eBook - ePub
Anterior Skull Base Tumors
- 256 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Anterior Skull Base Tumors
About this book
In recent years, interest in the management of anterior skull base tumors has been fostered by the introduction and subsequent rapid expansion of indications of transnasal endoscopic techniques. In parallel, extraordinary progress has been made in all the other disciplines which are involved in the complex process of managing anterior skull base tumors, leading to substantial improvements in diagnosis and treatment. The anterior skull base can be involved in a large variety of tumors of a varying nature and histology, which in the majority of cases originate from adjacent anatomic sites. In fact, primary lesions of the anterior skull base mainly include osteoma and other tumors originating from bone and cartilage. Tumors involving the anterior skull base have their origin prevalently in the sinonasal tract, but lesions developing on the intracranial site, such as meningiomas, may also extend caudally to encroach upon this area. All these lesions are rare, which means that the pertinent literature does not include prospective studies or treatment guidelines based on a high level of evidence. In view of the extreme histological variability of lesions involving the anterior skull base, much emphasis has been placed on addressing the different nuances of treatment in relation to histology, especially for malignant tumors. The chapters focusing on surgery provide divergent views on selection criteria for a specific surgical technique, which is the aim of this publication.
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Nicolai P, Bradley PJ (eds): Anterior Skull Base Tumors. Adv Otorhinolaryngol. Basel, Karger, 2020, vol 84, pp 87–105 (DOI: 10.1159/000457929)
______________________
Particle Therapy: Protons and Heavy Ions
Alexandra D. Jensen
Department of Radiation Oncology, University Hospitals Giessen and Marburg (UKGM), Marburg, Germany
______________________
Abstract
Particle therapy is a comparatively new radiotherapy treatment form allowing the application of extremely conformal treatment plans. In head and neck malignancies, especially in the paranasal sinus and anterior skull base, this technology is very valuable in order to maintain normal tissue tolerance of critical structures while still applying high doses to the tumour. The following paper will further explore the rationale of particle therapy and available clinical experience of the most commonly treated malignancies arising in these anatomical sites. The potential of particle therapy for re-irradiation in tumours of the paranasal sinus and anterior skull base will also be summarised.
© 2020 S. Karger AG, Basel
Squamous cell carcinoma (SCC), adenocarcinoma (AC), and mucosal melanoma, adenoid cystic carcinoma (ACC), and olfactory neuroblastoma are the most frequent malignancies in the paranasal sinuses and anterior skull base [1, 2]. These tumours require high radiation doses to achieve durable local control and hence present a huge challenge due to the close proximity of critical structures such as the optic nerves, chiasm, and brain stem. The radiation tolerance of organs at risk critically limit the application of high radiation doses in (standard) photon radiotherapy.
As pointed out by Hermann Holthusen in the 1930s, radiation oncologists act within a narrow therapeutic window of compromise between tumour control and acceptable toxicity (Fig. 1). In the past decades technical developments such as 3D conformal therapy, stereotactic radiotherapy, intensity-modulated radiotherapy (IMRT) in combination with image-guided and adaptive radiotherapy (IGRT and ART) have produced increasingly conformal dose distributions which have reduced treatment toxicity, but so far been unable to substantially improve tumour control [3, 4].
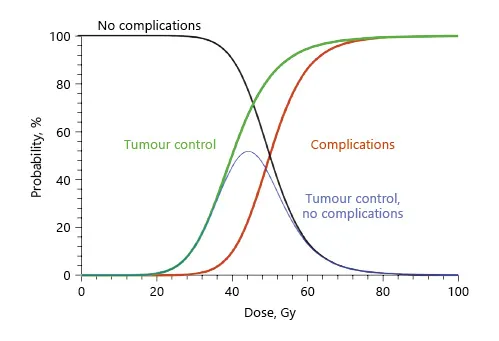
Fig. 1. Holthusen curve.
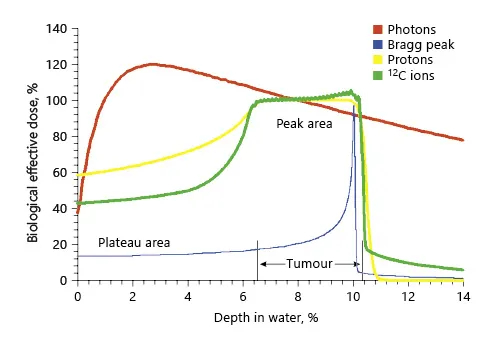
Fig. 2. Bragg peak and SOBP. Reprinted with permission from Springer [5].
