in PDF and/or ePUB format, as well as other popular books in Physical Sciences & Inorganic Chemistry. We have over one million books available in our catalogue for you to explore.