![]()
CHAPTER 1
Engineering the Architecture of 3D Graphene-based Macrostructures
S. CHANDRASEKARANa, M. R. CERÓNa AND M. A. WORSLEY*a
a Physical and Life Sciences Directorate, Lawrence Livermore National Laboratory, 7000 East Ave. L-367, Livermore, CA, USA,
*E-mail:
[email protected] Graphene aerogels are promising materials for the next generation of energy and environmental technologies. They exhibit good electrical conductivity, large surface areas, extraordinary mechanical properties, and as composites can possess a wide range of novel functionalities. However, in order to truly harness their potential, one must understand how the design and assembly of these 3D graphene networks impact their final properties. In this chapter, we explore the various types of graphene-based aerogels reported to date and how their architecture impacts their ultimate performance.
1.1 Introduction
Carbon is one of the most abundant elements on Earth with unique mechanical, thermal and electrical properties. Carbon has raised a lot of interest in the scientific community because of its intriguing properties and structural variability. The possibility of forming three different strong covalent bonds (with sp1, sp2 and sp3 hybridization) makes carbon a very promising element for both material scientists and engineers.
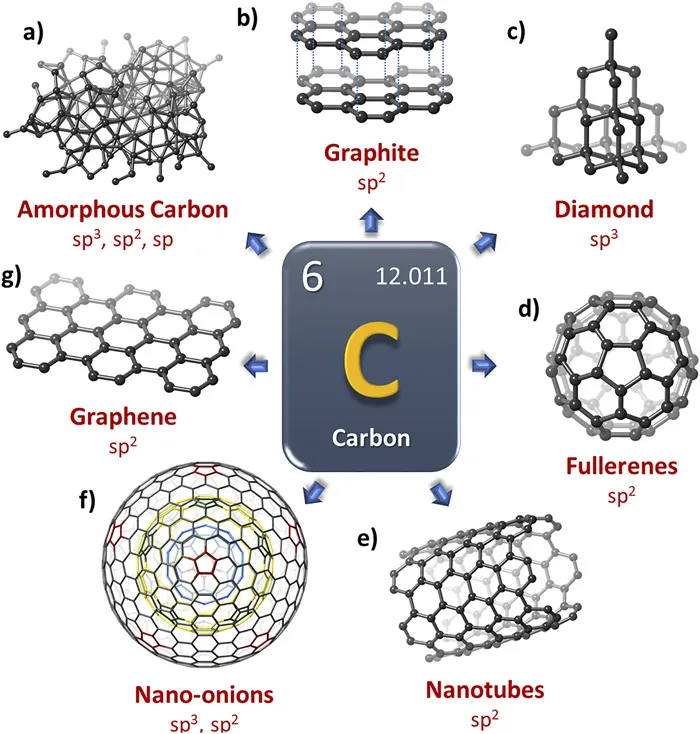
Carbon can be found in different allotropic forms depending on its hybridization and crystalline structure (Figure 1.1). For instance, the first non-amorphous allotropic form of carbon discovered was graphite. In graphite, the carbon atoms are arranged in a hexagonal structure through sp2 hybridization (Figure 1.1b). In diamond, the carbon atoms have an sp3 hybridization forming four tetrahedral bonds with the four nearest neighbors to create diamond cubic unit cells (Figure 1.1c). In 1985, fullerenes or buckyballs were discovered by Kroto et al. as an unexpected result of investigating particles found in space.1 The 0D fullerene carbon allotrope forms a hollow cage of carbon atoms connected by single and double bonds, resulting in twelve pentagons, where each pentagon is surrounded by five hexagons (Figure 1.1d).2
Figure 1.1Allotropic forms of carbon. (a) Amorphous carbon. (b) Graphite. (c) Diamond. (d) Fullerenes. (e) Nanotubes. (f) Nano-onions.
Soon after the discovery of fullerenes, carbon nanotubes (CNTs)3,5 and carbon nano-onions (CNOs)6 were reported in 1991 and 1992, respectively (Figure 1.1e and f). CNOs consist of multi-layered spherical or polyhedral shaped closed carbon shells with a structure resembling that of an onion (Figure 1.1f). The 1D CNTs consist of a tube with diameters generally in the nanometer range and are broadly distinguished by the number of concentric walls that make up the tube (Figure 1.1e). Generally, there are two different types of CNTs, single wall carbon nanotubes (SWCNTs) and multi-wall carbon nanotubes (MWCNTs).
