
- 192 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
About this book
Mixed oxides are the most widely used catalyst materials for industrial catalytic processes. The principal objective of this book is to describe systematically the mixed oxide catalysts, from their fundamentals through their practical applications. After describing concisely general items concerning mixed oxide and mixed oxide catalysts, two important mixed oxide catalyst materials, namely, heteropolyacids and perovskites, are taken as typical examples and discussed in detail.
These two materials have several advantages: 1. They are, respectively, typical examples of salts of oxoacids and double oxide, that is, the two main categories of mixed oxides in solid state chemistry. 2. Both exhibit excellent catalytic performance in nearly crystalline state and are used in several industrial applications. 3. They have studied for many years.
In addition, metal oxides functioning as a catalyst support (carrier) are included. Although the supports are very important in practical applications, and tremendous progress has been made in the past decades, few systematic reviews exist. It is notable that heteropolyacids and perovskite exhibit unique performance when used as a support.
Fundamental catalytic science and technology and solid state chemistry necessary is presented for the proper understanding of mixed oxide catalysts as well as for R&D. For the latter, the concept of design of practical catalysts is very important. This is considered throughout the book.
- Systematically describes design principles of mixed oxide catalysts
- Shows how catalysis and solid-state chemistry of metal oxides are inter-related
- Covers all useful basic concepts of mixed oxide catalysis
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Chapter 1
Basis of Heterogeneous Catalysis
Abstract
A concise introduction is given to the fundamental knowledge required to understand the heterogeneous catalysis of mixed oxides and to the concept of catalyst design. The fundamental knowledge includes the rate versus equilibrium relation; rate equation; the mechanism of catalytic reaction; essential functions of a catalyst, namely, activity, selectivity, and durability (or life); and catalytic reaction engineering such as mass and heat transfer. Typical methods for the preparation and characterization of catalysts and for the elucidation of the reaction mechanism, as well as a short history of industrial catalysts, are also provided.
Keywords
Rate equation; Kinetics; Reaction mechanism; Essential functions of catalyst; Industrial catalyst; Catalyst design; Catalytic reaction engineering
Fundamental concepts and principles indispensable to understand the heterogeneous catalysis of mixed oxides are described in this chapter [1].
1.1 Catalyst and Catalysis
Catalyst is a substance that is present in a small amount in the reaction system and accelerates the desired chemical reaction(s), but little changes during the reaction. Catalysis is a general term for the function of catalyst. Almost all materials and goods that are daily used are produced through catalytic processes from various raw materials.
Many high-performance catalysts are used, in order to utilize efficiently raw materials, including recycling of used materials, and to utilize efficiently the existing as well as “new” energy sources. Catalysts are used not only for chemical synthetic processes but also for other uses such as electrodes of fuel cells and batteries. In addition, catalysts are used to protect and improve the environment. The latter catalysts are called environmental catalysts, that is, “Kankyou Shokubai” in Japanese which is a new word first used by the author in the late 1970s.
1.1.1 Rate and Equilibrium of Chemical Reaction and Role of Catalyst
A catalyst changes (accelerates in most cases) the rate of chemical reactions to approach the equilibrium but does not change the equilibrium itself. It is important to distinguish between the kinetics (rate of reaction) and thermodynamics (equilibrium constant of reaction). The difference may be evident in Fig. 1.1, which schematically shows the case of a simple reversible reaction of A ↔ B. The rate to approach the equilibrium composition changes from a catalyst to another, but the equilibrium finally attained is the same. If the forward and reverse reactions are first order, the rates are expressed by Eq. (1.1) as
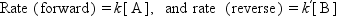
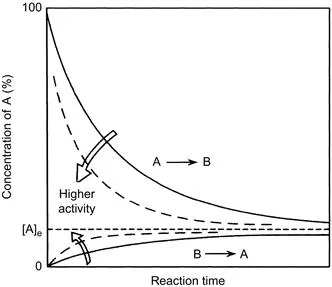
Figure 1.1 Time course of a reversible reaction, A ↔ B. The concentration reaches faster the equilibrium concentration [A]e over a catalyst with a higher activity, but the equilibrium concentration is the same.
At the equilibrium, the rates of forward and reverse reactions are identical, then,
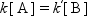
where the ratio of [B]/[A] is being determined by the equilibrium constant, K = k/k′. With an active catalyst, the rate to approach the equilibrium is fast, and for a less active catalyst it is slower. Therefore, both forward and reverse reactions are faster over a more active catalyst at the equilibrium, while both rates are identical for each case.
1.1.2 Three Essential Functions of Catalyst
Catalytic activity, selectivity, and durability (= catalyst life) are the three most important functions of catalyst.
Activity is most fundamental. A reaction which does not occur in the absence of catalyst could proceed in the presence of catalyst. With a more active catalyst, the production rate per volume of reactor becomes larger and the reactor volume can be made smaller.
Selectivity is the most interesting and attractive function of catalyst, which selects one (or more) desirable reaction(s) among many reactions that would possibly occur. Choosing a reaction which produces a thermodynamically unfavorable but valuable product is one of the most attractive functions of catalyst. A fascinating function of catalyst is stereoselectivity that produces one of the two stereoisomers which have thermodynamically the same stability.
The selectivity may be divided into two categories (Eq. 1.3). One is to select one product among several possible products starting from one reacting molecule, e.g., choosing only A to B (Eq. 1.3a). This may be called (a) product-selectivity. Another type is to select one reactant in a mixture of several reactants choosing A from a mixture of A and B (Eq. 1.3...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- Preface
- Chapter 1. Basis of Heterogeneous Catalysis
- Chapter 2. Chemistry and Catalysis of Mixed Oxides
- Chapter 3. Catalysis of Perovskite and Related Mixed Oxides
- Chapter 4. Catalysis of Heteropoly Compounds (Polyoxometalates)
- Chapter 5. Mixed Oxides as Catalyst Supports
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Heterogeneous Catalysis of Mixed Oxides by M. Misono in PDF and/or ePUB format, as well as other popular books in Technology & Engineering & Inorganic Chemistry. We have over 1.5 million books available in our catalogue for you to explore.