The development of therapy with anti-angiogenics or vascular endothelial growth factor inhibitors (anti-VEGF) has marked the beginning of a new era in neovascularization and macular edema treatment. Its main goals are the inhibition of growth and development of new vessels along with the reduction of vascular permeability. The advantages over conventional laser photocoagulation are evident as laser treatment always leaves scars and causes a retinal sensitivity deterioration.Starting with an outline of treatment principles, this volume covers all aspects of anti-VEGF therapy for ophthalmological disorders. In particular, specific chapters are dedicated to age-related macular degeneration, degenerative myopia, angioid streaks, inflammatory diseases, hereditary dystrophies, retinal vein occlusion, diabetic retinopathy, ocular tumors, as well as anterior segment neovascularization.The book gives an update on the application of anti-VEGF in ocular diseases to general ophthalmologists as well as retina specialists.

eBook - ePub
Anti-VEGF
- 154 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
MedicineSubtopic
Opthalmology & OptometryBandello F, Battaglia Parodi M (eds): Anti-VEGF.
Dev Ophthalmol. Basel, Karger, 2010, vol 46, pp 4-20
Dev Ophthalmol. Basel, Karger, 2010, vol 46, pp 4-20
______________________
Mechanisms of Ocular Angiogenesis and Its Molecular Mediators
Martin J. Siemerinka · Albert J. Augustinb · Reinier O. Schlingemanna,c
aOcular Angiogenesis Group, Department of Ophthalmology, Academic Medical Center, Amsterdam, The Netherlands; bAugenklinik, Karlsruhe, Germany, and cNetherlands Institute for Neuroscience, Royal Netherlands Academy of Arts and Sciences, Amsterdam, The Netherlands
______________________
Abstract
Angiogenesis is defined as the formation of new blood vessels from the existing vasculature. It is a highly coordinated process occurring during development of the retinal vasculature, ocular wound healing, and in pathological conditions. Complex interactions are involved between non-vascular and microvascular cells, such as endothelial cells and pericytes, via several angiogenic growth factors and inhibitors. Of these growth factors, vascular endothelial growth factor (VEGF) has emerged as the single most important causal agent of angiogenesis in health and disease in the eye. During the angiogenic process, endothelial cells shift from a homogeneous quiescent population into a population of heterogeneous phenotypes, each with a distinct cellular fate. So far, three angiogenic specialized phenotypes have been identified: (1) ‘tip cells’, which pick up guidance signals and migrate through the extracellular matrix; (2) ‘stalk cells’, which proliferate, form junctions, produce extracellular matrix, and form a lumen, and (3)‘phalanx cells’, which do not proliferate, but align and form a smooth monolayer. Eventually, a robust mature new blood vessel is formed which is capable of supplying blood and oxygen to tissues. Pathological angiogenesis is a key component of several irreversible causes of blindness. In most of these conditions, angiogenesis is part of a wound healing response culminating, via an angiofibrotic switch, in fibrosis and scar formation which leads to blindness. Currently, VEGF-A antagonists are standard care in the treatment of exudative age-related macular degeneration, and have been found to be a valuable additional treatment strategy in several other vascular retinal diseases.
Copyright © 2010 S. Karger AG, Basel
Blood vessels form an intricate hollow network of arteries, capillaries, and veins for the transport of liquids, solutes, gases, macromolecules, and cells throughout the vertebrate body. The vascular network is formed during early stages of development, and its correct and early function is absolutely critical for survival of the embryo. New blood vessels originate from endothelial precursor cells (angioblasts) by a process called vasculogenesis or from preexisting blood vessels by angiogenesis [1, 2]. Once a functional adult vascular system has been formed completely, blood vessels become quiescent. The growth potential of smaller blood vessels, however, is retained and is employed during wound healing and tissue regeneration.
Beyond its physiological roles, angiogenesis is also a hallmark of many pathological conditions, including neovascular diseases in the eye [3-5]. Excessive angiogenesis occurs when diseased cells produce abnormal amounts of angiogenic factors, overwhelming the effects of natural angiogenesis inhibitors. As the newly formed vessels mainly serve a role in a wound healing response, they usually do not restore the tissue integrity, but rather cause visual impairment when they are located in normally avascular, transparent tissues such as the cornea and vitreous. Strategies for inhibition of angiogenesis include approaches that can block the angiogenesis cascade at several steps [4, 6].
