This book covers different aspects of Inorganic Chemistry in terms of 10 Chapters with in-depth and up-to-date coverage. Starting with the VSEPR theory in the first chapter, the book symmetrically presents delocalized p-bonding in polyatomic molecules; structure, bonding and topology of borane and related compounds; synthesis and reactivity of metal clusters and their bonding; some aspects of stability constants of metal complexes; magnetochemistry; mechanism of inorganic reactions; molecular orbital (MO) approach of bonding in transition metals; bonding in organometallic sandwich compounds based on MO approach. Safe and economical inorganic experiments at UG and PG Levels are also presented in the last chapter. At the end, five relevant topics are included as appendices for updating students and faculty members.

- 663 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
Physical SciencesSubtopic
Atomic & Molecular PhysicsChapter I Valence shell electron pair repulsion (VSEPR) theory: principles and applications
1.1 Introduction
This theory was first formulated by Sidgwick and Powell (1940) based on the repulsions between electron pairs, known as valence shell electron pair repulsion (VSEPR) theory to explain molecular shapes and bond angles of molecules of non-transition elements. Later on Gillespie and Nyholm (1957) developed an extensive rationale (basis/underlying principle) called VSEPR model of molecular geometry.
According to this theory, the shape of a given species (molecule or ion) depends on the number and nature of electron pairs surrounding the central atom of the species.
1.2 Postulates of VSEPR theory: Sidgwick and Powell
The various postulates of this theory are as follows:
- The unpaired electrons in the valence shell of central atom form bond pairs (bps) with surrounding atoms while paired electrons remain as lone pairs (lps).
- The electron pairs surrounding the central atom repel each other. Consequently, they stay as far apart as possible in space to attain stability.
- The geometry and shape of the molecule depend upon the number of electron pairs (bond pair as well as lone pair) around the central atom.
- The geometrical arrangements of electron pairs with different number of electron pairs around central atom are given in Table 1.1.
Table 1.1:Shapes of the various molecules depending upon the number of shared electrons around the central metal atom.
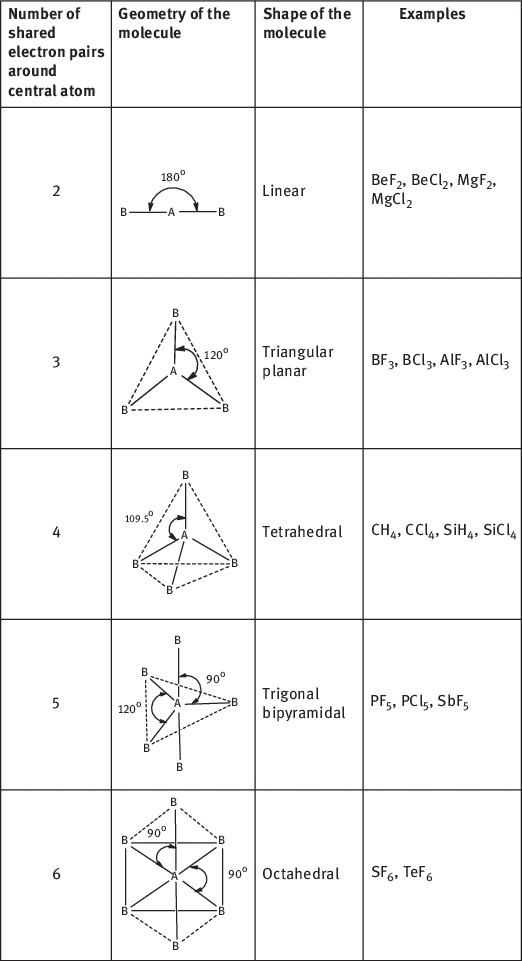 |
1.3 Rules proposed by Gillespie and Nyholm
The following rules have been proposed by Ronald Gillespie and Ronald Sydney Nyholm of University College of London to explain the shape of a number of polyatomic molecules or ions.
1.3.1 Rule 1. Spatial arrangement of electron pairs around the central atom of a given molecule/ion
The electron already present in the valence shell of the central atom of the given species plus the electron acquired by the central atom as a result of bonding with other atoms are called the valence shell electrons. Half of this gives the number of electron pairspresent in the valence shell of the central atom of the given species.
Electron pairs present in the valence shell of the central atom occupy localized orbitals which arrange themselves in space in such a way that they keep apart from one another as for as possible. This gives minimum energy and maximum stability to the species. As there can be only one definite orientation of orbitals corresponding to minimum energy, a molecule or ion of a given substance has a definite shape, that is, a definite geometry.
When the central atom in a molecule is surrounded by bonded electron pair only, the molecule will have a regular geometry or shape. The geometry depends on the number of bonded electron pairs as given in Table 1.1.
1.3.2 Rule 2. Regular and irregular geometry: presence of hybrid orbitals containing bond pairs and lone pairs
If...
Table of contents
- Title Page
- Copyright
- Contents
- Preface
- Chapter I Valence shell electron pair repulsion (VSEPR) theory: principles and applications
- Chapter II Delocalized π-bonding in polyatomic molecules: molecular orbital approach
- Chapter III Chemistry of borane and related compounds: structure, bonding and topology
- Chapter IV Synthesis and reactivity of metal clusters, and their bonding based on molecular orbital approach
- Chapter V Stability constants of metal complexes: some aspects
- Chapter VI Principles of magnetochemistry and its multiple applications in coordination compounds
- Chapter VII Mechanism of inorganic reactions: a study of metal complexes in solution
- Chapter VIII Bonding in transition metal complexes: molecular orbital theory approach
- Chapter IX Bonding in organometallic sandwich compounds: molecular orbital theory approach
- Chapter X Some aspects of safe and economical inorganic experiments at UG and PG levels
- Appendix III Nomenclature of inorganic compounds: the rules
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Inorganic Chemistry by Ram Charitra Maurya in PDF and/or ePUB format, as well as other popular books in Physical Sciences & Atomic & Molecular Physics. We have over 1.5 million books available in our catalogue for you to explore.