
- 288 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
About this book
Progress in Nuclear Physics, Volume 3 covers the advances in the experimental and theoretical studies on some aspects of nuclear physics. This volume is divided into nine chapters that include discussions on diffusion cloud chamber, precision instruments, and ?erenkov radiation. The opening chapter deals with the theory, design, and experimental applications of diffusion cloud chamber. The next chapters explored the energy measurements using proportional and solid conduction counters; the methods for studying orienting nuclei or nucleons; and the theoretical interpretation and practical applications of ?erenkov radiation. These topics are followed by discussions on positron annihilation, the role of stripping reactions in nuclear physics, and the production of intense ion beams. The final chapter considers the importance of the study of nucleon-deuteron collisions in nuclear physics. This book is of value to nuclear physicists and researchers in the allied fields.
Trusted by 375,005 students
Access to over 1 million titles for a fair monthly price.
Study more efficiently using our study tools.
1
THE DIFFUSION CLOUD CHAMBER
M. Snowden
Publisher Summary
This chapter discusses the diffusion cloud chamber. Expansion cloud chambers have been extensively used in cosmic ray research, and early experiments used randomly operated chambers. The rate of amassing data was limited by the short duration of the sensitive time after expansion and also by the long recovery time between expansions. It was the work of Langsdorf, who used a thermal gradient to obtain conditions of supersaturation, that gave the most promising results. To use one-dimensional equations to describe the diffusion and heat transfer, the effect of the walls of the chamber must be neglected, and for further simplification of the theory, the vapor is treated as a perfect gas. For stable conditions in the diffusion chamber, the density of the gas vapor mixture must decrease with the height above the base. Diffusion chambers have been largely operated using solid carbon dioxide as the primary coolant and cold temperatures have consequently been limited to about −70°C. It is desirable to achieve as great a sensitive depth as possible to utilize the chamber efficiently. Diffusion chambers have rather limited application in the field of cosmic ray research. However, it is possible that they may prove useful in the study of the structure of extensive showers where the simplicity of operation would allow several chambers to be employed in an experiment. They may also be used with counter control.
Although it is only during the past few years that diffusion cloud chambers have come into common use, it was as long ago as 1936 that experiments with chambers operating on the diffusion principle were started. A measure of success was achieved by LANGSDORF (1936–7) and later (1939) he published an account of a satisfactory, though rather complicated, diffusion cloud chamber. No further work was reported until NEEDELS and NIELSEN (1950), COWAN (1950) and MILLER, FOWLER and SHUTT (1951) described satisfactory results obtained with fairly simple designs of chamber. Since then numerous workers have constructed similar chambers and their application for research in nuclear physics is becoming widespread. In addition small chambers of very rudimentary design are being used for demonstration purposes in many laboratories.
Expansion cloud chambers have been extensively used in cosmic ray research and early experiments used randomly operated chambers. The rate of amassing data was limited by the short duration of the sensitive time after expansion and also by the long recovery time between expansions. Much effort therefore was directed towards developing a chamber which had a longer sensitive time (BEARDEN, 1935; FRISCH, 1935 and MAIER-LEIBNITZ, 1939) or required a shorter interval between expansions (BRINKMAN, 1936; SHIMIZU, 1921; GAERTTNER and YEATER, 1949). At the same time other workers looked for new principles of operation which might give continuous sensitivity. VOLLRATH (1936) obtained some success using a chemical method in which hydrochloric acid vapour and water vapour are allowed to diffuse together. The resulting mixture is supersaturated and droplets formed on cosmic ray tracks were observed but not photographed. By passing air over water at 70°C and thence into an observation channel maintained at room temperature by a surrounding water jacket, HOXTON (1934) produced conditions of continuous supersaturation. Droplet condensation was produced in the presence of electrical discharges but not observed by radioactive ionization.
It was the work of LANGSDORF (1936) who used a thermal gradient to obtain conditions of supersaturation which gave the most promising results. Methyl alcohol produced at the top of the chamber was allowed to diffuse downwards to the base of the chamber which was kept at a low temperature. An intermediate region of the chamber was observed where the supersaturation was sufficient to give condensation on ion tracks formed by cosmic rays but there was a background of continuous rain making the contrast insufficient for photographic records to be obtained.
It is this last type of continuously sensitive cloud chamber which has been rapidly developed during the past few years and is now universally referred to as the diffusion cloud chamber. Let us consider the basic requirements of such a chamber and look into the conditions necessary for satisfactory operation.
In Fig. 1 the essential details of the diffusion cloud chamber are illustrated. The body of the chamber containing the gas-vapour mixture is thermally connected to the base which is cooled to some low temperature T0. Near the glass top of the chamber is a trough containing a liquid which is vaporized by maintaining it at some temperature T1 greater than T0; a thermal gradient is therefore set up between top and bottom of the chamber.
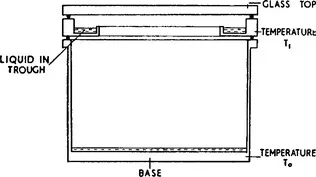
Fig. 1 Basic design of diffusion cloud chamber.
The gas at the top of the chamber is saturated with the vapour of the liquid contained in the trough and this vapour will diffuse downwards towards the cooled base. At the lower temperatures the gas becomes supersaturated with the vapour, the value of the supersaturation increasing as the temperature decreases. With a suitable gas-vapour combination and for appropriate values of T1 and T0 the super-saturation can exceed the critical value necessary to cause drop-wise condensation upon ions. This value was shown by THOMSON (1888) to be given by the expression
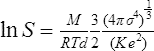
where S is the supersaturation, M is the gram molecular weight, d the density, σ the surface tension and K the dielectric constant of the liquid; T is the absolute temperature, R the gas constant and e the charge carried by the ions.
THEORY
LANGSDORF (1939) has determined the supersaturation resulting from certain temperature and vapour density distributions within the chamber. For simplicity he considered the special case where drop-wise condensation of the vapour can be neglected, a condition which could only be attained in practice by the elimination of all condensation nuclei, whether charged or uncharged, from the volume of the chamber. In order to use one-dimensional equations to describe the diffusion and heat transfer, the effect of the walls of the chamber must be neglected and for further simplification of the theory the vapour is treated as a perfect gas.
The total energy flux through the chamber can then be expressed by the equation
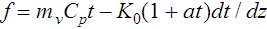
where Cp is the specific heat of the vapour, mv the vapour flux in g cm−2 sec−1 and K = K0(1 + at) the relation between the thermal conductivity of the gas vapour mixture and the temperature t. t = (T – T0) is the difference in temperature between some height z in the chamber and the base temperature T0, and dt/dz the temperature gradient along the z-axis of the cha...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- FOREWORD
- ACKNOWLEDGMENTS
- Chapter 1: THE DIFFUSION CLOUD CHAMBER
- Chapter 2: ENERGY MEASUREMENTS WITH PROPORTIONAL COUNTERS
- Chapter 3: ORIENTED NUCLEAR SYSTEMS
- Chapter 4: ČERENKOV RADIATION
- Chapter 5: ANNIHILATION OF POSITRONS
- Chapter 6: SOLID CONDUCTION COUNTERS
- Chapter 7: STRIPPING REACTIONS
- Chapter 8: THE PRODUCTION OF INTENSE ION BEAMS
- Chapter 9: THE COLLISIONS OF DEUTERONS WITH NUCLEONS
- NAME INDEX
- SUBJECT INDEX
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.4M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Progress in Nuclear Physics by O. R. Frisch in PDF and/or ePUB format, as well as other popular books in Physical Sciences & Physics. We have over one million books available in our catalogue for you to explore.