Fundamentals of Nuclear Science and Engineering, Third Edition, presents the nuclear science concepts needed to understand and quantify the whole range of nuclear phenomena. Noted for its accessible level and approach, the Third Edition of this long-time bestselling textbook provides overviews of nuclear physics, nuclear power, medicine, propulsion, and radiation detection. Its flexible organization allows for use with Nuclear Engineering majors and those in other disciplines. The Third Edition features updated coverage of the newest nuclear reactor designs, fusion reactors, radiation health risks, and expanded discussion of basic reactor physics with added examples. A complete Solutions Manual and figure slides for classroom projection are available for instructors adopting the text.

- 660 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Fundamentals of Nuclear Science and Engineering
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Chapter 1
Fundamental Concepts
The last half of the twentieth century was a time in which tremendous advances in science and technology revolutionized our entire way of life. Many new technologies were invented and developed in this time period from basic laboratory research to widespread commercial application. Communication technology, genetic engineering, personal computers, medical diagnostics and therapy, bioengineering, and material sciences are just a few areas that were greatly affected.
Nuclear science and engineering is another technology that has been transformed in less than fifty years from laboratory research into practical applications encountered in almost all aspects of our modern technological society. Nuclear power, from the first experimental reactor built in 1942, has become an important source of electrical power in many countries. Nuclear technology is widely used in medical imaging, diagnostics, and therapy. Agriculture and many other industries make wide use of radioisotopes and other radiation sources. Finally, nuclear applications are found in a wide range of research endeavors such as archaeology, biology, physics, chemistry, cosmology, and, of course, engineering.
The discipline of nuclear science and engineering is concerned with quantifying how various types of radiation interact with matter and how these interactions affect matter. In this book, we will describe sources of radiation, radiation interactions, and the results of such interactions. As the word “nuclear” suggests, we will address phenomena at a microscopic level, involving individual atoms and their constituent nuclei and electrons. The radiation we are concerned with is generally very penetrating and arises from physical processes at the atomic level.
However, before we begin our exploration of the atomic world, it is necessary to introduce some basic fundamental atomic concepts, properties, nomenclature, and units used to quantify the phenomena we will encounter. Such is the purpose of this introductory chapter.
1.1 Modern Units
With only a few exceptions, units used in nuclear science and engineering are those defined by the SI system of metric units. This system is known as the “International System of Units” with the abbreviation SI taken from the French “Le Système International d’Unitès.” In this system, there are four categories of units: (1) base units of which there are seven, (2) derived units which are combinations of the base units, (3) supplementary units, and (4) temporary units which are in widespread use for special applications. These units are shown in Table 1.1. To accommodate very small and large quantities, the SI units and their symbols are scaled by using the SI prefixes given in Table 1.2.
Table 1.1. The SI system of units and their four categories.
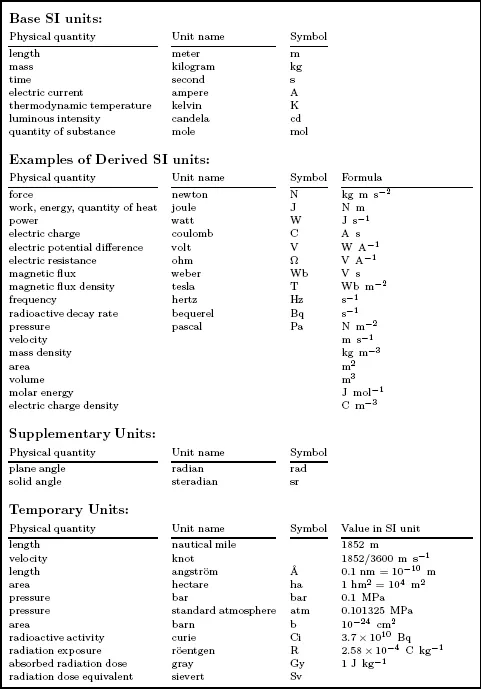
Source: NBS Special Publication 330, National Bureau of Standards, Washington, DC, 1977.
There are several units outside the SI which are in wide use. These include the time units day (d), hour (h), and minute (min); the liter (L or ℓ); plane angle degree (°), minute (′), and second (″); and, of great use in nuclear and atomic physics, the electron volt (eV) and the atomic mass unit (u). Conversion factors to convert some non-SI units to their SI equivalent are given in Table 1.3.
Finally it should be noted that correct use of SI units requires some “grammar” on how to properly combine different units and the prefixes. A summary of the SI grammar is presented in Table 1.4.
Table 1.2. SI pr...
Table of contents
- Cover
- Half Title
- Title Page
- Copyright Page
- Table of Contents
- 1 Fundamental Concepts
- 2 Modern Physics Concepts
- 3 Atomic/Nuclear Models
- 4 Nuclear Energetics
- 5 Radioactivity
- 6 Binary Nuclear Reactions
- 7 Radiation Interactions with Matter
- 8 Detection and Measurement of Radiation
- 9 Radiation Doses and Hazard Assessment
- 10 Principles of Nuclear Reactors
- 11 Nuclear Power
- 12 Fusion Reactors and Other Conversion Devices
- 13 Nuclear Technology in Industry and Research
- 14 Medical Applications of Nuclear Technology
- A Fundamental Atomic Data
- B Atomic Mass Table
- C Cross Sections and Related Data
- D Decay Characteristics of Selected Radionuclides
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Fundamentals of Nuclear Science and Engineering by J. Kenneth Shultis,Richard E. Faw in PDF and/or ePUB format, as well as other popular books in Technology & Engineering & Radiology, Radiotherapy & Nuclear Medicine. We have over 1.5 million books available in our catalogue for you to explore.