Two-dimensional (2D) materials for photocatalytic applications have attracted attention in recent years due to their unique thickness-dependent physiochemical properties. 2D materials offer enhanced functionality over traditional three-dimensional (3D) photocatalysts due to modified chemical composition and electronic structures, as well as abundant surface active sites. This book reviews the applications of 2D-related nano-materials in solar-driven catalysis, providing an up-to-date introduction to the design and use of 2D-related photo(electro)catalysts. This includes not only application areas such as fine chemicals synthesis, water splitting, CO2 reduction, and N2 fixation, but also catalyst design and preparation. Some typical 2D and 2D-related materials (such as layered double hydroxides (LDHs), layered metal oxides, transition metal dichalcogenide (TMDs), metal–organic frameworks (MOFs), graphene, g-C3N4, etc.) are classified, and relationships between structure and property are demonstrated, with emphasis on how to improve 2D-related materials performance for practical applications. While the focus of this book will primarily be on experimental studies, computational results will serve as a necessary reference. With chapters written by expert researchers in their fields, Photocatalysis Using 2D Nanomaterials will provide advanced undergraduates, postgraduates and other researchers convenient introductions to these topics.

- 304 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Photocatalysis Using 2D Nanomaterials
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Chapter 1
Recent Developments and Perspectives on Solar-driven Fine Chemicals Synthesis: From the Reaction System to 2D Photocatalysts
a State Key Laboratory of Chemical Resource Engineering, Beijing University of Chemical Technology, 100029 Beijing, P. R. Chinssa;
b Department of Chemistry, Tsinghua University, 100084 Beijing, P. R. China Email:[email protected];[email protected]
1.1 Introduction
Fine chemicals are complex, single and pure chemicals produced in limited quantities by a multi-step batch chemical or biological process (from Wikipedia, 2021, https://en.wikipedia.org/wiki/Fine_chemical). In most cases, the production of these fine chemicals is limited (<1000 tons per year)under strict specifications, leading to high prices (>$10 kg−1). These high value-added products can be used as the starting materials for speciality chemicals such as agrochemicals and pharmaceuticals (Figure 1.1). Fine chemicals have become a significant part of the chemical industry since the late 1970s. However, so far, most of their production on the industrial scale is operated under high temperature and high pressure, resulting in environmental pollution and high energy consumption. In this regard, photo(electro)catalysis enables a more sustainable and economical approach by virtue of the solar energy used. At present, many photocatalytic reaction systems using semiconductors have been proven to be feasible under mild conditions. Simple processes, green reaction conditions and renewable solar energy bring promising prospects for photocatalyst applications in CO2 reduction, water splitting to produce clean H2, fine chemicals synthesis, etc. However, the low conversion and selectivity still hinder the popularisation and the industrial application of photocatalytic technology.1 In recent years, great progress has been made in heterogeneous photocatalyst synthesis for enhanced catalytic activity under visible light irradiation.2 By manipulating the morphology of heterogeneous catalysts, nanoscale photocatalysts with higher intrinsic activity, larger specific surface areas and more active sites can be synthesised. Among them, two-dimensional (2D) nanomaterials have attracted special research interest due to their unique structural advantages and excellent photocatalytic performance.
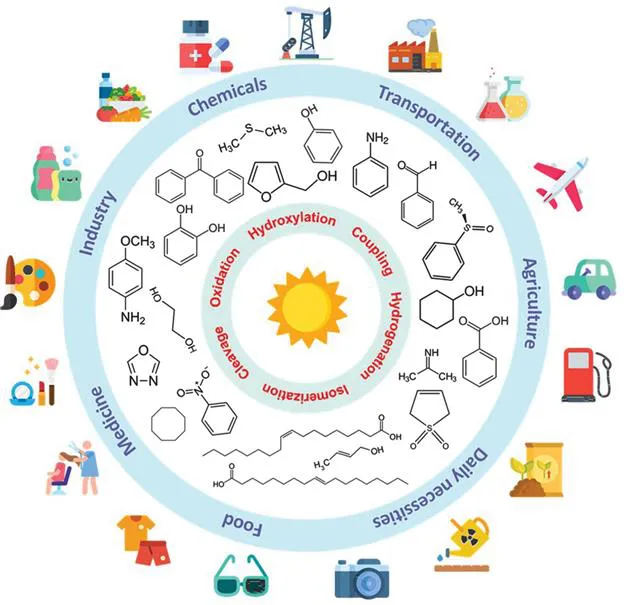
Figure 1.1 Reactions to synthesise fine chemicals and their corresponding applications in industry and modern life. Icons made by Freepik, Smashicons, Pixel perfect, Flat Icons and photo3idea_studio from www.flaticon.com.