Unrolling a SWCNT results in the last carbon allotrope discovered: graphene (Figure 1.1g). The so-called 2D graphene describes a single layer of carbon taken from a 3D graphite block, first observed using electron microscopy in 1962.7 Forty-two years later graphene was “rediscovered”, isolated, and characterized by Novoselov et al.8 Graphene has a special set of properties, such as high electrical and thermal conductivity,9 enormous specific surface area (1168 m2 g−1),9 and larger nonlinear diamagnetism than graphite,10 which set it apart from other allotropes of carbon. It is approximately 100 times stronger than steel, yet with a much lower density, having one of the largest strength-to-weight ratios observed.11
Given these impressive properties, graphene and graphene-based materials have great potential in numerous applications12 such as energy storage,13,17 nanoelectronics,18,19 sensors,20,21 catalysis,22,23 and composites,24,26 among others.27 However, in many cases we do not observe the full theoretical potential of the synthesized composites.28 These sub-par outcomes have been attributed to poor graphene dispersion due to π–π stacking interactions between several graphene sheets. One of the best strategies to overcome poor graphene dispersion, while keeping the intrinsic properties of graphene, is breaking the symmetry of the single sheets by introducing oxygen functional groups in the backbone. In this context, graphene oxide (GO) is widely used as a precursor of graphene composites because it is economical to fabricate on a large scale and easy to process.29
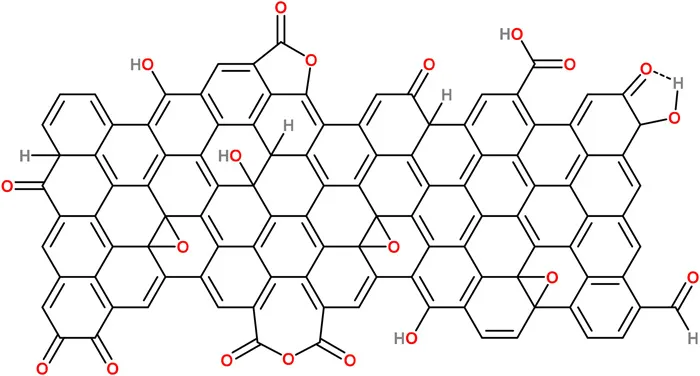
Today GO is produced in large quantities through chemical exfoliation of graphite, known as the Hummers' method.30,31 The technique is based on the principle of oxidizing graphite by treatment in a mixture of strong acids (e.g. H2SO4) and oxidizing agents (e.g., NaNO2, KMnO4) for 2 hours at 45 °C. Then the reaction mixture is washed in an ice-bath with H2O2 to remove the residual KMnO4. This process introduces several oxygen functional groups such as epoxides, hydroxyl groups, and carboxylic acid, among others (Figure 1.2),32 making GO hydrophilic and easily dispersible in water via ultra-sonication. The oxidized carbons (i.e., sp3 carbons) and lattice defects of GO can be later eliminated by a reduction step (e.g., chemical, or thermal) to recover the graphene-like properties (i.e., sp2 carbons).
Figure 1.2Surface groups on graphene oxide.
However, like any other carbon allotrope, graphene as a bulk material has a strong propensity to form irretrievable agglomerates due to strong π–π interactions among individual graphene sheets. This leads to inadequate exploitation of isolated graphene layers for practical applications. In order to overcome this problem, the integration of 2D graphene nanosheets into 3D macrostructures, and ultimately into a functional system, such as aerogels has been recognized as a progressively critical approach during the past five years. Their intensively interconnected networks, enormous surface area, intense porosity, remarkable sturdiness, and superior graphene building blocks endow a plethora of exciting features that make them extremely suitable for a broad range of clean energy and environmental applications. However, in order to truly harness their potential, one must understand how the design and assembly of these 3D graphene networks impact their final properties. In this chapter, we explore the various types of graphene-based aerogels reported to date and how their architecture impacts their ultimate performance.
1.2 Graphene Aerogels
Graphene aerogels (GA) were first synthesized in 2010 by two independent groups, Xu et al.33 and Worsley et al.34 Both groups used GO as the precursor but different gelation processes (e.g., covalent vs. noncovalent). Here we describe the different methods to synthesize GAs.
1.2.1 Sol–Gel Hydrogels, Freeze-drying, Gelation Methods
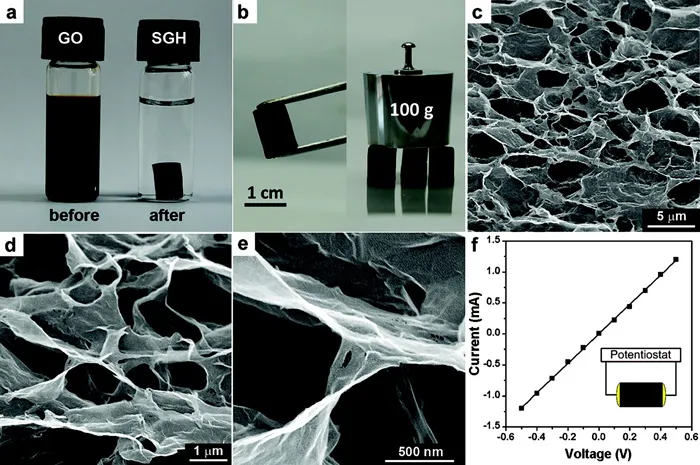
Xu and co-workers reported a hydrothermal route involving a 180 °C treatment in a pressure vessel for 12 hours to simultaneously reduce and gel an aqueous GO suspension.33 For the hydrothermal route, gelation occurs upon reduction because the electrostatic repulsion is eliminated due to loss of oxygen functionalities in GO. Local regions on the GO sheets then become hydrophobic and are prone to noncovalent bonding (e.g., π–π stacking) with reduced regions on nearby sheets forming a physically crosslinked gel. The properties of the aerogel achieved by freeze-drying were highly correlated to the starting GO concentration of the suspension. If the concentration was below 1 mg mL−1, no gel was formed. The duration of the hydrothermal treatment also proved to be significant in determining the properties of the aerogel. Both the maximum pressure and treatment time determined the degree of reduction, which impacted density, conductivity, and crosslinking (Figure 1.3).33
Figure 1.3(a) Photographs of a 2 mg mL−1 homogeneous GO aqueous dispersion before and after hydr...