![]()
CHAPTER
By the End of This Chapter You Will:
•Start to recognise the importance of stereochemistry.
•Start to understand what stereoisomerism means.
•Gain a feeling for the ways we draw and describe stereoisomers.
•Understand how to use this book.
What You Will Get from This Chapter
This short chapter is really just to set the scene for the rest of the book, highlighting the importance of stereochemistry and the approaches we can use to study the topic. Don't worry if the new material appears daunting at this point. We'll go through it slowly in the relevant chapters. Perhaps the most important bit of Chapter 1 is the final section, Section 1.4, explaining ‘how to use this book’.
![]()
Stereochemistry
1.1 Thalidomide – Why Stereochemistry Is Important
In the 1960s, thalidomide was an over-the-counter drug that was given to pregnant women, mostly in Germany, to treat morning sickness. The use of this drug led to 10 000 babies being born with phocomelia (deformed limbs). Approximately half of the children survived and have had to live with this condition.
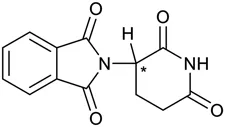
Representing the structure of thalidomide as shown in Figure 1.1 does not explain why this happened, because it gives the impression that it is a flat molecule. Importantly, one of the carbon atoms (marked *) is bound to four different groups. This means that two types of isomer can form, based on the 3D orientation of the four groups.
Figure 1.1Planar representation of thalidomide.
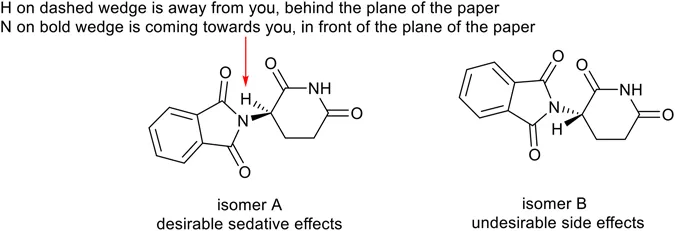
A better representation to show this 3D effect is to represent the bonds as wedged to show groups pointing towards you, and as hashed to show groups pointing away, as in Figure 1.2. Here we can see the two isomers that thalidomide can have. It turns out that the isomer on the right is the one responsible for phocomelia. If the company that produced the drug had been aware of this then it is possible that they could have sold the drug as only the bioactive isomer (although interconversion of the isomers in the body, known as racemisation, might still have complicated matters).
Figure 1.22D ‘hashed and wedged’ representation of thalidomide shows that it exists as two isomers.
This story, perhaps more than any other in history, signifies why it is important to have an understanding of molecules in 3D. This topic is known as stereochemistry and throughout this book we will introduce the main concepts in this area.
1.2 Stereoisomerism
You may well have come across types of isomerism, such as structural isomerism, where the atoms in two molecules with the same molecular formula are joined together in different ways. This book will introduce another type of isomerism, called stereoisomerism. This is when the atoms are connected in the same way but they adopt different positions in space.
Once you've understood what kinds of stereoisomers there are, it's important to consider how readily they can interconvert. If they basically can't, we have a set of configurational isomers that we may even be able to separate and study separately. Even if they can interconvert, we may be able to observe different conformational isomers by spectroscopy – understanding which isomer is of lowest energy can be helpful in predicting certain properties of molecules, including how they react.
1.2.1 Configurational Isomerism
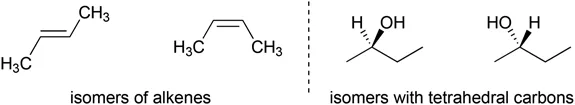
Configurational isomers (Figure 1.3) are stereoisomers that cannot interconvert readily. We will explain how these types of isomers can be seen in alkenes, tetrahedral carbons, planar organic molecules and inorganic complexes.
Figure 1.3Examples of configurational isomers.
Configuration as a topic is covered in Chapter 2 (alkenes), Chapter 3 (tetrahedral carbons), Chapter 6 (helicenes, biphenyls and allenes) and Chapter 7 (inorganic complexes).
1.2.2 Conformational Isomerism
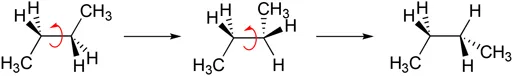
Any organic molecule that can have rotation about a bond can exist as conformational isomers (Figure 1.4). This topic relates to the different spatial arrangements in 3D that molecules can have through the rotation of bonds.
Figure 1.4Example of conformations.
box1 Don’t worry if Figure 1.4 isn’t making much sense at the moment. It’s difficult to visualise features like this, which is why we’ve written this book!
We'll be studying the conformations of acyclic (no ring structures) and cyclic compounds in Chapters 4 and 5, respectively.
Of course, all this requires us to represent the stereochemical information in the chemical structures we draw, as in Figures 1.2–1.4. This isn't easy, but we'll guide you through it step by step.
1.3 Representing Molecules in 3D
As we've said, as we discuss different concepts of stereochemistry and, essentially, the 3D structure of molecules, we will be introducing the different ways of drawing and naming molecules that represent their 3D character. One of the most unfamiliar and tricky areas is representing conformations, but we'll get there!
1.3.1 New Ways of Drawing Molecules
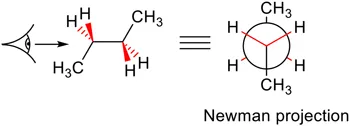
A Newman projection is the perspective obtained by looking down a specific bond in a molecule. It is used most often with alkanes where the central C–C bond is considered. The C atom closer to you has the lines for the bonds running to it and the further C atom is represented by a circle, as shown in Figure 1.5. Newman projections are useful in explaining effects such as the Karplus relationship in NMR spectroscopy, and to help us identify the lowest-energy conformations of molecules that arise from steric and electronic interactions.
Figure 1.5Newman projection.
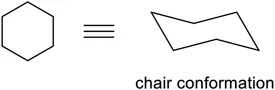
Chair conformations (Figure 1.6) represent a more accurate view of what a six-membered ring, such as a cyclohexane, looks like in 3D. By looking at chair conformations, we can tell what the most stable form of a particular six-membered ring is. This can then allow us to predict what will form from a reaction, and hence how we can control the stereochemistry of the products.
Figure 1.6Chair conformation.
1.3.2 Stere...