2D nanomaterials can be nanoscale thick or even as thin as one atom.3 The family of 2D nanomaterials include graphene,4–8 graphitic carbon nitride (g-C3N4),9–11 transition metal dichalcogenides (TMDs),12–15 layered double hydroxides (LDHs),16,17 black phosphorus (BP),18,19 MXenes,20 hexagonal boron nitride (h-BN),21–23 perovskites,24–27 metal–organic frameworks (MOFs),28–30 covalent–organic frameworks (COFs)31 and other sheet-like materials32–41 (Figure 1.2). Because electrons are confined to a two-dimensional scale, 2D materials have unique physical and chemical properties, such as excellent electrical conductivities, carrier mobility, etc.42 The nanosheet structure and atomic-level thickness endow 2D materials with high specific surface area and full exposure of the surface active sites, thus showing high catalytic performance in photo(electro)catalysis such as selective oxidation of alcohols, hydrogenation, and C–C and C–N coupling that will be discussed later.43
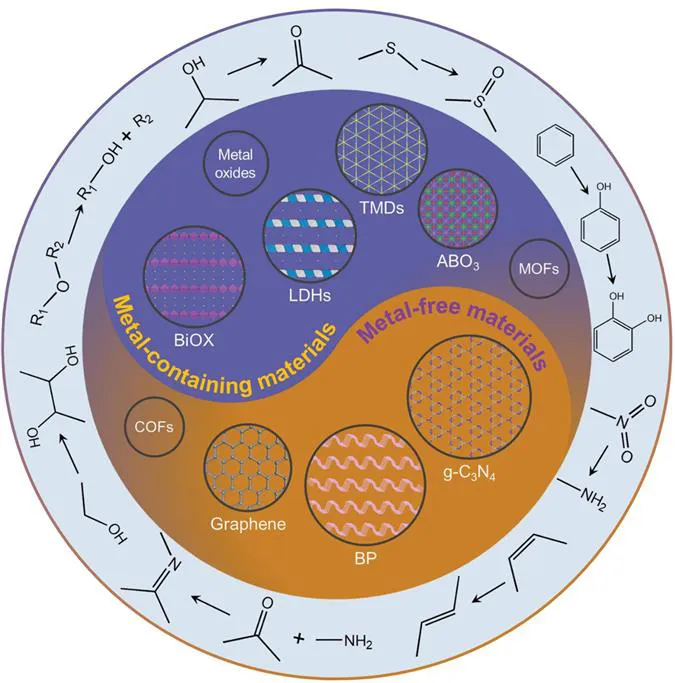
Figure 1.2 Types of 2D materials and crystal structures of several typical 2D materials for efficient fine chemicals synthesis.