Angiogenesis: Mechanisms and Molecular Mediators
Endothelial Cell Differentiation
All blood vessels are lined by endothelial cells (ECs), which form the interface between circulating blood in the lumen and the rest of the vessel wall. Under normal conditions, ECs are a remarkably quiescent cell type, undergoing division approximately once every 1,000 days, but when activated, cell division can occur every 1-2 days [7]. Sprouting angiogenesis requires selection of ECs from an existing blood vessel which will be activated to form the new vessel, while at the same time, surrounding ECs remain quiescent in their current position. From recent studies a model has emerged in which ECs differentiate into three specialized cell types with distinct phenotypes during angiogenesis (fig. 1) [8-10]. First, a single ‘tip cell’ develops. This EC breaks down the basal lamina, emerges from its parent blood vessel and becomes the leading cell of the sprouting vessel. The tip cell migrates into the extracellular matrix and senses microenvironmental attractive and repulsive signals for guidance. Secondly, following directly behind the migrating tip cell, other ECs differentiate under the influence of the adjacent tip cell into ‘stalk cells’ that proliferate and bridge the gap between the tip cell and the parent vasculature. Stalk cells generate the blood vessel lumen through the formation of intracellular vacuoles, a process called ‘lumenogenesis’. Thirdly, ECs behind the stalk cells differentiate into ‘phalanx cells’, and align in a smooth cobblestone monolayer, becoming the most inner cell layer in the new blood vessel. Phalanx cells no longer proliferate, express tight junctions and make contact with mural cells.
Angiogenesis Inducers and Inhibitors
Angiogenesis is tightly controlled by closely interacting angiogenic and angiostatic factors, and their balance ultimately determines if, where and when the ‘angiogenic switch’ is turned on with angiogenesis as the result [2, 9]. Over the past decades, numerous inducers of angiogenesis have been identified, including the members of the vascular endothelial growth factor (VEGF) family, angiopoietins, transforming growth factors (TGF), epidermal growth factor (EGF), platelet-derived growth factor (PDGF), tumor necrosis factor-α (TNF-α), insulin-like growth factor (IGF), vascular endothelial-cadherin (VE-cadherin), interleukins and the members of the fibroblast growth factor (FGF) family. In addition, there is a plethora of growth factors, hormones and metabolites that have been reported to directly or indirectly stimulate physiological and pathological angiogenesis (table 1) [11, 12]. Not all of these factors are specific for ECs. Consistent with a major role for hypoxia in the overall process of angiogenesis, a large number of angiogenic factors involved in various stages of angiogenesis are independently responsive to hypoxia [13]. The VEGF family of proteins is the most important family of angiogenic factors that controls blood vessel formation.
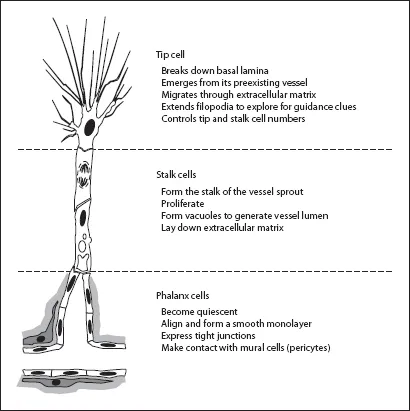
Fig. 1. Representative model of sprouting angiogenesis. At least three different angiogenic specialized endothelial cells (white) are required, each with ...
Table of contents
- Cover Page
- Front Matter
- Angiostatic and Angiogenic Factors
- Mechanisms of Ocular Angiogenesis and Its Molecular Mediators
- Antivascular Endothelial Growth Factors in Age-Related Macular Degeneration
- Antivascular Endothelial Growth Factor in Diabetic Retinopathy
- Retinal Vein Occlusions
- Antivascular Endothelial Growth Factor for Choroidal Neovascularization in Pathologic Myopia
- Antivascular Endothelial Growth Factors for Inflammatory Chorioretinal Disorders
- Antivascular Endothelial Growth Factor Treatment in Pseudoxanthoma Elasticum Patients
- Antivascular Endothelial Growth Factor in Hereditary Dystrophies
- Antivascular Endothelial Growth Factor as an Approach for Macular Edema
- Angiogenesis and Vascular Endothelial Growth Factors in Intraocular Tumors
- Antivascular Endothelial Growth Factors in Anterior Segment Diseases
- Author Index
- Subject Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Anti-VEGF by F. Bandello,M. Battaglia Parodi,A. J. Augustin,P. Iacono,R. O. Schlingemann,U. Schmidt-Erfurth,F. D. Verbraak, F. Bandello in PDF and/or ePUB format, as well as other popular books in Medicine & Opthalmology & Optometry. We have over 1.5 million books available in our catalogue for you to explore.