The physicist Robert R. Wilson was the first to propose protons and heavy ions for treatment malignant tumours and pioneered this idea at the Berkeley Radiation Laboratory where more than 2,500 patients were treated with this technique. As opposed to photons and neutrons, radiotherapy with charged particles has several advantages. Charged particles lose most of their energy toward the end of their path (“Bragg peak”), and their penetration depth is characterised by their specific mass and charge as well as their kinetic energy on entering tissue. Beyond the Bragg peak, the dose drops to almost zero leading to extremely sharp dose gradients towards normal tissues. In order to use charged particles for clinical treatments, multiple Bragg peaks need to be superposed, creating a so-called spread-out Bragg peak (SOBP; Fig. 2) [5]. This is achieved by modulating particle energy in an accelerator system (active beam application). In view of their physical properties, charged particle beams are ideally suited for dose escalation in complex anatomical sites such as paranasal sinuses or the base of the skull where tumours are located close to critical and radiosensitive organs at risk. The use of this highly conformal radiation technique therefore allows to increase the therapeutic window by increasing the dose to the tumour without consequently increasing the dose and thereby toxicity to critical structures such as the brain stem, optic nerves, or optic chiasm (Fig. 3a, b). Multiple planning comparisons of IMRT and protons or carbon ions support the dosimetric advantages [5–8]. Heavy ion beams have some advantages over proton radiotherapy: while both can produce highly conformal dose distributions, heavy charged particles such as carbon ions have a higher mass than protons. Therefore, Coulomb scatter and hence lateral penumbra is much smaller in heavy ions as compared to protons, which may further improve their toxicity profile.
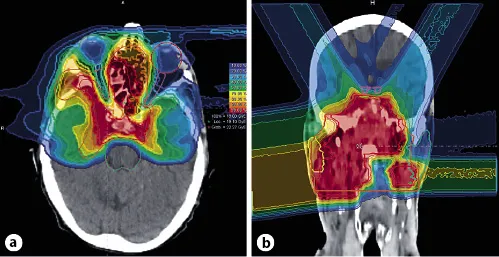
Fig. 3. Four-field intensity-controlled carbon ion therapy with good sparing of optic nerves (blue, pink), chiasm (pink), and brain stem (bright green) in a patient with SNUC. a Axial dose distribution. b Coronal dose distribution. Courtesy of Heidelberg Ion Beam Therapy Centre (HIT).
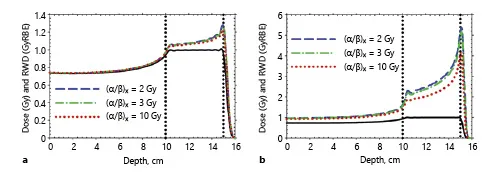
Fig. 4. RBE as a function of the position within the particle track. a Protons. b Carbon ions. Reprinted from Frese et al. [9], with permission from Elsevier.
In addition, particle beams also offer radiobiological benefits as compared to standard photon beams. High-LET beams such as ion or neutron beams show an increased relative biological effectiveness (RBE). However, the RBE changes with the position within the particle track, as shown in Figure 4 [9]. Particle beams achieve their maximum RBE with the Bragg peak towards the end of their penetration depth. In scanned particle beams, the RBE not only depends on the position within the beam, but also on the particle charge, energy, and absorbed dose in the tissue. Especially with multiple irradiation fields and dose contributions by different particle tracks, the RBE is no longer intuitive: sophisticated calculation models are an integral part of the respective treatment planning system [10–13]. While the effects of photon irradiation depend on the presence of oxygen in cells, this is not the main mechanism of action in heavy charged particles, which may be especially advantageous in hypoxic tumours. In addition, radiosensitivity in particle radiotherapy varies less within the cell cycle. Due to massive energy depositions by single particle tracks, cell repair phenomena are less prominent, therefore fractionation effects are much smaller in ion radiotherapy. Also, as particle radiation allows less cell repair, it is selectively more efficient against tumour cells with high repair capacities (low alpha/beta) such as chordomas, chondrosarcomas, prostate carcinomas, etc.
Planning comparisons show extensively that particle dose distributions are indeed superior to conventional and IMRT treatment plans [5, 7, 8], but clinical data to show that these dosimetric advantages do indeed translate into improved clinical outcomes are not available for all histologies. Although there is increasing interest in particle radiotherapy and more facilities being projected and opened throughout the USA, Europe, and Asia [14], the technology remains expensive and availability is still limited to specialised centres. The following paragraphs describe available published evidence on particle therapy for the most common head and neck malignancies in the paranasal sinus and anterior skull base.
Salivary Gland Malignancies
The treatment of malignant salivary gland tumours (MSGT) of the head and neck is complex: while the majority of salivary gland malignancies arise in the minor salivary glands and most commonly in the paranasal sinuses, they are also comparatively radioresistant. Doses in excess of 60 Gy are needed to achieve long-term local control in this disease [15–18].
