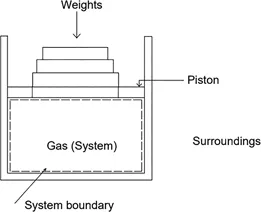
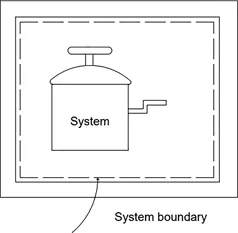
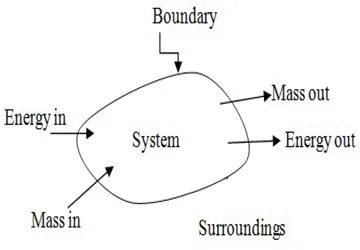
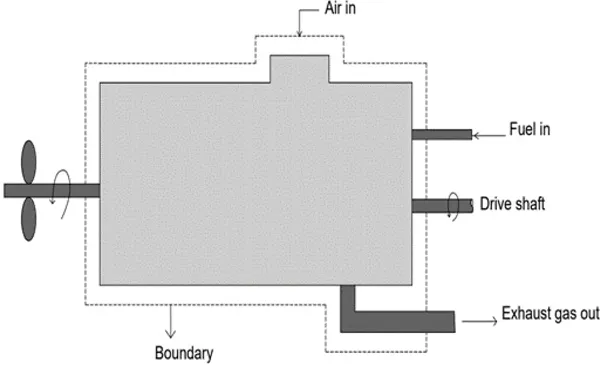
This textbook comprehensively covers the fundamentals and advanced concepts of thermodynamics in a single volume.
It provides a detailed discussion of advanced concepts that include energy efficiency, energy sustainability, energy security, organic Rankine cycle, combined cycle power plants, combined cycle power plant integrated with organic Rankine cycle and absorption refrigeration system, integrated coal gasification combined cycle power plants, energy conservation in domestic refrigerators, and next-generation low-global warming potential refrigerants. Pedagogical features include solved problems and unsolved exercises interspersed throughout the text for better understanding.
This textbook is primarily written for senior undergraduate students in the fields of mechanical, automobile, chemical, civil, and aerospace engineering for courses on engineering thermodynamics/thermodynamics and for graduate students in thermal engineering and energy engineering for courses on advanced thermodynamics. It is accompanied by teaching resources, including a solutions manual for instructors.
FEATURES
- Provides design and experimental problems for better understanding
- Comprehensively discusses power cycles and refrigeration cycles and their advancements
- Explores the design of energy-efficient buildings to reduce energy consumption
Property tables, charts, and multiple-choice questions comprise appendices of the book and are available at https://www.routledge.com/9780367646288.