Among the metal-free 2D materials, g-C3N4 has the advantages of low cost, easy synthesis, unique band structure and broad adsorption in the visible light range. As a visible-light-responsive photocatalyst, it has been widely studied in the field of photocatalysis.44 To enhance its photocatalytic performance, researchers have been modifying and optimising g-C3N4 through element doping,45,46 morphology regulation47,48 and heterogeneous junction construction.49,50 Different from g-C3N4 and graphene, BP has a wide band response and shows a strong light conduction efficiency. BP is a natural p-type semiconductor with a direct bandgap, which can be adjusted by changing the number of stacked phosphorus layers, and has significant anisotropy. Among the two-dimensional materials, BP has high electron mobility, and its optical and photoelectric properties have greater advantages for further photocatalysis.51–53
Layered double hydroxides (LDHs) are a kind of layered clay material, and their structure can be optimised by adjusting the type and proportion of metal elements for enhanced catalytic performance. Through the calcination strategy, LDHs can be further transformed into mixed-metal oxides (MMO) or metal-supported materials, which expose more active sites and improve catalytic performance.54,55 Similarly, TMDs,56,57 bismuth oxyhalides (BiOX),58,59 perovskite (ABO3)60,61 and metal-containing layered materials62–65 are composed of polyhedral coordination units of metallic and non-metallic elements. They can be modified by controlling the morphology, elements and crystal surface, and constructing a heterojunction to improve the exposure of active elements or defects, enhance the light absorption capacity of materials and the separation and transfer efficiency of photogenerated carriers, change the adsorption ability of reactants and products, and improve the conversion rate and selectivity of photocatalytic reactions. Herein, this review summarises the recent development of 2D materials in the photocatalytic synthesis of fine chemicals and biomass valorisation, such as selective oxidation, C–C/C–N coupling, hydrogenation, cis–trans isomerisation and C–C/C–O cleavage, etc. In order to facilitate the reader's understanding, a brief history of the development of some common fine chemicals synthesis reactions is also provided.
1.2 Selective Oxidation Reactions
Oxidation of alcohol liquids is one of the most common and key organic reactions, and the corresponding aldehydes, ketones or carboxylic acids produced by it are widely used in large-scale industrial chemical production and fine chemicals production (Figure 1.3). In the oxidation products of alcohols, the value of aldehydes or ketones is higher than that of carboxylic acids. Therefore, controlling the degree of oxidation reaction and avoiding over-oxidation products (such as CO2) is the key to preparing high value-added products of selectively oxidised alcohols. The selective oxidation of alcohols discussed in this section refers to the oxidation of alcohols in liquid form.
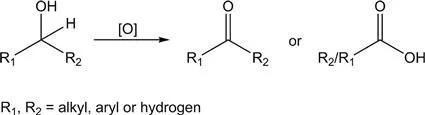
Figure 1.3 Selective oxidation of alcohols to aldehydes, ketones, or carboxylic acids.
1.2.1 Selective Oxidation of Alcohols
1.2.1.1 The Development of Selective Oxidation of Alcohols
In early organic synthesis, the oxidation reaction mostly used K[MnO4], K2[Cr2O7] and some other oxidants. However, these oxidants always suffer from strong oxidation ability, poor selectivity and serious pollution, which limit their application scope. Catalytic oxidation has made it possible to use cheap and readily available oxidants such as air, O2, or H2O2. These green oxidants improve the atomic economy of oxidation reaction and make the oxidation of alcohol more environmentally friendly and efficient. Due to the advantages of low cost, high yield, good selectivity and low pollution, catalytic oxidation technology using these oxidants has gradually become the mainstream of alcohol selective oxidation research.
As an important medium for reactions and post-treatment, organic solvents have been widely used. Most of these solvents (such as trifluorotoluene, toluene, acetonitrile) are volatile and cause soil, water and air pollution when they escape into the environment, causing great harm to the human body. Therefore, it is highly desirable to choose non-toxic and harmless solvents and develop solv...
Table of contents
- Cover
- Title
- Copyright
- Contents
- Chapter 1 Recent Developments and Perspectives on Solar-driven Fine Chemicals Synthesis: From the Reaction System to 2D Photocatalysts
- Chapter 2 Opportunities for Ultrathin 2D Catalysts in Promoting CO2 Photoreduction
- Chapter 3 Photocatalysis by Graphenes
- Chapter 4 2D Inorganic Nanosheet-based Hybrid Photocatalysts for Water Splitting
- Chapter 5 2D Photocatalytic Materials for Environmental Applications
- Subject Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Photocatalysis Using 2D Nanomaterials by Yufei Zhao, Haohong Duan, Yufei Zhao,Haohong Duan in PDF and/or ePUB format, as well as other popular books in Physical Sciences & Physical & Theoretical Chemistry. We have over 1.5 million books available in our catalogue for you to explore.