In principle, there are three strategies to increase therapeutic effects of radiation: the addition of radiosensitising agents (chemoradiation), dose escalation, or the use of heavy particles in order to enhance the radiobiological effectiveness of radiation.
Data on radiosensitisation in MSGTs and especially ACC is clearly limited. Only a few groups have reported their experience with chemoradiation using platin compounds [19–24] or bioradiation [25, 26]. Reports are mostly retrospective and patient numbers are small. There is increasing interest in chemoradiation for the treatment for MSGTs: RTOG 1008 is a prospective randomised multicentric phase II trial currently recruiting patients to investigate this issue further [27]. Until this trial has been completed and the results are mature, chemoradiation remains experimental.
There are two other options to increase biological effectiveness: either by employing other radiotherapy modalities such as neutrons or heavy charged particles, or by dose escalation. The RTOG-MRC trial randomised patients with head and neck malignancies to radiotherapy with photons versus neutrons. Treatment consisted of either 70-Gy standard fractionated or 55-Gy hypofractionated photons versus 16.5- to 22-Gy neutrons in 12 fractions over 4 weeks. Following the interim analysis that showed locoregional control was significantly higher in the neutron arm as compared to the photon arm (67 vs. 17% at 2 years), trial recruitment was terminated early. Overall survival did show a trend towards improvement in the neutron arm (62 vs. 25% at 2 years) but the benefit was not statistically significant [28] and no longer detectable at 5 and 10 years of follow-up [29]. The Seattle group updated their prior neutron experience [30, 31] in 2000 and reported outcomes of a large patient cohort with ACC (159 patients) who were treated between 1985 and 1997 with a median dose of 19.2 nGy (1.05–1.7 nGy/fraction). Forty-eight percent of patients had T4 disease, and 95% had gross residual or unresectable disease. Local control in patients with gross residual disease was 57% at 5 years but varied considerably according to subsites in the head and neck with between 21% for ACCs of the nasopharynx and 67 and 68% for ACCs of the parotid and oral cavity [32]. Also, Douglas et al. [32] saw local control rates as high as 80% for ACCs of the lacrimal gland. With a median follow-up of 75 months, these results have recently been updated by Gensheimer et al. [33]. Local control in these 11 patients was 80% at 3 and 5 years and an estimated 55% at 10 years. Overall survival at 5 years was estimated to be 71% in the complete cohort [32] and 90% in lacrimal gland ACC [33].
The European neutron centres have also shared their expertise from the 1980s and early 1990s. Patient numbers ranged from 8 to 72 with a follow-up between 36 and 52 months [34–36]. Patients, mostly with gross residual or unresectable disease, received between 13.7 and 19 nGy in 3–4 fractions per week. L...
Table of contents
- Cover Page
- Front Matter
- Macroscopic and Endoscopic Anatomy of the Anterior Skull Base and Adjacent Structures
- Pathology and Differential Diagnosis of Anterior Skull Base Tumours
- The Role of Morphologic and Functional Imaging in Pretreatment Assessment
- Transnasal Endoscopic Surgery: Surgical Techniques and Complications
- Anterior Skull Base Surgery in the 21st Century: The Role of Open Approaches
- Update of Radiation Techniques Using Photons for Anterior Skull Base Tumors
- The Evolving Role of Systemic Therapy in the Primary Treatment of Sinonasal Cancer
- Particle Therapy: Protons and Heavy Ions
- Benign Tumors of the Anterior Cranial Base
- Endoscopic Endonasal Approaches for Anterior Skull Base Meningiomas
- Squamous Cell Carcinoma and Its Variants
- A Comprehensive Update on Intestinal- and Non-Intestinal-Type Adenocarcinomas
- Olfactory Neuroblastoma
- Neuroendocrine Carcinoma and Sinonasal Undifferentiated Carcinoma
- Sinonasal Malignant Melanoma
- Adenoid Cystic Carcinoma
- Other Rare Sinonasal Malignant Tumours Involving the Anterior Skull Base
- Posttreatment Imaging Surveillance
- Treatment Options for Recurrent Anterior Skull Base Tumors
- Future Perspectives in the Management of Tumors of the Anterior Skull Base
- Author Index
- Subject Index
- Back Cover Page
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Anterior Skull Base Tumors by Piero Nicolai,Patrick J. Bradley,P. Nicolai,P.J. Bradley,Piero, Nicolai,Patrick J., Bradley, Patrick J. Bradley,Patrick J., Bradley,Piero, Nicolai,Patrick J., Bradley in PDF and/or ePUB format, as well as other popular books in Medicine & Oncology. We have over 1.5 million books available in our catalogue for you to explore.